Abstract
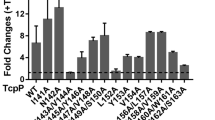
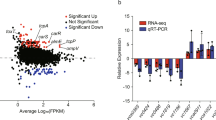
The epidemic diarrheal disease cholera is caused by the Gram-negative bacterium Vibrio cholerae. V. cholerae virulence factors include the toxin-coregulated pilus (TCP) and cholera toxin, which are major factors responsible for host colonization and production of diarrhea. Expression of cholera toxin and TCP genes is controlled by the ToxR regulon. The ToxR regulon includes the transcriptional activators ToxR, TcpP, and ToxT. ToxT directly initiates transcription of the cholera toxin and TCP genes. TcpP and ToxR are necessary for expression of toxT. TcpP and ToxR activity requires TcpH and ToxS, respectively. Additionally, ToxR is able to directly initiate transcription of the cholera toxin genes independent of TcpP and ToxT. TCP is required early in infection to colonize the small intestine, then cholera toxin is expressed later in infection to produce diarrhea. We tested whether stringent response, the low nutrient stress response, was involved in regulation of virulence genes. Using an infant mouse model, we found that V. cholerae strains with deletions of the stringent response genes were unable to colonize the small intestine. We further tested these stringent response-null mutants and found that stringent response was necessary for TCP expression, although effects on cholera toxin expression were not significant. We then tested whether stringent response regulation of TCP occurred through the ToxR regulon. We found that stringent response induced toxT and tcpPH expression, while repressing toxRS. This differential regulation of ToxR and TcpP may explain the differential expression of TCP and cholera toxin in vivo.






Similar content being viewed by others
References
Abuaita BH, Withey JH (2009) Bicarbonate induces Vibrio cholerae virulence gene expression by enhancing ToxT activity. Infect Immun 77:4111–4120. https://doi.org/10.1128/IAI.00409-09
Acosta N, Pukatzki S, Raivio TL (2015) The Cpx system regulates virulence gene expression in Vibrio cholerae. Infect Immun 83:2396–2408. https://doi.org/10.1128/IAI.03056-14
Almagro-Moreno S, Root MZ, Taylor RK (2015) Role of ToxS in the proteolytic cascade of virulence regulator ToxR in Vibrio cholerae. Mol Microbiol 98:963–976. https://doi.org/10.1111/mmi.13170
Basu P, Bhadra RK (2019) Post-transcriptional regulation of cholera toxin production in Vibrio cholerae by the stringent response regulator DksA. Microbiology 165:102–112. https://doi.org/10.1099/mic.0.000743
Bina J, Zhu J, Dziejman M, Faruque S, Calderwood S, Mekalanos J (2003) ToxR regulon of Vibrio cholerae and its expression in vibrios shed by cholera patients. Proc Natl Acad Sci USA 100:2801–2806. https://doi.org/10.1073/pnas.2628026100
Bina XR, Taylor DL, Vikram A, Ante VM, Bina JE (2013) Vibrio cholerae ToxR downregulates virulence factor production in response to cyclo(Phe-Pro). MBio 4:e00366–e1313. https://doi.org/10.1128/mBio.00366-13
Brown RC, Taylor RK (1995) Organization of tcp, acf, and toxT genes within a ToxT-dependent operon. Mol Microbiol 16:425–439
Burnette WN (1994) AB5 ADP-ribosylating toxins: comparative anatomy and physiology. Structure 2:151–158
Butler SM, Camilli A (2004) Both chemotaxis and net motility greatly influence the infectivity of Vibrio cholerae. Proc Natl Acad Sci USA 101:5018–5023. https://doi.org/10.1073/pnas.0308052101
Clemens JD, Nair GB, Ahmed T, Qadri F, Holmgren J (2017) Cholera Lancet 390:1539–1549. https://doi.org/10.1016/S0140-6736(17)30559-7
Conner JG, Teschler JK, Jones CJ, Yildiz FH (2016) Staying alive: Vibrio cholerae's cycle of environmental survival. Transm Dissemin Microbiol Spectr. https://doi.org/10.1128/microbiolspec.VMBF-0015-2015
Crawford JA, Krukonis ES, DiRita VJ (2003) Membrane localization of the ToxR winged-helix domain is required for TcpP-mediated virulence gene activation in Vibrio cholerae. Mol Microbiol 47:1459–1473
Dalebroux ZD, Swanson MS (2012) ppGpp: magic beyond RNA polymerase. Nat Rev Microbiol 10:203–212. https://doi.org/10.1038/nrmicro2720
Das B, Bhadra RK (2008) Molecular characterization of vibrio cholerae DeltarelA DeltaspoT double mutants. Arch Microbiol 189:227–238. https://doi.org/10.1007/s00203-007-0312-z
Das B, Pal RR, Bag S, Bhadra RK (2009) Stringent response in Vibrio cholerae: genetic analysis of spoT gene function and identification of a novel (p)ppGpp synthetase gene. Mol Microbiol 72:380–398. https://doi.org/10.1111/j.1365-2958.2009.06653.x
DiRita VJ, Neely M, Taylor RK, Bruss PM (1996) Differential expression of the ToxR regulon in classical and E1 Tor biotypes of Vibrio cholerae is due to biotype-specific control over toxT expression. Proc Natl Acad Sci USA 93:7991–7995. https://doi.org/10.1073/pnas.93.15.7991
Dorman MJ, Dorman CJ (2018) Regulatory hierarchies controlling virulence gene expression in Shigella flexneri and Vibrio cholerae. Front Microbiol 9:2686. https://doi.org/10.3389/fmicb.2018.02686
Fullner KJ, Mekalanos JJ (1999) Genetic characterization of a new type IV-A pilus gene cluster found in both classical and El Tor biotypes of Vibrio cholerae. Infect Immun 67:1393–1404
Gardel CL, Mekalanos JJ (1994) Regulation of cholera toxin by temperature, pH, and osmolarity. Methods Enzymol 235:517–526
Gourse RL, Chen AY, Gopalkrishnan S, Sanchez-Vazquez P, Myers A, Ross W (2018) Transcriptional Responses to ppGpp and DksA. Annu Rev Microbiol 72:163–184. https://doi.org/10.1146/annurev-micro-090817-062444
Haralalka S, Nandi S, Bhadra RK (2003) Mutation in the relA gene of Vibrio cholerae affects in vitro and in vivo expression of virulence factors. J Bacteriol 185:4672–4682
Hase CC, Mekalanos JJ (1998) TcpP protein is a positive regulator of virulence gene expression in Vibrio cholerae. Proc Natl Acad Sci USA 95:730–734. https://doi.org/10.1073/pnas.95.2.730
He H, Cooper JN, Mishra A, Raskin DM (2012) Stringent response regulation of biofilm formation in Vibrio cholerae. J Bacteriol 194:2962–2972. https://doi.org/10.1128/JB.00014-12
Howard MF, Bina XR, Bina JE (2019) Indole inhibits ToxR regulon expression in Vibrio cholerae. Infect Immun 87:e00776–e1718. https://doi.org/10.1128/IAI.00776-18
Hung DT, Mekalanos JJ (2005) Bile acids induce cholera toxin expression in Vibrio cholerae in a ToxT-independent manner. Proc Natl Acad Sci USA 102:3028–3033. https://doi.org/10.1073/pnas.0409559102
Iwanaga M, Yamamoto K (1985) New medium for the production of cholera toxin by Vibrio cholerae O1 biotype El Tor. J Clin Microbiol 22:405–408
Kanjee U, Ogata K, Houry WA (2012) Direct binding targets of the stringent response alarmone (p)ppGpp. Mol Microbiol 85:1029–1043. https://doi.org/10.1111/j.1365-2958.2012.08177.x
Kanjilal S, Citorik R, LaRocque RC, Ramoni MF, Calderwood SB (2010) A systems biology approach to modeling vibrio cholerae gene expression under virulence-inducing conditions. J Bacteriol 192:4300–4310. https://doi.org/10.1128/JB.00182-10
Kovacikova G, Skorupski K (1999) A Vibrio cholerae LysR homolog, AphB, cooperates with AphA at the tcpPH promoter to activate expression of the ToxR virulence cascade. J Bacteriol 181:4250–4256
Krukonis ES, Yu RR, Dirita VJ (2000) The Vibrio cholerae ToxR/TcpP/ToxT virulence cascade: distinct roles for two membrane-localized transcriptional activators on a single promoter. Mol Microbiol 38:67–84
Larocque RC et al (2005) Transcriptional profiling of Vibrio cholerae recovered directly from patient specimens during early and late stages of human infection. Infect Immun 73:4488–4493. https://doi.org/10.1128/IAI.73.8.4488-4493.2005
Lippi D, Gotuzzo E, Caini S (2016) Cholera Microbiol Spectr 4 doi:10.1128/microbiolspec.PoH-0012–2015
Matson JS, DiRita VJ (2005) Degradation of the membrane-localized virulence activator TcpP by the YaeL protease in Vibrio cholerae. Proc Natl Acad Sci USA 102:16403–16408. https://doi.org/10.1073/pnas.0505818102
Matson JS, Withey JH, DiRita VJ (2007) Regulatory networks controlling Vibrio cholerae virulence gene expression. Infect Immun 75:5542–5549. https://doi.org/10.1128/IAI.01094-07
Matz C, McDougald D, Moreno AM, Yung PY, Yildiz FH, Kjelleberg S (2005) Biofilm formation and phenotypic variation enhance predation-driven persistence of Vibrio cholerae. Proc Natl Acad Sci USA 102:16819–16824. https://doi.org/10.1073/pnas.0505350102
Mekalanos JJ, Swartz DJ, Pearson GD, Harford N, Groyne F, de Wilde M (1983) Cholera toxin genes: nucleotide sequence, deletion analysis and vaccine development. Nature 306:551–557
Metcalf WW, Jiang W, Daniels LL, Kim SK, Haldimann A, Wanner BL (1996) Conditionally replicative and conjugative plasmids carrying lacZ alpha for cloning, mutagenesis, and allele replacement in bacteria. Plasmid 35:1–13. https://doi.org/10.1006/plas.1996.0001
Miller VL, Mekalanos JJ (1984) Synthesis of cholera toxin is positively regulated at the transcriptional level by toxR. Proc Natl Acad Sci USA 81:3471–3475
Miller VL, Mekalanos JJ (1988) A novel suicide vector and its use in construction of insertion mutations: osmoregulation of outer membrane proteins and virulence determinants in Vibrio cholerae requires toxR. J Bacteriol 170:2575–2583
Oh YT et al (2014) Cholera toxin production during anaerobic trimethylamine N-oxide respiration is mediated by stringent response in Vibrio cholerae. J Biol Chem 289:13232–13242. https://doi.org/10.1074/jbc.M113.540088
Pal RR, Bag S, Dasgupta S, Das B, Bhadra RK (2012) Functional characterization of the stringent response regulatory gene dksA of Vibrio cholerae and its role in modulation of virulence phenotypes. J Bacteriol 194:5638–5648. https://doi.org/10.1128/JB.00518-12
Pal RR, Das B, Dasgupta S, Bhadra RK (2011) Genetic components of stringent response in Vibrio cholerae. Indian J Med Res 133:212–217
Peterson KM, Gellings PS (2018) Multiple intraintestinal signals coordinate the regulation of Vibrio cholerae virulence determinants. Pathog Dis. https://doi.org/10.1093/femspd/ftx126
Potrykus K, Cashel M (2008) (p)ppGpp: still magical? Annu Rev Microbiol 62:35–51. https://doi.org/10.1146/annurev.micro.62.081307.162903
Raskin DM, Judson N, Mekalanos JJ (2007) Regulation of the stringent response is the essential function of the conserved bacterial G protein CgtA in Vibrio cholerae. Proc Natl Acad Sci USA 104:4636–4641. https://doi.org/10.1073/pnas.0611650104
Ronneau S, Hallez R (2019) Make and break the alarmone: regulation of (p)ppGpp synthetase/hydrolase enzymes in bacteria. FEMS Microbiol Rev. https://doi.org/10.1093/femsre/fuz009
Sanchez J, Holmgren J (2011) Cholera toxin—a foe & a friend. Indian J Med Res 133:153–163
Silva AJ, Benitez JA (2006) A Vibrio cholerae relaxed (relA) mutant expresses major virulence factors, exhibits biofilm formation and motility, and colonizes the suckling mouse intestine. J Bacteriol 188:794–800. https://doi.org/10.1128/JB.188.2.794-800.2006
Steinchen W, Bange G (2016) The magic dance of the alarmones (p)ppGpp. Mol Microbiol 101:531–544. https://doi.org/10.1111/mmi.13412
Tamayo R, Patimalla B, Camilli A (2010) Growth in a biofilm induces a hyperinfectious phenotype in Vibrio cholerae. Infect Immun 78:3560–3569. https://doi.org/10.1128/IAI.00048-10
Thelin KH, Taylor RK (1996) Toxin-coregulated pilus, but not mannose-sensitive hemagglutinin, is required for colonization by Vibrio cholerae O1 El Tor biotype and O139 strains. Infect Immun 64:2853–2856
Weber GG, Klose KE (2011) The complexity of ToxT-dependent transcription in Vibrio cholerae. Indian J Med Res 133:201–206
Xu X, Stern AM, Liu Z, Kan B, Zhu J (2010) Virulence regulator AphB enhances toxR transcription in Vibrio cholerae. BMC Microbiol 10:3. https://doi.org/10.1186/1471-2180-10-3
Zhu J, Mekalanos JJ (2003) Quorum sensing-dependent biofilms enhance colonization in Vibrio cholerae. Dev Cell 5:647–656
Acknowledgements
We thank Jim Bina for the gift of the pWM91-tcpPH and pMD1 plasmids. We also thank Swathi Mall for plasmid and strain construction and Samina Akbar for comments on the manuscript.
Author information
Authors and Affiliations
Contributions
Conceptualization, methodology, validation, formal analysis, investigation, writing of the manuscript, and visualization were performed by David Raskin; methodology, validation, and investigation were performed by Huajun He and Arunima Mishra; investigation was performed by Zachary Lundy.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Ethical approval
The animal studies performed in this work were approved by Houston Methodist Research Institute IACUC, the institution where Huajun He and David Raskin were employed when the mouse studies were performed.
Additional information
Communicated by Erko Stackebrandt.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Raskin, D.M., Mishra, A., He, H. et al. Stringent response interacts with the ToxR regulon to regulate Vibrio cholerae virulence factor expression. Arch Microbiol 202, 1359–1368 (2020). https://doi.org/10.1007/s00203-020-01847-6
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-020-01847-6