Abstract
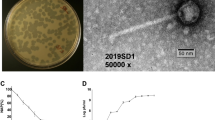
A novel F-specific RNA bacteriophage (FRNAPH) YM1, affiliating to genogroup I (GI) of Levivirus, is isolated for the first time from human stool samples using double-layer agar plates with the Escherichia coli ATCC700891 as the host. The complete genomic sequence of YM1 is 3551 nt in length, obtained through next-generation sequencing, and contains four genes encoding for maturation protein, coat protein, lysis protein, and RNA-dependent RNA polymerase (RdRp). The genomic sequence of YM1 shares the highest similarity of 95.3% with that of a GI FRNAPH DL16 isolated from surface water of Great Bay. The YM1 possesses a non-enveloped, icosahedral virion of 23 ± 0.45 nm in diameter. One-step growth curve analysis shows that the burst time of YM1 is 30 min post-infection (p.i.) with the average burst size of 264 PFU/cell. The YM1 lyses only E. coli strains tested, revealing high host specificity. This newly discovered phage may serve as a candidate for viral indicator to monitor human enteric virus, especially norovirus, contamination in the environments.


Similar content being viewed by others
References
Amarasiri M, Hashiba S, Miura T, Nakagomi T, Nakagomi O, Ishii S, Okabe S, Sano D (2016) Bacterial histo-blood group antigens contributing to genotype-dependent removal of human noroviruses with a microfiltration membrane. Water Res 95:383–391. https://doi.org/10.1016/j.watres.2016.04.018
Barrios ME, Blanco Fernández MD, Cammarata RV, Torres C, Mbayed VA (2018) Viral tools for detection of fecal contamination and microbial source tracking in wastewater from food industries and domestic sewage. J Virol Methods 262:79–88. https://doi.org/10.1016/j.jviromet.2018.10.002
Bolger AM, Lohse M, Usadel B (2014) Trimmomatic: a flexible trimmer for Illumina sequence data. Bioinformatics 30(15):2114–2120. https://doi.org/10.1093/bioinformatics/btu170
Bollback JP, Huelsenbeck JP (2001) Phylogeny, genome evolution, and host specificity of single-stranded RNA bacteriophage (family Leviviridae). J Mol Evol 52:117–128. https://doi.org/10.1007/s002390010140
Butler AJ, Thomas MK, Pintar KD (2015) Expert elicitation as a means to attribute 28 enteric pathogens to foodborne, waterborne, animal contact, and person-to-person transmission routes in Canada. Foodborne Pathog Dis 12(4):335–344. https://doi.org/10.1089/fpd.2014.1856
Crawford EM, Gesteland RF (1964) The adsorption of bacteriophage R-17. Virology 22(1):165–167. https://doi.org/10.1016/0042-6822(64)90063-7
Cross T, Schoff C, Chudoff D, Graves L, Broomell H, Terry K, Farina J, Correa A, Shade D, Dunbar D (2015) An optimized enrichment technique for the isolation of Arthrobacter bacteriophage species from soil sample isolates. J vis Exp 9(98):52781. https://doi.org/10.3791/52781
De Carlo S, Harris JR (2011) Negative staining and cryo-negative staining of macromolecules and viruses for TEM. Micron 42(2):117–131. https://doi.org/10.1016/j.micron.2010.06.003
de Graaf M, van Beek J, Koopmans MP (2016) Human norovirus transmission and evolution in a changing world. Nat Rev Microbiol 14(7):421–433. https://doi.org/10.1038/nrmicro.2016.48
Debartolomeis J, Cabelli VJ (1991) Evaluation of an Escherichia coli host strain for enumeration of F male-specific bacteriophages. Appl Environ Microbiol 57:1301–1305. https://doi.org/10.1128/aem.57.5.1301-1305.1991
Friedman SD, Cooper EM, Casanova L, Sobsey MD, Genthner FJ (2009) A reverse transcription-PCR assay to distinguish the four genogroups of male-specific (F+) RNA coliphages. J Virol Methods 159(1):47–52. https://doi.org/10.1016/j.jviromet.2009.02.028
Golmohammadi R, Valegård K, Fridborg K, Liljas L (1993) The refined structure of bacteriophage MS2 at 2.8 A resolution. J Mol Biol 234(3):620–639. https://doi.org/10.1006/jmbi.1993.1616
Groeneveld H, Oudot F, van Duin JV (1996) RNA phage KU1 has an insertion of 18 nucleotides in the start codon of its lysis gene. Virology 218(1):141–147. https://doi.org/10.1006/viro.1996.0174
Guo P, Yu Y, Pan Y, Yan S, Wang Y (2018) Design and evaluation of nested PCR primers for specific detection of genogroup I noroviruses in oysters. Mol Cell Probes 40:40–43. https://doi.org/10.1016/j.mcp.2018.06.002
Haramoto E, Otagiri M, Morita H, Kitajima M (2012) Genogroup distribution of F-specific coliphages in wastewater and river water in the Kofu basin in Japan. Lett Appl Microbiol 54(4):367–373. https://doi.org/10.1111/j.1472-765X.2012.03221.x
Hartard C, Rivet R, Banas S, Gantzer C (2015) Occurrence of and sequence variation among F-specific RNA bacteriophage subgroups in feces and wastewater of urban and animal origins. Appl Environ Microbiol 81:6505–6515. https://doi.org/10.1128/aem.01905-15
Hartard C, Banas S, Loutreul J, Rincé A, Benoit F, Boudaud N, Gantzer C (2016) Relevance of F-Specific RNA bacteriophages in assessing human norovirus risk in shellfish and environmental waters. Appl Environ Microbiol 82:5709–5719. https://doi.org/10.1128/aem.01528-16
Hartard C, Leclerc M, Rivet R, Maul A, Loutreul J, Banas S, Boudaud N, Gantzer C (2017) F-Specific RNA bacteriophages, especially members of subgroup II, should be reconsidered as good indicators of viral pollution of oysters. Appl Environ Microbiol 84:1817–1866. https://doi.org/10.1128/AEM.01866-17
Harwood VJ, Staley C, Badgley BD, Borges K, Korajkic A (2014) Microbial source tracking markers for detection of fecal contamination in environmental waters: relationships between pathogens and human health outcomes. FEMS Microbiol Rev 38(1):1–40. https://doi.org/10.1111/1574-6976
Hodgson KR, Torok VA, Turnbull AR (2017) Bacteriophages as enteric viral indicators in bivalve mollusc management. Food Microbiol 65:284–293. https://doi.org/10.1016/j.fm.2017.03.003
Hurwitz BL, Deng L, Poulos BT, Sullivan MB (2013) Evaluation of methods to concentrate and purify ocean virus communities through comparative, replicated metagenomics. Environ Microbiol 15(5):1428–1440. https://doi.org/10.1111/j.1462-2920.2012.02836.x
Kazaks A, Voronkova T, Rumnieks J, Dishlers A, Tars K (2011) Genome structure of caulobacter phage phiCb5. J Virol 85(9):4628–4631. https://doi.org/10.1128/jvi.02256-10
Kearse M, Moir R, Wilson A, Stones-Havas S, Cheung M, Sturrock S, Buxton S, Cooper A, Markowitz S, Duran C, Thierer T, Ashton B, Meintjes P, Drummond A (2012) Geneious basic: an integrated and extendable desktop software platform for the organization and analysis of sequence data. Bioinformatics 28(12):1647–1649. https://doi.org/10.1093/bioinformatics/bts199
Klovins J, Overbeek GP, van den Worm SHE, Ackermann HW, van Duin J (2002) Nucleotide sequence of a ssRNA phage from Acinetobacter: kinship to coliphages. J Gen Virol 83:1523–1533. https://doi.org/10.1099/0022-1317-83-6-1523
Langmead B, Salzberg SL (2012) Fast gapped-read alignment with Bowtie 2. Nat Methods 9(4):357–359. https://doi.org/10.1038/nmeth.1923
Lee JE, Lee H, Cho YH, Hur HG, Ko G (2011) F+ RNA coliphage-based microbial source tracking in water resources of South Korea. Sci Total Environ 412–413:127–131. https://doi.org/10.1016/j.scitotenv.2011.09.061
Li D, Liu CM, Luo R, Sadakane K, Lam TW (2015) MEGAHIT: an ultra-fast single-node solution for large and complex metagenomics assembly via succinct de Bruijn graph. Bioinformatics 31(10):1674–1676. https://doi.org/10.1093/bioinformatics/btv033
Love DC, Sobsey MD (2007) Simple and rapid F+ coliphage culture, latex agglutination, and typing assay to detect and source track fecal contamination. Appl Environ Microbiol 73(13):4110–4118. https://doi.org/10.1128/aem.02546-06
Masson P, Hulo C, De Castro E, Bitter H, Gruenbaum L, Essioux L, Bougueleret L, Xenarios I, Le Mercier P (2013) ViralZone: recent updates to the virus knowledge resource. Nucleic Acids Res 41:D579-583. https://doi.org/10.1093/nar/gks1220
Murphy FA, Fauquet CM, Bishop DHL, Ghabrial SA, Jarvis AW, Martelli GP, Mayo MA, Summers MD (1995) Virus taxonomy, classification and nomenclature of viruses (Sixth Report of the international committee on taxonomy of viruses). Arch Virol 10:412–414
Ogorzaly L, Gantzer C (2006) Development of real-time RT-PCR methods for specific detection of F-specific RNA bacteriophage genogroups: application to urban raw wastewater. J Virol Methods 138:131–139. https://doi.org/10.1016/j.jviromet.2006.08.004
Ogorzaly L, Tissier A, Bertrand I, Maul A, Gantzer C (2009) Relationship between F-specific RNA phage genogroups, faecal pollution indicators and human adenoviruses in river water. Water Res 43(5):1257–1264. https://doi.org/10.1016/j.watres.2008.12.011
Ogorzaly L, Bertrand I, Paris M, Maul A, Gantzer C (2010) Occurrence, survival, and persistence of human adenoviruses and F-specific RNA phages in raw groundwater. Appl Environ Microbiol 76(24):8019–8025. https://doi.org/10.1128/AEM.00917-10
Quinlan AR, Hall IM (2010) BEDTools: a flexible suite of utilities for comparing genomic features. Bioinformatics 26(6):841–842. https://doi.org/10.1093/bioinformatics/btq033
Ravva SV, Sarreal CZ, Cooley MB (2015) Male-specific coliphages for source tracking fecal contamination in surface waters and prevalence of Shiga-toxigenic Escherichia coli in a major produce production region of the Central Coast of California. Environ Sci Process Impacts 17(7):1249–1256. https://doi.org/10.1039/c4em00537f
Robilotti E, Deresinski S, Pinsky BA (2015) Norovirus. Clin Microbiol Rev 28(1):134–164. https://doi.org/10.1128/CMR.00075-14
Ruokoranta TM, Grahn AM, Ravantti JJ, Poranen MM, Bamford DH (2006) Complete genome sequence of the broad host range single-stranded RNA phage PRR1 places it in the Levivirus genus with characteristics shared with Alloleviviruses. J Virol 80(18):9326–9330. https://doi.org/10.1128/jvi.01005-06
Schaeffer J, Le Saux JC, Lora M, Atmar RL, Le Guyader FS (2013) Norovirus contamination on French marketed oysters. Int J Food Microbiol 166(2):244–248. https://doi.org/10.1016/j.ijfoodmicro.2013.07.022
Stutman HR (1994) Salmonella, Shigella, and Campylobacter: common bacterial causes of infectious diarrhea. Pediatr Ann 23:538–543. https://doi.org/10.3928/0090-4481-19941001-07
Topka G, Bloch S, Nejman-Faleńczyk B, Gąsior T, Jurczak-Kurek A, Necel A, Dydecka A, Richert M, Węgrzyn G, Węgrzyn A (2018) Characterization of bacteriophage vB-EcoS-95, isolated from urban sewage and revealing extremely rapid lytic development. Front Microbiol 9:3326. https://doi.org/10.3389/fmicb.2018.03326
Towner KJ (2009) Acinetobacter: an old friend, but a new enemy. J Hosp Infect 73(4):355–363. https://doi.org/10.1016/j.jhin.2009.03.032
Wolf YI, Silas S, Wang Y, Wu S, Bocek M, Kazlauskas D, Krupovic M, Fire A, Dolja VV, Koonin EV (2020) Doubling of the known set of RNA viruses by metagenomic analysis of an aquatic virome. Nat Microbiol 5(10):1262–1270. https://doi.org/10.1038/s41564-020-0755-4
Wu GD, Bushmanc FD, Lewis JD (2013) Diet, the human gut microbiota, and IBD. Anaerobe 24:117–120. https://doi.org/10.1016/j.anaerobe.2013.03.011
Yu Y, Cai H, Hu L, Lei R, Pan Y, Yan S, Wang Y (2015) Molecular epidemiology of oyster-related human noroviruses and their global genetic diversity and temporal-geographical distribution from 1983 to 2014. Appl Environ Microbiol 81:7615–7624. https://doi.org/10.1128/aem.01729-15
Acknowledgements
We are grateful to Shijiazhuang Center for Disease Control and Prevention for providing the strain ATCC700891, and to Dr. Song Cong from Donghua University for providing the strain ATCC15597.
Funding
This study was funded by the National key R&D Program of China (2017YFC1600703) and the National Natural Science Foundation of China (31601570, 41876195).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Communicated by Erko Stackebrandt.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yang, M., Yu, Y. & Wang, Y. Characterization of a novel group I F-specific RNA bacteriophage isolated from human stool. Arch Microbiol 203, 4065–4072 (2021). https://doi.org/10.1007/s00203-021-02402-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-021-02402-7