Abstract
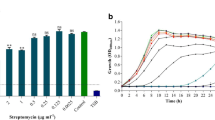
Microbes within an infection impact neighbors’ pathogenicity. This study aimed to address in vitro virulence activity of Pseudomonas aeruginosa under the binary interaction with Acinetobacter baumannii or Enterococcus faecium, co-isolated from two chronic wound infections. The biofilm formation of Pseudomonas was enhanced 1.5- and 1.4-fold when it was simultaneously cultured with Acinetobacter and Enterococcus, respectively. Pseudomonas motility was increased by 1.9- and 1.5-fold (swimming), 3.6- and 1.9-fold (swarming), and 1.5- and 1.5-fold (twitching) in the dual cultures with Acinetobacter and Enterococcus, respectively. The synergistic hemolysis activity of Pseudomonas was observed with the heat-killed Acinetobacter and Enterococcus cells. The minimum inhibitory concentration of ciprofloxacin against Pseudomonas was increased from (μg mL−1) 25 to 400 in the individual and mixed cultures, respectively. The pyocyanin production by Pseudomonas in the single and mixed cultures with Acinetobacter and Enterococcus was (μg/mL) 1.8, 2.3, and 2.9, respectively. The expression of lasI, rhlI, and pqsR genes was up-regulated by 1.0-, 1.9-, and 16.3-fold, and 4.9-, 1.0-, and 9.3-fold when Pseudomonas was incubated with Acinetobacter and Enterococcus, respectively. Considering the entire community instead of a single pathogen may lead to a more effective therapeutic design for persistent infections caused by Pseudomonas.




Similar content being viewed by others
Data Availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
References
Bottery MJ, Matthews JL, Wood AJ, Johansen HK, Pitchford JW, Friman VP (2022) Inter-species interactions alter antibiotic efficacy in bacterial communities. ISME 16:812–821. https://doi.org/10.1038/s41396-021-01130-6
Cai YM, Hutchin A, Craddock J, Walsh MA, Webb JS, Tews I (2020) Differential impact on motility and biofilm dispersal of closely related phosphodiesterases in Pseudomonas aeruginosa. Sci Rep 10:6232. https://doi.org/10.1038/s41598-020-63008-5
Chaurasia LK, Tirwa RK, Tamang B (2022) Potential of Enterococcus faecium LM5. 2 for lipopeptide biosurfactant production and its effect on the growth of maize (Zea mays L.). Arch Microbiol 204:223. https://doi.org/10.1007/s00203-022-02834-9
Cummings LA, Hoogestraat DR, Rassoulian-Barrett SL, Rosenthal CA, Salipante SJ, Cookson BT et al (2020) Comprehensive evaluation of complex polymicrobial specimens using next generation sequencing and standard microbiological culture. Sci Rep 10:5446. https://doi.org/10.1038/s41598-020-62424-x
D’Argenio DA, Calfee MW, Rainey PB, Pesci EC (2002) Autolysis and autoaggregation in Pseudomonas aeruginosa colony morphology mutants. J Bacteriol 184:6481–6489. https://doi.org/10.1128/JB.184.23.6481-6489.2002
DeBritto S, Gajbar TD, Satapute P, Sundaram L, Lakshmikantha RY, Jogaiah S et al (2020) Isolation and characterization of nutrient dependent pyocyanin from Pseudomonas aeruginosa and its dye and agrochemical properties. Sci Rep 10:1542. https://doi.org/10.1038/s41598-020-58335-6
Donlan RM (2002) Biofilms: microbial life on surfaces. Emerg Infect Dis 8:881–890. https://doi.org/10.3201/eid0809.020063
Fadwa AO, Alkoblan DK, Mateen A, Albarag AM (2021) Synergistic effects of zinc oxide nanoparticles and various antibiotics combination against Pseudomonas aeruginosa clinically isolated bacterial strains. Saudi J Biol Sci 28:928–935. https://doi.org/10.1016/j.sjbs.2020.09.064
Frykberg RG, Banks J (2015) Challenges in the treatment of chronic wounds. Adv Wound Care 4:560–582. https://doi.org/10.1089/wound.2015.0635
Glessner A, Smith RS, Iglewski BH, Robinson JB (1999) Roles of Pseudomonas aeruginosa las and rhl quorum-sensing systems in control of twitching motility. J Bacteriol 181:1623–1629. https://doi.org/10.1128/JB.181.5.1623-1629.1999
Gupta B, Puri S, Thakur IS, Kaur J (2020) Enhanced pyrene degradation by a biosurfactant producing Acinetobacter baumannii BJ5: growth kinetics, toxicity and substrate inhibition studies. Environ Technol Innov 19:100804. https://doi.org/10.1016/j.eti.2020.100804
Hall S, McDermott C, Anoopkumar-Dukie S, McFarland AJ, Forbes A, Perkins AV et al (2016) Cellular effects of Pyocyanin, a secreted virulence factor of Pseudomonas aeruginosa. Toxins (basel) 8:236. https://doi.org/10.3390/toxins8080236
Høiby N, Bjarnsholt T, Moser C, Bassi GL, Coenye T, Donelli G et al (2015) ESCMID∗ guideline for the diagnosis and treatment of biofilm infections 2014. Clin Microbiol Infect. https://doi.org/10.1016/j.cmi.2014.10.024
Josenhans C, Suerbaum S (2002) The role of motility as a virulence factor in bacteria. Int J Med Microbiol 291:605–614. https://doi.org/10.1078/1438-4221-00173
Kim SK, Lee JH (2016) Biofilm dispersion in Pseudomonas aeruginosa. J Microbiol 54:71–85. https://doi.org/10.1007/s12275-016-5528-7
Kostylev M, Kim DY, Smalley NE, Salukhe I, Greenberg EP, Dandekar AA (2019) Evolution of the Pseudomonas aeruginosa quorum-sensing hierarchy. Proc Natl Acad Sci 116:7027–7032. https://doi.org/10.1073/pnas.1819796116
Lee J, Zhang L (2015) The hierarchy quorum sensing network in Pseudomonas aeruginosa. Protein Cell 6:26–41. https://doi.org/10.1007/s13238-014-0100-x
Limoli DH, Hoffman LR (2018) Help, hinder, hide and harm: what can we learn from the interactions between Pseudomonas aeruginosa and Staphylococcus aureus during respiratory infections? Thorax 7:4684–4692. https://doi.org/10.1136/thoraxjnl-2018-212616
Overhage J, Bains M, Brazas MD, Hancock RE (2008) Swarming of Pseudomonas aeruginosa is a complex adaptation leading to increased production of virulence factors and antibiotic resistance. J Bacteriol 190:2671–2679. https://doi.org/10.1128/JB.01659-07
Peleg AY, Hogan DA, Mylonakis E (2010) Medically important bacterial-fungal interactions. Nat Rev Microbiol 8:340–349. https://doi.org/10.1038/nrmicro2313
Peters BM, Jabra-Rizk MA, O’May GA, Costerton JW, Shirtliff ME (2012) Polymicrobial interactions: impact on pathogenesis and human disease. Clin Microbiol Rev 25:193–213. https://doi.org/10.1128/CMR.00013-11
Reigada I, San-Martin-Galindo P, Gilbert-Girard S, Chiaro J, Cerullo V, Savijoki K et al (2021) Surfaceome and exoproteome dynamics in dual-species Pseudomonas aeruginosa and Staphylococcus aureus Biofilms. Front Microbiol 12:672975. https://doi.org/10.3389/fmicb.2021.672975
Rezaie P, Pourhajibagher M, Chiniforush N, Hosseini N, Bahador A (2018) The effect of quorum-sensing and efflux pumps interactions in Pseudomonas aeruginosa against photo oxidative stress. J Lasers Med Sci 9:161–167. https://doi.org/10.15171/jlms.2018.30
Shi N, Gao Y, Yin D, Song Y, Kang J, Li X et al (2019) The effect of the sub-minimal inhibitory concentration and the concentrations within resistant mutation window of ciprofloxacin on MIC, swimming motility and biofilm formation of Pseudomonas aeruginosa. Microb Pathog 137:103765. https://doi.org/10.1016/j.micpath.2019.103765
Solis-Velazquez OA, Gutiérrez-Lomelí M, Guerreo-Medina PJ, Rosas-García ML, Iñiguez-Moreno M, Avila-Novoa MG (2021) Nosocomial pathogen biofilms on biomaterials: Different growth medium conditions and components of biofilms produced in vitro. J Microbiol Immunol Infect 54:1038–1047. https://doi.org/10.1016/j.jmii.2020.07.002
Sorroche FG, Spesia MB, Zorreguieta A, Giordano W (2012) A positive correlation between bacterial autoaggregation and biofilm formation in native Sinorhizobium meliloti isolates from Argentina. Appl Environ Microbiol 78:4092–4101. https://doi.org/10.1128/AEM.07826-11
Sperandio D, Rossignol G, Guerillon J, Connil N, Orange N, Feuilloley MG et al (2010) Cell-associated hemolysis activity in the clinical strain of Pseudomonas fluorescens MFN1032. BMC Microbiol. https://doi.org/10.1186/1471-2180-10-124
Trizna EY, Yarullina MN, Baidamshina DR, Mironova AV, Akhatova FS, Rozhina EV et al (2020) Bidirectional alterations in antibiotics susceptibility in Staphylococcus aureus-Pseudomonas aeruginosa dual-species biofilm. Sci Rep 10:14849. https://doi.org/10.1038/s41598-020-71834-w
Warrier A, Satyamoorthy K, Murali TS (2021) Quorum-sensing regulation of virulence factors in bacterial biofilm. Future Microbiol 16:1003–1021. https://doi.org/10.2217/fmb-2020-0301
Acknowledgements
This work was supported by a grant from Ferdowsi University of Mashhad (50614/3).
Funding
The authors have not disclosed any funding.
Author information
Authors and Affiliations
Contributions
A.M and S.A contributed to the study conception and design. G.H and A.M contributed to Material preparation, data collection and analysis. The first draft of the manuscript was written by A.M and G.H. All authors read and approved the final manuscript
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Communicated by Muhammad Bilal.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Laliany, G., Jamehdar, S.A. & Makhdoumi, A. In vitro virulence activity of Pseudomonas aeruginosa, enhanced by either Acinetobacter baumannii or Enterococcus faecium through the polymicrobial interactions. Arch Microbiol 204, 709 (2022). https://doi.org/10.1007/s00203-022-03308-8
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00203-022-03308-8