Abstract
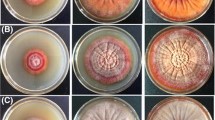
The development of practices that enhance the potential of actinomycetes as major antibiotic producers is a challenge in discovering new secondary metabolites. Light, an essential external stimulus for most microorganisms, could be exploited to manipulate their physiological processes. However, the effects of monochromatic green light on the production of secondary metabolites in actinomycetes have not yet been reported. In this paper, we report a novel and simple method that uses high-intensity monochromatic green light to potentially induce the production of cryptic secondary metabolites in the model actinomycete Streptomyces coelicolor A3(2). Using actinorhodin (ACT), a blue-pigmented antibiotic, and undecylprodigiosin (RED), a red-pigmented antibiotic, as indicators, we found that irradiation with high-intensity monochromatic green light-emitting diodes promoted sporulation, significantly decreased RED production, and increased ACT production. Semi-quantitative reverse transcription-polymerase chain reaction and western blot analyses revealed, for the first time, that stimulation with green light accelerated the expression of ActII-ORF4, a pathway-specific regulator of ACT biosynthesis in S. coelicolor A3(2). This approach of stimulating secondary metabolite biosynthesis pathways in actinomycetes by irradiation with high-intensity monochromatic green light is expected to facilitate the discovery of cryptic antibiotics that are not typically produced under conventional dark culture conditions. However, the effective intensity and duration of irradiation with green light that are required to activate these metabolite pathways may vary markedly among actinomycetes.
Graphical abstract





Similar content being viewed by others
Data availability
All data generated or analyzed in this study are included in this published article and the supplemental information file, and further inquiries can be directed to the corresponding author.
References
Alam K, Mazumder A, Sikdar S et al (2022) Streptomyces: The biofactory of secondary metabolites. Front Microbiol 13:968053. https://doi.org/10.3389/fmicb.2022.968053
Allenby NEE, Laing E, Bucca G et al (2012) Diverse control of metabolism and other cellular processes in Streptomyces coelicolor by the PhoP transcription factor: genome-wide identification of in vivo targets. Nucleic Acids Res 40:9543–9556. https://doi.org/10.1093/nar/gks766
Bentley SD, Chater KF, Cerdeño-Tárraga AM, Challis GL, Thomson NR, James KD, Harris DE, Quail MA, Kieser H, Harper D, Bateman A, Brown S, Chandra G, Chen CW, Collins M, Cronin A, Fraser A, Goble A, Hidalgo J, Hornsby T, Howarth S, Huang CH, Kieser T, Larke L, Murphy L, Oliver K, O’Neil S, Rabbinowitsch E, Rajandream MA, Rutherford K, Rutter S, Seeger K, Saunders D, Sharp S, Squares R, Squares S, Taylor K, Warren T, Wietzorrek A, Woodward J, Barrell BG, Parkhill J, Hopwood DA (2002) Complete genome sequence of the model actinomycete Streptomyces coelicolor A3(2). Nature 417:141–147. https://doi.org/10.1038/417141a
Bérdy J (2012) Thoughts and facts about antibiotics: where we are now and where we are heading. J Antibiot 65:385–395. https://doi.org/10.1038/ja.2012.27
Bibb MJ (2005) Regulation of secondary metabolism in Streptomycetes. Curr Opin Microbiol 8:208–215. https://doi.org/10.1016/j.mib.2005.02.016
Bode HB, Bethe B, Höfs R, Zeeck A (2002) Big effects from small changes: Possible ways to explore nature’s chemical diversity. ChemBioChem 3:619–627. https://doi.org/10.1002/1439-7633(20020703)3:7%3c;619::aid-cbic619%3e3.0.co;2-9
Chakraburtty R, Bibb M (1997) The ppGpp synthetase gene (relA) of Streptomyces coelicolor A3(2) plays a conditional role in antibiotic production and morphological differentiation. J Bacteriol 179:5854–5861. https://doi.org/10.1128/jb.179.18.5854-5861.1997
Endler JA (1993) The color of light in forests and its implications. Ecol Monogr 63:1–27. https://doi.org/10.2307/2937121
Gwynne PJ, Gallagher MP (2018) Light as a broad-spectrum antimicrobial. Front Microbiol 9:7603–7609. https://doi.org/10.3389/fmicb.2018.00119
Hoshino K, Imai Y, Mukai K, Hamauzu R, Ochi K, Hosaka T (2020) A putative mechanism underlying secondary metabolite overproduction by Streptomyces strains with a 23S rRNA mutation conferring erythromycin resistance. Appl Microbiol Biotechnol 104:2193–2203. https://doi.org/10.1007/s00253-019-10288-1
Huang J, Lih CJ, Pan KH, Cohen SN (2001) Global analysis of growth phase responsive gene expression and regulation of antibiotic biosynthetic pathways in Streptomyces coelicolor using DNA microarrays. Genes Dev 15:3183–3192. https://doi.org/10.1101/gad.943401
Imai Y, Fujiwara T, Ochi K, Hosaka T (2012) Development of the ability to produce secondary metabolites in Streptomyces through the acquisition of erythromycin resistance. J Antibiot 65:323–326. https://doi.org/10.1038/ja.2012.16
Imai Y, Sato S, Tanaka Y, Ochi K, Hosaka T (2015) Lincomycin at subinhibitory concentrations potentiates secondary metabolite production by Streptomyces spp. Appl Environ Microbiol 81:3869–3879. https://doi.org/10.1128/AEM.04214-14
Imbert M, Blondeau R (1999) Effect of light on germinating spores of Streptomyces viridosporus. FEMS Microbiol Lett 181:159–163. https://doi.org/10.1111/j.1574-6968.1999.tb08839.x
Ishizuka M, Imai Y, Mukai K, Shimono K, Hamauzu R, Ochi K, Hosaka T (2018) A possible mechanism for lincomycin induction of secondary metabolism in Streptomyces coelicolor A3(2). Antonie Van Leeuwenhoek 111:705–716. https://doi.org/10.1007/s10482-018-1021-0
Kieser T, Bibb MJ, Buttner MJ, Chater KF, Hopwood DA (2000) Practical Streptomyces genetics. The John Innes Foundation
Kojima M, Kimura N, Miura R (2015) Regulation of primary metabolic pathways in oyster mushroom mycelia induced by blue light stimulation: accumulation of shikimic acid. Sci Rep 5:559. https://doi.org/10.1038/srep08630
Kojima M, Hosaka T (2017) Method for producing useful metabolites and for screening conditions for producing metabolites. Publication of patent application JP 2018–57371A
Liu G, Chater KF, Chandra G, Niu G, Tan H (2013) Molecular regulation of antibiotic biosynthesis in Streptomyces. Microbiol Mol Biol Rev 77:112–143. https://doi.org/10.1128/MMBR.00054-12
Liu P, Zhu H, Zheng G et al (2017) Metabolic engineering of Streptomyces coelicolor for enhanced prodigiosins (RED) production. Sci China Life Sci 60:948–957. https://doi.org/10.1007/s11427-017-9117-x
Nakano Y, Fujii H, Kojima M (2010) Identification of blue-light photoresponse genes in oyster mushroom mycelia. Biosci Biotechnol Biochem 74:2160–2165. https://doi.org/10.1271/bbb.100565
Nakano Y, Fujii H, Kojima M (2011) Effects of light wavelength and intensity on the expression of photoresponse genes in oyster mushroom mycelia. J Light vis Environ 35:90–94. https://doi.org/10.2150/jlve.35.90
Ochi K, Hosaka T (2012) New strategies for drug discovery: activation of silent or weakly expressed microbial gene clusters. Appl Microbiol Biotechnol 97:87–98. https://doi.org/10.1007/s00253-012-4551-9
Ohlendorf R, Möglich A (2022) Light-regulated gene expression in bacteria: Fundamentals, advances, and perspectives. Front Bioeng Biotechnol 10:1029403. https://doi.org/10.3389/fbioe.2022.1029403
Omura S (1986) Philosophy of new drug discovery. Microbiol Rev 50:259–279. https://doi.org/10.1128/mr.50.3.259-279.1986
Onaka H, Mori Y, Igarashi Y, Furumai T (2011) Mycolic acid-containing bacteria induce natural-product biosynthesis in Streptomyces species. Appl Environ Microbiol 77:400–406. https://doi.org/10.1128/aem.01337-10
Padmanabhan S, Pérez-Castaño R, Osete-Alcaraz L et al (2022) Vitamin B12 photoreceptors. In: Litwack G (ed) Vitamin B12. Academic Press, pp 149–184
Schneider CA, Rasband WS, Eliceiri KW (2012) NIH Image to ImageJ: 25 years of image analysis. Nat Meth 9:671–675. https://doi.org/10.1038/nmeth.2089
Sevcikova B, Rezuchova B, Mazurakova V et al (2021) Cross-recognition of promoters by the nine SigB homologues present in Streptomyces coelicolor A3(2). Int J Mol Sci 22:7849. https://doi.org/10.3390/ijms22157849
Takano H (2016) The regulatory mechanism underlying light-inducible production of carotenoids in nonphototrophic bacteria. Biosci Biotechnol Biochem 80:1264–1273. https://doi.org/10.1080/09168451.2016.1156478
Takano H, Obitsu S, Beppu T, Ueda K (2005) Light-induced carotenogenesis in Streptomyces coelicolor A3(2): identification of an extracytoplasmic function sigma factor that directs photodependent transcription of the carotenoid biosynthesis gene cluster. J Bacteriol 187:1825–1832. https://doi.org/10.1128/JB.187.5.1825-1832.2005
Takano H, Hagiwara K, Ueda K (2015) Fundamental role of cobalamin biosynthesis in the developmental growth of Streptomyces coelicolor A3 (2). Appl Microbiol Biotechnol 99:2329–2337. https://doi.org/10.1007/s00253-014-6325-z
Tanaka Y, Hosaka T, Ochi K (2010) Rare earth elements activate the secondary metabolite–biosynthetic gene clusters in Streptomyces coelicolor A3(2). J Antibiot 63:477–481. https://doi.org/10.1038/ja.2010.53
Tenconi E, Traxler MF, Hoebreck C, van Wezel GP, Rigali S (2018) Production of prodiginines is part of a programmed cell death process in Streptomyces coelicolor. Front Microbiol 9:1742. https://doi.org/10.3389/fmicb.2018.01742
Tenconi E, Traxler M, Tellatin D et al (2020) Prodiginines postpone the onset of sporulation in Streptomyces coelicolor. Antibiotics (basel) 9:847. https://doi.org/10.3390/antibiotics9120847
Thu TTM, Vinh DTT, Dung NA, Tu NHK (2021) Effect of Lactic Acid produced by lactic acid bacteria on prodigiosin production from Streptomyces coelicolor. Res J Pharm Technol. https://doi.org/10.52711/0974-360x.2021.00345
Xia H, Li X, Li Z et al (2020) The Application of regulatory cascades in Streptomyces: yield enhancement and metabolite mining. Front Microbiol 11:406. https://doi.org/10.3389/fmicb.2020.00406
Xu F, Wang J, Zhao G-P (2017) Alpha-ketoglutarate protects Streptomyces coelicolor from visible light-induced phototoxicity. Biochem Biophys Rep 9:22–28. https://doi.org/10.1016/j.bbrep.2016.11.002
Zerikly M, Challis GL (2009) Strategies for the discovery of new natural products by genome mining. ChemBioChem 10:625–633. https://doi.org/10.1002/cbic.200800389
Zong G, Fu J, Zhang P et al (2022) Use of elicitors to enhance or activate the antibiotic production in Streptomyces. Crit Rev Biotechnol 42:1260–1283. https://doi.org/10.1080/07388551.2021.1987856
Acknowledgements
The authors express their gratitude to Y. Imai, K. Mukai, K. Hoshino, R. Hamauzu, and N. Kimura for their assistance with the experiments.
Funding
This work was supported by the Ohsumi Frontier Science Foundation (to TH), the Hitachi Scholarship Research Support Program 2019 through the Hitachi Global Foundation (to CK), and the Chulalongkorn University Ratchadaphiseksomphot Fund (to CK).
Author information
Authors and Affiliations
Contributions
MK conceived and designed the research. CK and TH conducted the experiments. MK, CK, and TH analyzed the data. MK and CK wrote the manuscript. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by El-Sayed.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kanchanabanca, C., Hosaka, T. & Kojima, M. High-intensity green light potentially activates the actinorhodin biosynthetic pathway in Streptomyces coelicolor A3(2). Arch Microbiol 206, 8 (2024). https://doi.org/10.1007/s00203-023-03730-6
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00203-023-03730-6