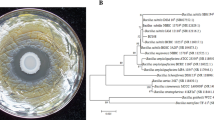
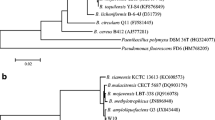
Abstract
Antimicrobial proteins and peptides are an alternative to current antibiotics. Here, we report an antimicrobial activity in a low-molecular-weight protein secreted naturally by Streptomyces lividans TK24 when glucose or glycerol were used as carbon sources. The antimicrobial activity was demonstrated against Bacillus subtilis, Bacillus cereus, Kokuria rhizophila, Clostridium sporogenes and Clavibacter michiganensis, causal pathogen of tomato bacterial canker; one of the most destructive bacterial diseases of this crop. The protein fraction with antimicrobial activity was identified and quantified by LC–MS/MS. From a total of 155 proteins, 11 were found to be within the range of 11.3–13.9 kDa of which four proteins were selected by functional analysis as possibly responsible for the antimicrobial activity. Protein fractionation, correlation analysis between antimicrobial activity and abundance of selected proteins, as well as transcriptional expression analysis, indicate that 50S ribosomal protein L19 is the main candidate responsible for antimicrobial activity.






Similar content being viewed by others
Data availability
The datasets generated during and analyzed during the current study are available from the corresponding author on reasonable request.
References
Brenner C (2002) Hint, Fhit, and GalT: Function, structure, evolution, and mechanism of three branches of the histidine triad superfamily of nucleotide hydrolases and transferases. Biochemistry 41:9003–9014. https://doi.org/10.1021/bi025942q
Butler MS, Blaskovich MA, Cooper MA (2017) Antibiotics in the clinical pipeline at the end of 2015. J Antibiot 70:3–24. https://doi.org/10.1038/ja.2016.72
Chen Y, Yao L, Wang Y et al (2021) Identification of ribosomal protein L30 as an uncharacterized antimicrobial protein. Dev Comp Immunol 120:104067. https://doi.org/10.1016/j.dci.2021.104067
Dahl C, Engels S, Pott-Sperling AS et al (2005) Novel genes of the dsr gene cluster and evidence for close interaction of Dsr proteins during sulfur oxidation in the phototrophic sulfur bacterium Allochromatium vinosum. J Bacteriol 187:1392–1404. https://doi.org/10.1128/JB.187.4.1392-1404.2005
Das R, Romi W, Das R et al (2018) Antimicrobial potentiality of actinobacteria isolated from two microbiologically unexplored forest ecosystems of Northeast India. BMC Microbiol 18:1–16. https://doi.org/10.1186/s12866-018-1215-7
Davies J (2006) Where have all the antibiotics gone? Can J Infect Dis Med Microbiol 17:287–290. https://doi.org/10.1155/2006/707296
De Carvalho KG, Bambirra FHS, Kruger MF et al (2010) Antimicrobial compounds produced by Lactobacillus sakei subsp. sakei 2a, a bacteriocinogenic strain isolated from a Brazilian meat product. J Ind Microbiol Biotechnol 37:381–390. https://doi.org/10.1007/s10295-009-0684-y
de Vos WM, Kuipers OP, Roelof Van Der Meer J, Siezen RJ (1995) Maturation pathway of nisin and other lantibiotics: post-translationally modified antimicrobial peptides exported by Gram-positive bacteria. Mol Microbiol 17:427–437. https://doi.org/10.1111/j.1365-2958.1995.mmi_17030427.x
García-Cano I, Rocha-Mendoza D, Ortega-Anaya J et al (2019) Lactic acid bacteria isolated from dairy products as potential producers of lipolytic, proteolytic and antibacterial proteins. Appl Microbiol Biotechnol 103:5243–5257. https://doi.org/10.1007/s00253-019-09844-6
Grewal S, Bhagat M, Vakhlu J (2014) Antimicrobial protein produced by Pseudomonas aeruginosa JU-Ch 1, with a broad spectrum of antimicrobial activity. Biocatal Agric Biotechnol 3:332–337. https://doi.org/10.1016/j.bcab.2014.04.006
Hancock REW, Diamond G (2000) The role of cationic antimicrobial peptides in innate host defences. Trends Microbiol 8:402–410. https://doi.org/10.1016/S0966-842X(00)01823-0
Hopfe M, Hegemann JH, Henrich B (2005) HinT proteins and their putative interaction partners in Mollicutes and Chlamydiaceae. BMC Microbiol 5:1–14. https://doi.org/10.1186/1471-2180-5-27
Hurtado-Rios JJ, Carrasco-Navarro U, Almanza-Pérez JC, Ponce-Alquicira E (2022) Ribosomes: the new role of ribosomal proteins as natural antimicrobials. Int J Mol Sci 23:9123. https://doi.org/10.3390/ijms23169123
Ishihama Y, Schmidt T, Rappsilber J et al (2008) Protein abundance profiling of the Escherichia coli cytosol. BMC Genomics 9:102. https://doi.org/10.1186/1471-2164-9-102
Jeffery CJ (1999) Moonlighning proteins. Trends Biochem Sci 24:8–11. https://doi.org/10.1016/S0968-0004(98)01335-8
Jeffery CJ (2017) Protein moonlighting: what is it, and why is it important? Phil Trans R Soc B 373:20160523. https://doi.org/10.1098/rstb.2016.0523
Jenssen H, Hamill P, Hancock REW (2006) Peptide antimicrobial agents. Clin Microbiol Rev 19:491–511. https://doi.org/10.1128/CMR.00056-05
Karaiskos I, Giamarellou H (2014) Multidrug-resistant and extensively drug-resistant Gram-negative pathogens: current and emerging therapeutic approaches. Expert Opin Pharmacother 15:1351–1370. https://doi.org/10.1517/14656566.2014.914172
Keller A, Nesvizhskii AI, Kolker E, Aebersold R (2002) Empirical statistical model to estimate the accuracy of peptide identifications made by MS/MS and database search. Anal Chem 74:5383–5392. https://doi.org/10.1021/ac025747h
Kieser T, Bibb MJ, Buttner MJ, et al (2000) Practical Streptomyces genetics. The John Innes Foundation, Norwich
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔCT method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Ma Z, Qu B, Yao L et al (2020) Identification and functional characterization of ribosomal protein S23 as a new member of antimicrobial protein. Dev Comp Immunol 110:103730. https://doi.org/10.1016/j.dci.2020.103730
Malik H, Sur B, Singhal N, Bihari V (2008) Antimicrobial protein from Streptomyces fulvissimus inhibitory to methicillin resistant Staphylococcus aureus. Indian J Exp Biol 46:254–257
Matsumoto H, Haniu H, Komori N (2019) Determination of protein molecular weights on SDS-PAGE. Methods Mol Biol 1855:101–105. https://doi.org/10.1007/978-1-4939-8793-1_10
Miao V, Davies J (2010) Actinobacteria: The good, the bad, and the ugly. Antonie Van Leeuwenhoek 98:143–150. https://doi.org/10.1007/s10482-010-9440-6
Nesvizhskii AI, Keller A, Kolker E, Aebersold R (2003) A statistical model for identifying proteins by tandem mass spectrometry. Anal Chem 75:4646–4658. https://doi.org/10.1021/ac0341261
Pidutti P, Federici F, Brandi J et al (2017) Purification and characterization of ribosomal proteins L27 and L30 having antimicrobial activity produced by the Lactobacillus salivarius SGL 03. J Appl Microbiol 124:398–407. https://doi.org/10.1111/jam.13646
Pott AS, Dahl C (1998) Sirohaem sulfite reductase and other proteins encoded by genes at the dsr locus of Chromatium vinosum are involved in the oxidation of intracellular sulfur. Microbiology 144:1881–1894. https://doi.org/10.1099/00221287-144-7-1881
Putsep K, Branden CI, Boman HG, Normark S (1999) Antibacterial peptide from H. pylori. Nature 398:671–672. https://doi.org/10.1038/19439
Qu B, Ma Z, Yao L et al (2020) Preserved antibacterial activity of ribosomal protein S15 during evolution. Mol Immunol 127:57–66. https://doi.org/10.1016/j.molimm.2020.08.024
Rodríguez JM (1996) Review: Antimicrobial spectrum, structure, properties and mode of action of nisin, a bacteriocin produced by Lactococcus lactis. Food Sci Technol Int 2:61–68. https://doi.org/10.1177/108201329600200202
Seo JK, Kim DG, Oh R et al (2017) Antimicrobial effect of the 60S ribosomal protein L29 (cgRPL29), purified from the gill of pacific oyster, Crassostrea gigas. Fish Shellfish Immunol 67:675–683. https://doi.org/10.1016/j.fsi.2017.06.058
Sidders B, Withers M, Kendall SL et al (2007) Quantification of global transcription patterns in prokaryotes using spotted microarrays. Genome Biol 8:R265. https://doi.org/10.1186/gb-2007-8-12-r265
Sierra JM, Fusté E, Rabanal F et al (2017) An overview of antimicrobial peptides and the latest advances in their development. Expert Opin Biol Ther 17:663–676. https://doi.org/10.1080/14712598.2017.1315402
Yaghoubi A, Khazaei M, Ghazvini K et al (2021) Peptides with dual antimicrobial-anticancer activity derived from the N-terminal region of H. pylori ribosomal protein L1 (RpL1). Int J Pept Res Ther 27:1057–1067. https://doi.org/10.1007/s10989-020-10150-3
Zhou X, Liao WJ, Liao JM et al (2015) Ribosomal proteins: functions beyond the ribosome. J Mol Cell Biol 7:92–104. https://doi.org/10.1093/jmcb/mjv014
Acknowledgements
This work was supported by “Programa Especial de Apoyo a la Investigación” Rectoría General, UAM, I21-2019. Fernanda J Calderón-de la Sancha thanks Consejo Nacional de Ciencia y Tecnología (CONACYT-México) for the scholarship (No. 718101). We thank Dr. Denis Faubert at the Proteomics Facility of the Institut de Recherches Cliniques de Montreal, Canada, for his assistance in the LC–MS/MS analysis. Finally, to the Divisional Laboratory of Molecular Biology (LDBM) of the Universidad Autónoma Metropolitana-Iztapalapa, México.
Funding
This work was supported by “Programa Especial de Apoyo a la Investigación” Rectoría General, UAM, I21-2019. Fernanda J Calderón-de la Sancha has received the doctoral scholarship No. 718101 from Consejo Nacional de Ciencia y Tecnología (CONACYT-Mexico).
Author information
Authors and Affiliations
Contributions
FJCS and AM participated in the study conception. Material preparation, data collection and analysis were performed by FJCS, UCN and GS. FJCS and UCN had full access to all the data in the study and take responsibility for the integrity of the data and accuracy of the data analysis. FJCS, UCN, JBG and AM drafted and revised the paper. All authors read and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Erko Stackebrandt.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Calderón-de la Sancha, F.J., Carrasco-Navarro, U., Santander, G. et al. Novel antimicrobial activity of protein produced by Streptomyces lividans TK24 against the phytopathogen Clavibacter michiganensis. Arch Microbiol 204, 687 (2022). https://doi.org/10.1007/s00203-022-03290-1
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00203-022-03290-1