Abstract
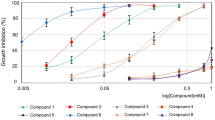
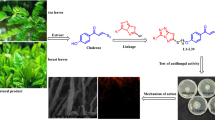
Fungal infections can be serious or life threatening in severe cases, and the need to discover and find novel antifungal agents persists. Chalcones are plant-derived aromatic compounds that have been appealing synthons for pharmaceutical industry as they have good anticancer, antibacterial, antifungal and anti-inflammatory properties. Although there are few structure–activity relationship studies on chalcones, studies that link the structural features of these compounds to their mode of action are scant. Thus, in this study, we aim to clarify the relationship between chalcone derivatives and their cellular target within the yeast cell Saccharomyces cerevisiae. We observed that some chalcone compounds lead to disruption of cell membrane and cause ion leakage out of the cell. Moreover, chalcones alter the biochemical composition of yeast cells detectable by FTIR spectroscopy and bind to the DNA as shown by our titration experiments based on UV–Vis absorbance spectroscopy. Thus, their interaction with the DNA may be the major impact of these compounds on yeast cells.




Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Andrade JT, Santos FRS, Lima WG, Sousa CDF, Oliveira LSFM, Ribeiro RIMA, Gomes AJPS, Araújo MGF, Villar JAFP (2018) Ferreira JMS design, synthesis, biological activity and structure-activity relationship studies of chalcone derivatives as potential anti-Candida agents. J Antibiot 71(8):702–712. https://doi.org/10.1038/s41429-018-0048-9
Arif R, Rana M, Yasmeen S, Amaduddin KMS, Abid M, Khan MS, Rahisuddin, (2020) Facile synthesis of chalcone derivatives as antibacterial agents: synthesis, DNA binding, molecular docking, DFT and antioxidant studies. J Mol Str 1208:127905. https://doi.org/10.1016/j.molstruc.2020.127905
Cushnie TP, Lamb AJ (2005) Antimicrobial activity of flavonoids. Int J Antimic Agents 26:343–356
Cushnie TP, Cushnie B, Lamb AJ (2008) Alkaloids: an overview of their antibacterial, antibiotic-enhancing and antivirulence activities. Int J Antimic Agents 32:288–293
Ergüden B, Ünver Y (2022) Phenolic chalcones lead to ion leakage from Gram-positive bacteria prior to cell death. Arch Microbiol 204:1–6. https://doi.org/10.1007/s00203-021-02603-0
Ergüden B, Lüleci HB, Ünver Y (2023) Chalcone Schiff bases disrupt cell membrane integrity of saccharomyces cerevisiae and Candida albicans cells. Arch Microbiol 205:246
Ghasemzadeh A, Jaafar HZ, Rahmat A (2016) Antioxidant activities, total phenolics and flavonoids content in two varieties of Malaysia young ginger (Zingiber officinale Roscoe). Molecules 17:4324–4333
Gupta D, Jain DK (2015) Chalcone derivatives as potential antifungal agents: synthesis, and antifungal activity. J Adv Pharm Technol Res 6:114–117
Harborne JB (1993) Introduction to ecological biochemistry. Academic Press
Havsteen BH (2002) The biochemistry and medical significance of the flavonoids. Pharmacol Therap 96:67–202
Kumar H, Chattopadhyay A, Prasath R, Devaraji V, Joshi R, Bhavana P, Saini P, Ghosh SK (2014) Design, synthesis, physicochemical studies, solvation, and dna damage of quinoline-appended chalcone derivative: comprehensive spectroscopic approach toward drug discovery. J Phys Chem B 118:7257–7266
Li K, Zhao S, Long J, Su J, Wu L, Tao J, Zhou J, Zhang JL, Chen X, Peng C (2020) A novel chalcone derivative has antitumor activity in melanoma by inducing DNA damage through the upregulation of ROS products. Cancer Cell Int 20:36. https://doi.org/10.1186/s12935-020-1114-5
Manach C, Scalbert A, Morand C, Rémésy C, Jiménez L (2004) Polyphenols: food sources and bioavailability. American J Clin Nutrit 79:727–747
Mellado M, Espinoza L, Madrid A, Mella J, Chávez-Weisser E, Diaz K, Cuellar M (2020) Design, synthesis, antifungal activity, and structure–activity relationship studies of chalcones and hybrid dihydrochromane–chalcones. Mol Div 24:603–615
Mukherjee A, Ghosh S, Ghosh S, Mahato S, Pal M, Sen SK, Majee A, Singh B (2021) Molecular recognition of synthesized halogenated chalcone by calf thymus DNA through multispectroscopic studies and analysis the anti-cancer, anti bacterial activity of the compounds. J Mol Liq 337:116504
Nijveldt RJ, van Nood E, van Hoorn DE, Boelens PG, van Norren K, van Leeuwen PA (2001) Flavonoids: a review of probable mechanisms of action and potential applications. Am J Clin Nutrit 74:418–425
Pietta PG (2000) Flavonoids as antioxidants. J Nat Prod 63:1035–1042
Shamsudin NF, Ahmed QU, Mahmood S, Ali Shah SA, Khatib A, Mukhtar S, Alsharif MA, Parveen H, Zakaria ZA (2022) Antibacterial effects of flavonoids and their structure-activity relationship study: a comparative interpretation. Molecules 27:1149. https://doi.org/10.3390/molecules27041149
Sockalingum GD, Sandt C, Toubas D, Gomez J, Pina P, Beguinot I, Witthuhn F, Aubert D, Allouch P, Pinon JM, Manfait M (2002) FTIR characterization of Candida species: a study on some reference strains and pathogenic C. albicans isolates from HIV+ patients. Vibrat Spect 28:137–146. https://doi.org/10.1016/S0924-2031(01)00152-7
Wang Y-H, Dong H-H, Zhao F, Wang J, Yan F, Jiang Y-Y, Jin Y-S (2016) The synthesis and synergistic antifungal effects of chalcones against drug resistant Candida albicans. Bioorg Med Chem Lett 26:3098–3102
Zarate X, Schott E, Escobar CA, Lopez-Castro R, Echeveriia C, Alvarado-Soto L, Ramirez-Tagle R (2016) Interaction of chalcones with ctDNA by spectrophotometric analysis and theoretical simulations. Quim Nova 39(8):914–918
Zheng Y, Wang X, Gao S, Ma M, Ren G, Liu H, Chen X (2015) Synthesis and antifungal activity of chalcone derivatives. Nat Prod Res 29:1804–1810
Funding
No funding was received.
Author information
Authors and Affiliations
Contributions
SA and BE designed and performed the experiments and BE wrote the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Communicated by Yusuf Akhter.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Alreqeb, S., Ergüden, B. Chalcone derivatives disrupt cell membrane integrity of Saccharomyces cerevisiae cells and alter their biochemical composition. Arch Microbiol 206, 34 (2024). https://doi.org/10.1007/s00203-023-03747-x
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00203-023-03747-x