Abstract
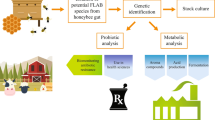
Lactic acid bacteria and bifidobacteria (LAB and Bifido), isolated from the gastrointestinal tract of Apis mellifera intermissa (BGIT), honey (H), propolis (P) and bee bread (BB) of hives set in different vegetations (wildflowers, caraway, orange blossom, Marrubium vulgare, Eucalyptus and Erica cinerea), were subjected to analysis of their antibacterial potential. Isolates able to inhibit Staphylococcus aureus were selected and identified with MALDI-TOF MS leading to 154 strains representing 12 LAB and Bifido species. Lactiplantibacillus plantarum, Pediococcus pentosaceus and Enterococcus faecalis were predominantly found in all matrices. BGIT showed the highest LAB and Bifido diversity with exclusive occurrences of five species (including Bifidobacterium asteroides and Limosilactobacillus fermentum). Honey was the second origin harboring an important variety of LAB species of which Apilactobacillus kunkeei and Enterococcus mundtii were characteristic of both H and BGIT. Principal components analysis revealed associations between antibacterial activities of LAB and Bifido, matrices and honey bee forage plants. Inhibition trends of S. aureus and Citrobacter freundii were highlighted with: L. plantarum from BGIT, P, H of bees feeding on E. cinerea; Pediococcus pentosaceus from BGIT, P, BB associated with E. cinerea; and Bifidobacterium asteroides from BGIT/orange blossom system. However, Enterococcus faecium associated with BGIT/Eucalyptus system antagonized Escherichia coli, Klebsiella pneumoniae, Pseudomonas aeruginosa and Acinetobacter baumannii. Our findings highlighted noteworthy effects of bee forage plants on the antibacterial activity of LAB and Bifido. Our approach could be useful to identify multiple conditions promoting antibacterial potency of LAB and Bifido under the combined effects of feeding plants and living matrices.








Similar content being viewed by others
Data availability statement
Data generated or analyzed during this study are provided in full within the published article.
References
Abriouel H, Lerma LL, Casado Muñoz M, Montoro BP, Kabisch J, Pichner R, Cho GS, Neve H, Fusco V, Franz CM, Gálvez A, Benomar N (2015) The controversial nature of the Weissella genus: technological and functional aspects versus whole genome analysis-based pathogenic potential for their application in food and health. Front Microbiol 6:1197. https://doi.org/10.3389/fmicb.2015.01197
Ağagündüz D, Yılmaz B, Şahin TÖ, Güneşliol BE, Ayten Ş, Russo P, Spano G, Rocha JM, Bartkiene E, Özogul F (2021) Dairy lactic acid bacteria and their potential function in dietetics: the food-gut-health axis. Foods 10(12):3099. https://doi.org/10.3390/foods10123099
Anderson KE, Ricigliano VA, Copeland DC, Mott BM, Maes P (2023) Social interaction is unnecessary for hindgut microbiome transmission in honey bees: the effect of diet and social exposure on tissue-specific microbiome assembly. Microb Ecol 85(4):1498–1513. https://doi.org/10.1007/s00248-022-02025-5
Asama T, Arima TH, Gomi T, Keishi T, Tani H, Kimura Y, Tatefuji T, Hashimoto K (2015) Lactobacillus kunkeei YB38 from honeybee products enhances IgA production in healthy adults. J Appl Microbiol 119:818–826. https://doi.org/10.1111/jam.12889
Bottacini F, Ventura M, van Sinderen D, O’Connell Motherway M (2014) Diversity, ecology and intestinal function of bifidobacteria. Microb Cell Fact 13(Suppl 1):S4. https://doi.org/10.1186/1475-2859-13-S1-S4
Bulgasem BY, Lani MN, Hassan Z, Wan Yusoff WM, Fnaish SG (2016) Antifungal activity of lactic acid bacteria strains isolated from natural honey against pathogenic Candida species. Mycobiology 44:302–309. https://doi.org/10.5941/MYCO.2016.44.4.302
Butler É, Oien RF, Lindholm C, Olofsson TC, Nilson B, Vásquez A (2016) A pilot study investigating lactic acid bacterial symbionts from the honeybee in inhibiting human chronic wound pathogens. Int Wound J 13:729–737. https://doi.org/10.1111/iwj.12360
Castelli L, Branchiccela B, Garrido M, Invernizzi C, Porrini M, Romero H, Santos E, Zunino P, Antúnez K (2020) Impact of nutritional stress on honeybee gut microbiota, immunity, and nosema ceranae infection. Microb Ecol 80(4):908–919. https://doi.org/10.1007/s00248-020-01538-1
Chen J, Chen X, Ho CL (2021) Recent development of probiotic Bifidobacteria for treating human diseases. Front Bioeng Biotechnol 9:770248. https://doi.org/10.3389/fbioe.2021.770248
Cornara L, Biagi M, Xiao J, Burlando B (2017) Therapeutic properties of bioactive compounds from different honeybee products. Front Pharmacol 8:412. https://doi.org/10.3389/fphar.2017.00412
Cuesta-Maté A, Renelies-Hamilton J, Kryger P, Jensen AB, Sinotte VM, Poulsen M (2021) Resistance and vulnerability of honeybee (Apis mellifera) gut bacteria to commonly used pesticides. Front Microbiol. 12:717990. https://doi.org/10.3389/fmicb.2021.717990
Dapkevicius M, Sgardioli B, Câmara S, Poeta P, Malcata FX (2021) Current trends of enterococci in dairy products: a comprehensive review of their multiple roles. Foods 10(4):821. https://doi.org/10.3390/foods10040821
De Filippis F, Pasolli E, Ercolini D (2020) The food-gut axis: lactic acid bacteria and their link to food, the gut microbiome and human health. FEMS Microbiol Rev 44:454–489. https://doi.org/10.1093/femsre/fuaa015
Dušková M, Šedo O, Kšicová K, Zdráhal Z, Karpíšková R (2012) Identification of lactobacilli isolated from food by genotypic methods and MALDI-TOF MS. Int J Food Microbiol 159(2):107–114. https://doi.org/10.1016/j.ijfoodmicro.2012.07.029
Ellegaard KM, Engel P (2019) Genomic diversity landscape of the honey bee gut microbiota. Nat Commun 10(1):446. https://doi.org/10.1038/s41467-019-08303-0
El-Sohaimy AA, Masry S, Shehata MG, Al-Kahtani SN, Abdelwahab TE, Abdelmotaleb Y, Nour ME (2020) Isolation, identification and antimicrobial activity of unprecedented lactic acid bacterial isolates from honeybees. Pak J Biol Sci 23:467–477. https://doi.org/10.3923/pjbs.2020.467.477
Gaudreau AM, Labrie J, Goetz C, Dufour S, Jacques M (2018) Evaluation of MALDI-TOF mass spectrometry for the identification of bacteria growing as biofilms. J Microbiol Methods 145:79–81. https://doi.org/10.1016/j.mimet.2018.01.003
Gibb S, Strimmer K (2012) MALDIquant: a versatile R package for the analysis of mass spectrometry data. Bioinformatics 28:2270–2271. https://doi.org/10.1093/bioinformatics/bts447
Gorrochategui-Ortega J, Muñoz-Colmenero M, Kovačić M, Filipi J, Puškadija Z, Kezić N, Parejo M, Büchler R, Estonba A, Zarraonaindia I (2022) A short exposure to a semi-natural habitat alleviates the honey bee hive microbial imbalance caused by agricultural stress. Sci Rep 12(1):18832. https://doi.org/10.1038/s41598-022-23287-6
Gregersen T (1978) Rapid method for distinction of Gram-negative from Gram-positive bacteria. Eur J Appl Microbiol 5:123–127
Hernández-González JC, Martínez-Tapia A, Lazcano-Hernández G, García-Pérez BE, Castrejón-Jiménez NS (2021) Bacteriocins from lactic acid bacteria. A powerful alternative as antimicrobials, probiotics, and immunomodulators in veterinary medicine. Animals 11(4):979. https://doi.org/10.3390/ani11040979
Imade EE, Omonigho SE, Babalola OO, Enagbonma BJ (2021) Lactic acid bacterial bacteriocins and their bioactive properties against food-associated antibiotic-resistant bacteria. Ann Microbiol 71:44. https://doi.org/10.1186/s13213-021-01652-6
Iorizzo M, Letizia F, Sonia Ganassi S, Bruno Testa B, Sonia Petrarca S, Gianluca Albanese G, Di Criscio D, De Cristofaro A (2022) Functional properties and antimicrobial activity from lactic acid bacteria as resources to improve the health and welfare of honey bees. InSects 13(3):308. https://doi.org/10.3390/insects13030308
Iorizzo M, Pannella G, Lombardi SJ, Ganassi S, Testa B, Succi M, Sorrentino E, Petrarca S, De Cristofaro A, Coppola R, Tremonte P (2020) Inter- and intra-species diversity of lactic acid bacteria in Apis mellifera ligustica colonies. Microorganisms 8:1578. https://doi.org/10.3390/microorganisms8101578
Jolliffe IT (2002) Principal component analysis 518. Springer, New York
Kakumanu ML, Reeves AM, Anderson TD, Rodrigues RR, Williams MA (2016) Honey bee gut microbiome is altered by in-hive pesticide exposures. Front Microbiol 7:1255. https://doi.org/10.3389/fmicb.2016.01255
Kassambara A (2017) Practical guide to cluster analysis in R: unsupervised machine learning, vol 1. Sthda
Klein A-M, Vaissiere BE, Cane JH, Steffan-Dewenter I, Cunningham SA, Kremen C, Tscharntke T (2007) Importance of pollinators in changing landscapes for world crops. Proc R Soc B 274(1608):303–313. https://doi.org/10.1098/rspb.2006.3721
Kwong WK, Moran NA (2016) Gut microbial communities of social bees. Nat Rev Microbiol 14:374–384. https://doi.org/10.1038/nrmicro.2016.43
Li C, Tang M, Li X, Zhou X (2022) Community dynamics in structure and function of honey bee gut bacteria in response to winter dietary shift. Mbio 13(5):e0113122. https://doi.org/10.1128/mbio.01131-22
Liasi SA, Azmi TI, Hassan MD, Shuhaimi M, Rosfarizan M, Ariff AB (2009) Antimicrobial activity and antibiotic sensitivity of three isolates of lactic acid bacteria from fermented fish product, Budu. Malays J Microbiol 5: 33–37. https://doi.org/10.21161/mjm.15008
Lim HJ, Shin HS (2020) Antimicrobial and immunomodulatory effects of Bifidobacterium strains: a review. J Microbiol Biotechnol 30(12):1793–1800. https://doi.org/10.4014/jmb.2007.07046
Mahmood AL, Lani MN, Hassan Z, Razak SBA, Ahmad FT (2021) Antioxidant and antimicrobial properties of Indo-Malayan stingless bee (Heterotrigona itama) honey from different seasons and distribution of flowers. Food Res 5(2):498–507. https://doi.org/10.26656/fr.2017.5(2).546
Mahmoud Elzeini H, Ali AA, Nasr FN, Essam Elenany Y, Abdel Moneim Hassan A (2021) Isolation and identification of lactic acid bacteria from the intestinal tracts of honey bees, Apis mellifera L., in Egypt. J Apic Res 60:349–357. https://doi.org/10.1080/00218839.2020.1746019
Meradji M, Nadia Bachtarzi N, Diego Mora D, Karima Kharroub K (2023) Characterization of lactic acid bacteria strains isolated from Algerian honeybee and honey and exploration of their potential probiotic and functional features for human use. Foods 12(12):2312. https://doi.org/10.3390/foods12122312
Mohammad SM, Mahmud-Ab-Rashid NK, Zawawi N (2021) Stingless bee-collected pollen (bee bread): chemical and microbiology properties and health benefits. Molecules 26(4):957. https://doi.org/10.3390/molecules26040957
Mulaw G, Sisay Tessema T, Muleta D, Tesfaye A (2020) In Vitro evaluation of probiotic properties of lactic acid bacteria isolated from some traditionally fermented Ethiopian ood products. Int J Microbiol. https://doi.org/10.1155/2020/6401356
Nataraj BH, Ali SA, Behare PV, Yadav H (2020) Postbiotics-parabiotics: the new horizons in microbial biotherapy and functional foods. Microb Cell Fact 19:168. https://doi.org/10.1186/s12934-020-01426-w
Neov B, Georgieva A, Shumkova R, Radoslavov G, Hristov P (2019) Biotic and abiotic factors associated with colonies mortalities of managed honey bee (Apis mellifera). Diversity 11:237–253. https://doi.org/10.3390/d11120237
Nowak A, Szczuka D, Górczyńska A, Motyl I, Kręgiel D (2021) Characterization of Apis mellifera gastrointestinal microbiota and lactic acid bacteria for honeybee protection—a review. Cells 10:701. https://doi.org/10.3390/cells10030701
Olofsson TC, Butler È, Markowicz P, Lindholm C, Larsson L, Vásquez A (2016) Lactic acid bacterial symbionts in honeybees—an unknown key to honey’s antimicrobial and therapeutic activities. Int Wound J 13:668–679. https://doi.org/10.1111/iwj.12345
Papp M, Békési L, Farkas R, Makrai L, Judge MF, Maróti G, Tőzsér D, Solymosi N (2022) Natural diversity of the honey bee (Apis mellifera) gut bacteriome in various climatic and seasonal states. PLoS ONE 17(9):e0273844. https://doi.org/10.1371/journal.pone.0273844
Pasupuleti VR, Sammugam L, Ramesh N, Gan SH (2017) Honey, propolis, and royal jelly: a comprehensive review of their biological actions and health benefits. Oxid Med Cell Longev. https://doi.org/10.1155/2017/1259510
Pełka K, Worobo RW, Walkusz J, Szweda P (2021) Bee pollen and bee bread as a source of bacteria producing antimicrobials. Antibiotics 10:713. https://doi.org/10.3390/antibiotics10060713
Piccart K, Vásquez A, Piepers S, De Vliegher S, Olofsson TC (2016) Short communication: lactic acid bacteria from the honeybee inhibit the in vitro growth of mastitis pathogens. Int J Dairy Sci 99:2940–2944. https://doi.org/10.3168/jds.2015-10208
R Core Team (2021) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Rabaoui G, Sánchez-Juanes F, Tebini M, Naghmouchi K, Bellido J, Ben-Mahrez K, Réjiba S (2023) Potential probiotic lactic acid bacteria with anti-Penicillium expansum activity from different species of Tunisian edible snails. Probiot Antimicrob Proteins 15(1):82–106. https://doi.org/10.1007/s12602-021-09882-5
Ramos OY, Basualdo M, Libonatti C, Vega MF (2020) Current status and application of lactic acid bacteria in animal production systems with a focus on bacteria from honey bee colonies. J Appl Microbiol 128:1248–1260. https://doi.org/10.1111/jam.14469
Santorelli LA, Wilkinson T, Abdulmalik R, Rai Y, Creevey CJ, Huws S, Gutierrez-Merino J (2023) Beehives possess their own distinct microbiomes. Environ Microbiome 18(1):1. https://doi.org/10.1186/s40793-023-00460-6
Semmar N (2011) Computational metabolomics 238. Nova Science, New York
Shehata MG, Ahmad FT, Badr AN, Masry SH, El-Sohaimy SA (2020) Chemical analysis, antioxidant, cytotoxic and antimicrobial properties of propolis from different geographic regions. Ann Agric Sci 65(2):209–217. https://doi.org/10.1016/j.aoas.2020.12.001
Smutin D, Lebedev E, Selitskiy M, Panyushev N, Adonin L (2022) Micro"bee"ota: honey bee normal microbiota as a part of superorganism. Microorganisms 10(12):2359. https://doi.org/10.3390/microorganisms10122359
Subotic S, Boddicker AM, Nguyen VM, Rivers J, Briles CE, Mosier AC (2019) Honey bee microbiome associated with different hive and sample types over a honey production season. PLoS ONE 14(11):e0223834. https://doi.org/10.1371/journal.pone.0223834
Torres-Moreno R, Hernández-Sánchez HS, Méndez-Tenorio A, Palmeros-Sánchez B, Melgar-Lalanne G (2021) Characterization and identification of lactic acid bacteria from Mexican stingless bees (Apidae: Meliponini). IOP Conf Ser Earth Environ Sci 85:012010. https://doi.org/10.1088/1755-1315/858/1/012010
Truc L, Ngoc AT, Hong T, Thanh TN, Kim HH, Kim LP, Truong GH, Quoc PT, Ngoc T (2019) Selection of lactic acid bacteria (LAB) antagonizing Vibrio parahaemolyticus: the pathogen of acute hepatopancreatic necrosis disease (AHPND) in whiteleg shrimp (Penaeus vannamei). Biology 8:91. https://doi.org/10.3390/biology8040091
Vásquez A, Olofsson TC (2009) The lactic acid bacteria involved in the production of bee pollen and bee bread. J Apic Res 48:189–195. https://doi.org/10.3896/IBRA.1.48.3.07
Villalba A, Maggi M, Ondarza PM, Szawarski N, Miglioranza K (2020) Influence of land use on chlorpyrifos and persistent organic pollutant levels in honey bees, bee bread and honey: beehive exposure assessment. Sci Total Environ 713:136554. https://doi.org/10.1016/j.scitotenv.2020.136554
Wu J, Lang H, Mu X, Zhang Z, Su Q, Hu X, Zheng H (2021) Honey bee genetics shape the strain-level structure of gut microbiota in social transmission. Microbiome 9(1):225. https://doi.org/10.1186/s40168-021-01174-y
Yadav MK, Kumari I, Singh BI, Sharma KK, Tiwari SK (2022) Probiotics, prebiotics and synbiotics: safe options for next-generation therapeutics. Appl Microbiol Biotechnol 106(2):505–521. https://doi.org/10.1007/s00253-021-11646-8
Zendo T, Ohashi C, Maeno S, Piao X, Salminen S, Sonomoto K, Endo A (2020) Kunkecin A, a new nisin variant bacteriocin produced by the fructophilic lactic acid bacterium, Apilactobacillus kunkeei FF30-6 isolated from honey bees. Front Microbiol 11:571903. https://doi.org/10.3389/fmicb.2020.571903
Zhang H, HuangFu H, Wang X, Zhao S, Liu Y, Lv H, Qin G, Tan Z (2021) Antibacterial activity of lactic acid producing Leuconostoc mesenteroides QZ1178 against pathogenic Gallibacterium anatis. Front Vet Sci 8:630294. https://doi.org/10.3389/fvets.2021.630294
Zhang Z, Mu X, Cao Q, Shi Y, Hu X, Zheng H (2022) Honeybee gut Lactobacillus modulates host learning and memory behaviors via regulating tryptophan metabolism. Nat Commun 13(1):2037. https://doi.org/10.1038/s41467-022-29760-0
Zheng H, Powell JE, Steele MI, Dietrich C, Moran NA (2017) Honeybee gut microbiota promotes host weight gain via bacterial metabolism and hormonal signaling. Proc Natl Acad Sci USA 114(18):4775–4780. https://doi.org/10.1073/pnas.1701819114
Zheng H, Steele MI, Leonard SP, Motta E, Moran NA (2018) Honey bees as models for gut microbiota research. Lab Anim 47:317–325. https://doi.org/10.1038/s41684-018-0173-x
Acknowledgements
We thank Pr. Karim Naghmouchi, from Biochemistry and Biotechnology Laboratory, Faculty of Sciences of Tunis, University of Tunis El Manar and College of Clinical Pharmacy, Department of Pharmaceutical Chemistry, Al Baha University, Saudi Arabia, for suggesting us to investigate LAB and Bifido from honey bee; Dr. Lamia Thabet, from Traumatology and Great Burned Center, Tunis, Tunisia, for the gift of strains S. aureus, E. coli, K. pneumoniae, A. baumannii, C. freundii and P. aeruginosa (collection of 2018). MALDI-TOF MS experiment was performed at the Department of Pathology and Microbiology, Faculty of Veterinary Medicine, Université de Montréal.
Funding
The authors are grateful to The Tunisian Ministry of Higher Education and Scientific Research for financial support for the laboratory LR01ES05. The funder had no role in study design, data collection and interpretation, or the decision to submit the work for publication.
Author information
Authors and Affiliations
Contributions
HB-M designed the study, performed experiments and drafted the manuscript. NS realized the statistical analysis (distribution analysis, principal component analysis) of antibacterial activity and contributed to the manuscript writing and revision. MSC completed biostatistical analyses and contributed to manuscript writing. KB-M contributed to the manuscript revision. M-OB-B. contributed to study supervision, and manuscript writing and edition. SR supervised the work, wrote and edited the manuscript. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare there are no competing interests.
Consent to publish
Not applicable.
Additional information
Communicated by Adriano Cruz.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ben-Miled, H., Semmar, N., Castellanos, M.S. et al. Effect of honey bee forage plants in Tunisia on diversity and antibacterial potential of lactic acid bacteria and bifidobacteria from Apis mellifera intermissa and its products. Arch Microbiol 205, 295 (2023). https://doi.org/10.1007/s00203-023-03630-9
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00203-023-03630-9