Abstract
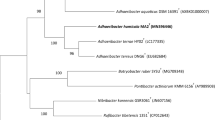
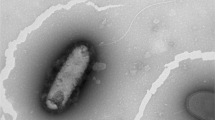
Two motile, rod-shaped, Gram-stain-negative bacterial strains, TNT11T and TNT19T, were isolated from soil samples collected at Deception Island, Antarctica. According to the 16S rRNA gene sequence similarity, both strains belong to the genus Pseudomonas. Further genomic analyses based on ANI and dDDH suggested that these strains were new species. Growth of strain TNT11T is observed at 0–30 ℃ (optimum, 20 ℃), pH 4.0–9.0 (optimum, pH 6.0) and in the presence of 0–5.0% NaCl (optimum, 1% NaCl), while for TNT19T is observed at 0–30 ℃ (optimum between 15 and 20 ℃), pH 5.0–9.0 (optimum, pH 6.0) and in the presence of 0–5.0% NaCl (optimum between 0 and 1% NaCl). The fatty acid profile consists of the major compounds; C16:0 and C16:1 ω6 for TNT11T, and C16:0 and C12:0 for TNT19T. Based on the draft genome sequences, the DNA G + C content for TNT11T is 60.43 mol% and 58.60 mol% for TNT19T. Based on this polyphasic study, TNT11T and TNT19T represent two novel species of the genus Pseudomonas, for which the proposed names are Pseudomonas violetae sp. nov. and Pseudomonas emilianonis sp. nov., respectively. The type strains are Pseudomonas violetae TNT11T (= RGM 3443T = LMG 32959T) and Pseudomonas emilianonis TNT19T (= RGM 3442T = LMG 32960T). Strains TNT11T and TNT19T were deposited to CChRGM and BCCM/LMG with entry numbers RGM 3443/LMG 32959 and RGM 3442/LMG 32960, respectively.


Similar content being viewed by others
Data availability
The data sets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request unless otherwise stated.
Abbreviations
- ANI:
-
Average nucleotide identity
- dDDH:
-
Digital DNA–DNA hybridization
- TNT:
-
Trinitrotoluene
References
Arkin AP, Cottingham RW, Henry CS, Harris NL, Stevens RL, Maslov S, Dehal P, Ware D, Perez F, Canon S, Sneddon MW, Henderson ML, Riehl WJ, Murphy-Olson D, Chan SY, Kamimura RT, Kumari S, Drake MM, Brettin TS, Yu D (2018) KBase: the United States department of energy systems biology knowledgebase. Nat Biotechnol 36(7):566–569. https://doi.org/10.1038/nbt.4163
Arnau VG, Sánchez LA, Delgado OD (2015) Pseudomonas yamanorum sp. Nov., a psychrotolerant bacterium isolated from a subantarctic environment. Int J Syst Evol Microbiol. 65(2):424–431. https://doi.org/10.1099/ijs.0.065201-0
Aziz RK, Bartels D, Best AA, DeJongh M, Disz T, Edwards RA, Formsma K, Gerdes S, Glass EM, Kubal M, Meyer F, Olsen GJ, Olson R, Osterman AL, Overbeek RA, McNeil LK, Paarmann D, Paczian T, Parrello B, Zagnitko O (2008) The RAST server: rapid annotations using subsystems technology. BMC Genomics 9(1):75. https://doi.org/10.1186/1471-2164-9-75
Baıda N, Yazourh A, Singer E, Izard D (2001) Pseudomonas brenneri sp. Nov., a new species isolated from natural mineral waters. Res Microbiol. 152(5):493–502. https://doi.org/10.1016/S0923-2508(01)01223-2
Bankevich A, Nurk S, Antipov D, Gurevich AA, Dvorkin M, Kulikov AS, Lesin VM, Nikolenko SI, Pham S, Prjibelski AD (2012) SPAdes: a new genome assembly algorithm and its applications to single-cell sequencing. J Comput Biol 19:455–477
Barrow GI, Feltham RKA (1993) Cowan and steel’s manual for the identification of medical bacteria, 3rd edn. Cambridge University Press, Cambridge. https://doi.org/10.1017/CBO9780511527104
Bligh EG, Dyer WJ (1959) A rapid method of total lipid extraction and purification. Can J Biochem Physiol 37(8):911–917. https://doi.org/10.1139/o59-099
Bozal N, Montes MJ, Mercade E (2007) Pseudomonas guineae sp Nov, a novel psychrotolerant bacterium from an Antarctic environment. Int J Syst Evol Microbiol. 57(11):2609–2612
Bushnell B (2014) BBMap: a fast, accurate, splice-aware aligner. Report Number: LBNL-7065E. https://www.osti.gov/biblio/1241166. Accessed 17 Mar 2023
Cabrera Ma Á, Márquez SL, Quezada CP, Osorio MI, Castro-Nallar E, González-Nilo FD, Pérez-Donoso JM (2020) Biotransformation of 2,4,6-Trinitrotoluene by Pseudomonas sp. TNT3 isolated from Deception Island Antarctica. Environ Pollut. 262:113922. https://doi.org/10.1016/j.envpol.2020.113922
Cabrera Ma. Á, Márquez SL, Pérez-Donoso JM (2022) Comparative genomic analysis of antarctic pseudomonas isolates with 2,4,6-trinitrotoluene transformation capabilities reveals their unique features for xenobiotics degradation. Genes 13(8):1354. https://doi.org/10.3390/genes13081354
Carrasco, V., Amarelle, V., Lagos-Moraga, S., Quezada, C. P., Espinoza-González, R., Faccio, R., Fabiano, E., and Pérez-Donoso, J. M. (2021). Production of cadmium sulfide quantum dots by the lithobiontic Antarctic strain Pedobacter sp. UYP1 and their application as photosensitizer in solar cells. Microbial Cell Factories, 20(1), 1–10.
Carrion O, Minana-Galbis D, Montes MJ, Mercade E (2011) Pseudomonas deceptionensis sp. Nov., a psychrotolerant bacterium from the Antarctic. Int J Syst Evol Microbiol 61:2401–2405
Dietz-Vargas C, Valenzuela-Ibaceta F, Carrasco V, Pérez-Donoso JM (2023) Solid medium for the direct isolation of bacterial colonies growing with polycyclic aromatic hydrocarbons or 2,4,6-trinitrotoluene (TNT). Arch Microbiol 205(7):271. https://doi.org/10.1007/s00203-023-03610-z
Duman M, Mulet M, Altun S, Saticioglu IB, Gomila M, Lalucat J, Garcia-Valdes E (2019) Pseudomonas piscium sp. Nov., ., Pseudomonas pisciculturae sp. Nov., Pseudomonas mucoides sp. Nov. and Pseudomonas neuropathica sp. Nov. Isolated from rainbow trout. Int J Syst Evol Microbiol. https://doi.org/10.1099/ijsem.0.004714
Elomari M, Coroler L, Hoste B, Gillis M, Izard D, Leclerc H (1996) DNA relatedness among pseudomonas strains isolated from natural mineral waters and proposal of Pseudomonas veronii sp. Nov. Int J Syst Bacteriol 46(4):1138–1144. https://doi.org/10.1099/00207713-46-4-1138
Farris JS (1972) Estimating phylogenetic trees from distance matrices. Am Nat 106(951):645–668
Felsenstein J (1985) Confidence limits on phylogenies: a justification. Evolution 39:783–791
Fulco AJ (1983) Fatty acid metabolism in bacteria. Prog Lipid Res 22(2):133–160. https://doi.org/10.1016/0163-7827(83)90005-X
Goris J, Konstantinidis KT, Klappenbach JA, Coenye T, Vandamme P, Tiedje JM (2007) DNA–DNA hybridization values and their relationship to whole-genome sequence similarities. Int J Syst Evol Microbiol 57(1):81–91. https://doi.org/10.1099/ijs.0.64483-0
Jang GI, Lee I, Ha TT, Yoon SJ, Hwang YJ, Yi H, Yun S, Lee WS, Hwang CY (2020) Pseudomonas neustonica sp. Nov, isolated from the sea surface microlayer of the Ross Sea (Antarctica). Int J Syst Evol Microbiol. 70(6):3832–3838
Kearse M, Moir R, Wilson A, Stones-Havas S, Cheung M, Sturrock S, Buxton S, Cooper A, Markowitz S, Duran C (2012) Geneious basic: an integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics 28(12):1647–1649
Kerr KG, Snelling AM (2009) Pseudomonas aeruginosa: a formidable and ever-present adversary. J Hosp Infect 73(4):338–344. https://doi.org/10.1016/j.jhin.2009.04.020
Kimura M (1980) A simple method for estimating evolutionary rates of base substitutions through comparative studies of nucleotide sequences. J Mol Evol 16(2):111–120. https://doi.org/10.1007/BF01731581
Köfeler HC (2016) Hydroxy fatty acid. In: Wenk MR (ed) Encyclopedia of lipidomics. Springer, Cham, pp 1–2. https://doi.org/10.1007/978-94-007-7864-1_22-1
Kosina M, Barták M, Mašlaňová I, Pascutti AV, Šedo O, Lexa M, Sedláček I (2013) Pseudomonas prosekii sp. Nov., a novel psychrotrophic bacterium from Antarctica. Curr Microbiol 67:637–646
Kosina M, Švec P, Černohlávková J, Barták M, Snopková K, De Vos P, Sedláček I (2016) Description of Pseudomonas gregormendelii sp. Nov., a novel psychrotrophic bacterium from James Ross Island. Antarctica. Curr Microbiol. 73(1):84–90. https://doi.org/10.1007/s00284-016-1029-5
Kumar S, Stecher G, Tamura K (2016) MEGA7: molecular evolutionary genetics analysis version 7.0 for bigger datasets. Mol Biol evol 33(7):1870–1874
Lefort V, Desper R, Gascuel O (2015) FastME 2.0: A comprehensive, accurate, and fast distance-based phylogeny inference program. Mol Biol Evol. 32(10):2798–2800
Li Y, Wu S, Wang L, Li Y, Shi F, Wang X (2010) Differentiation of bacteria using fatty acid profiles from gas chromatography–tandem mass spectrometry. J Sci Food Agric 90(8):1380–1383
López NI, Pettinari MJ, Stackebrandt E, Tribelli PM, Põtter M, Steinbüchel A, Méndez BS (2009) Pseudomonas extremaustralis sp. Nov., a Poly(3-hydroxybutyrate) producer isolated from an Antarctic environment. Curr Microbiol. 59(5):514–519. https://doi.org/10.1007/s00284-009-9469-9
Meier-Kolthoff JP, Göker M (2019) TYGS is an automated high-throughput platform for state-of-the-art genome-based taxonomy. Nat Commun 10(1):2182
Meier-Kolthoff JP, Auch AF, Klenk H-P, Göker M (2013) Genome sequence-based species delimitation with confidence intervals and improved distance functions. BMC Bioinformatics 14:1–14
Mező E, Hartmann-Balogh F, Madarászné Horváth I, Bufa A, Marosvölgyi T, Kocsis B, Makszin L (2022) Effect of culture conditions on fatty acid profiles of bacteria and lipopolysaccharides of the genus pseudomonas—GC-MS analysis on ionic liquid-based column. Molecules 27(20):6930. https://doi.org/10.3390/molecules27206930
Migula W (1895) Über ein neues System der Bakterien. Arb bakt Inst Karlsruhe 1:235–238
Nováková D, Koublová V, Sedlář K, Staňková E, Králová S, Švec P, Neumann-Schaal M, Wolf J, Koudelková S, Barták M (2023) Pseudomonas petrae sp. Nov. Isolated from regolith samples in Antarctica. Syst Appl Microbiol. 46(4):126424
Pavlov MS, Lira F, Martinez JL, Olivares-Pacheco J, Marshall SH (2020) Pseudomonas fildesensis sp. Nov., a psychrotolerant bacterium isolated from Antarctic soil of King George Island, South Shetland Islands. Int J Syst Evol Microbiol. 70(5):3255–3263. https://doi.org/10.1099/ijsem.0.004165
Poger D, Mark AE (2015) A ring to rule them all: the effect of cyclopropane fatty acids on the fluidity of lipid bilayers. J Phys Chem B 119(17):5487–5495. https://doi.org/10.1021/acs.jpcb.5b00958
Reasoner DJ, Geldreich EE (1985) A new medium for the enumeration and subculture of bacteria from potable water. Appl Environ Microbiol 49(1):1–7. https://doi.org/10.1128/aem.49.1.1-7.1985
Reddy GS, Matsumoto GI, Schumann P, Stackebrandt E, Shivaji S (2004) Psychrophilic pseudomonads from Antarctica: Pseudomonas antarctica sp. Nov., Pseudomonas meridiana sp Nov And Pseudomonas proteolytica sp Nov. Int J Syst Evol Microbiol 54(3):713–719
Rodriguez-R L, Konstantinidis KT (2016) The enveomics collection: a toolbox for specialized analyses of microbial genomes and metagenomes. PeerJ Preprints. 4:e1900v1
Saitou N, Nei M (1987) The neighbor-joining method: a new method for reconstructing phylogenetic trees. Mol Biol Evol. https://doi.org/10.1093/oxfordjournals.molbev.a040454
Sawada H, Fujikawa T, Satou M (2022) Pseudomonas aegrilactucae sp. Nov. and Pseudomonas morbosilactucae sp. Nov., pathogens causing bacterial rot of lettuce in Japan. Int J Syst Evol Microbiol. https://doi.org/10.1099/ijsem.0.005599
Serrano-González MY, Chandra R, Castillo-Zacarias C, Robledo-Padilla F, Rostro-Alanis MDJ, Parra-Saldivar R (2018) Biotransformation and degradation of 2,4,6-trinitrotoluene by microbial metabolism and their interaction. Defence Technol 14(2):151–164. https://doi.org/10.1016/j.dt.2018.01.004
Tindall BJ, Sikorski J, Smibert RA, Krieg NR (2007) Phenotypic characterization and the principles of comparative systematics. In: Methods for General and Molecular Microbiology, American Society of Microbiology, 3rd edn, pp 330–393
Van Dillewijn P, Wittich R-M, Caballero A, Ramos J-L (2008) Subfunctionality of hydride transferases of the old yellow enzyme family of flavoproteins of Pseudomonas putida. Appl Environ Microbiol 74(21):6703–6708. https://doi.org/10.1128/AEM.00386-08
Vásquez-Ponce F, Higuera-Llantén S, Pavlov MS, Marshall SH, Olivares-Pacheco J (2018) Phylogenetic MLSA and phenotypic analysis identification of three probable novel Pseudomonas species isolated on King George Island, South Shetland, Antarctica. Braz J Microbiol 49:695–702
Velly H, Bouix M, Passot S, Penicaud C, Beinsteiner H, Ghorbal S, Lieben P, Fonseca F (2015) Cyclopropanation of unsaturated fatty acids and membrane rigidification improve the freeze-drying resistance of Lactococcus lactis subsp. Lactis TOMSC161. Appl Microbiol Biotechnol. 99(2):907–918. https://doi.org/10.1007/s00253-014-6152-2
Wayne L, Brenner D, Colwell R, Grimont P, Kandler O, Krichevsky M, Moore L, Moore W, Murray R, Stackebrandt E (1987) Report of the ad hoc committee on reconciliation of approaches to bacterial systematics. Int J Syst Evol Microbiol 37(4):463–464
Wells T, Ragauskas AJ (2012) Biotechnological opportunities with the β-ketoadipate pathway. Trends Biotechnol 30(12):627–637. https://doi.org/10.1016/j.tibtech.2012.09.008
Yang Y, Guo Y (2018) Elucidating the molecular mechanisms mediating plant salt-stress responses. New Phytol 217(2):523–539. https://doi.org/10.1111/nph.14920
Yoon S-H, Ha S-M, Kwon S, Lim J, Kim Y, Seo H, Chun J (2017) Introducing EzBioCloud: a taxonomically united database of 16S rRNA gene sequences and whole-genome assemblies. Int J Syst Evol Microbiol 67(5):1613
Yu Y, Li C, Liu J, Zhu F, Wei S, Huang Y, Huang X, Qin Q (2020) Palmitic acid promotes virus replication in fish cell by modulating autophagy flux and TBK1-IRF3/7 pathway. Front Immunol 11:1764. https://doi.org/10.3389/fimmu.2020.01764
Funding
This study was financially supported by Fondecyt 1200870 (J.M.P-D.), DGI-UNAB DI-03-23/INI (VC), INACH DG_13-22 (V.C.), and ANID PIA Anillo INACH ACT192057 (J.M.P-D.).
Author information
Authors and Affiliations
Contributions
VC: conceptualization, methodology, validation, investigation, writing—original draft, writing—review and editing, visualization. DMR: conceptualization, methodology, validation, investigation, writing—original draft, writing—review and editing, visualization. FV-I: conceptualization, methodology, validation, investigation, writing—original draft, writing—review and editing, visualization. SL-M: conceptualization, methodology, validation, investigation, writing—original draft, writing—review and editing, visualization. CD-V: methodology, validation, writing—original draft, writing—review and editing, visualization. RJM: conceptualization, investigation, resources, writing—original draft, writing—review and editing, supervision. JMP-D: conceptualization, investigation, resources, writing—original draft, writing—review and editing, supervision, project administration, funding acquisition.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Additional information
Communicated by Yusuf Akhter.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The GenBank/EMBL/DDBJ accession number for the 16S rRNA gene sequence of strains TNT11T and TNT19T are OP846032.1 and OP846033.1, respectively. The Whole Genome Shotgun project has been deposited at DDBJ/ENA/GenBank under the accession NZ_JAKNRV000000000.1 (TNT11T) and NZ_JAKNRW000000000.1 (TNT19T).
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Carrasco, V., Roldán, D.M., Valenzuela-Ibaceta, F. et al. Pseudomonas violetae sp. nov. and Pseudomonas emilianonis sp. nov., two new species with the ability to degrade TNT isolated from soil samples at Deception Island, maritime Antarctica. Arch Microbiol 206, 39 (2024). https://doi.org/10.1007/s00203-023-03768-6
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00203-023-03768-6