Abstract
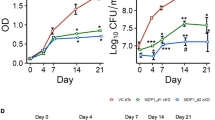
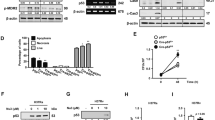
Genomic instability resulting from oxidative stress responses may be traced to chromosomal aberration. Oxidative stress suggests an imbalance between the systemic manifestation of reactive free radicals and biological system’s ability to repair resulting DNA damage and chromosomal aberration. Bacterial infection associated insult is considered as one of the major factors leading to such stress conditions. To study free radical responses by host cells, RAW 264.7 macrophages were infected with non-pathogenic M. smegmatis mc2155 at different time points. The infection process was followed up with an assessment of free radical stress, cytokine, toll-like receptors (TLRs) and the resulting DNA damage profiles. Results of CFU count showed that maximum infection in macrophages was achieved after 9 h of infection. Host responses to the infection across different time periods were validated from nitric oxide quantification and expression of iNOS and were plotted at regular intervals. IL-10 and TNF-α expression profile at protein and mRNA level showed a heightened pro-inflammatory response by host macrophages to combat M. smegmatis infection. The expression of TLR4, a receptor for recognition of mycobacteria, in infected macrophages reached the highest level at 9 h of infection. Furthermore, comet tail length, micronuclei and γ-H2AX foci recorded the highest level at 9 h of infection, pointing to the fact that breakage in DNA double strands in macrophage reaches its peak at 9 h of infection. In contrast, treatment with ROS inhibitor N-acetyl-l-cysteine (NAC) prevented host cell death through reduction in oxidative stress and DNA damage response during M. smegmatis infection. Therefore, it can be concluded that enhanced oxidative stress response in M. smegmatis infected macrophages might be correlated with DNA damage response.






Similar content being viewed by others
References
Anes E, Peyron P, Staali L, Jordao L, Gutierrez MG, Kress H, Hagedorn M, Maridonneau-Parini I, Skinner MA, Wildeman AG, Kalamidas SA, Kuehnel M, Griffiths G (2006) Dynamic life and death interactions between Mycobacterium smegmatis and J774 macrophages. Cell Microbiol 8:939–960
Anuja K, Roy S, Ghosh C, Gupta P, Bhattacharjee S, Banerjee B (2016) Prolonged inflammatory microenvironment is crucial for pro-neoplastic growth and genome instability: a detailed review. Inflamm Res 66:119–128
Bogdan C, Röllinghoff M, Diefenbach A (2000) Reactive oxygen and reactive nitrogen intermediates in innate and specific immunity. Curr Opin Immunol 12:64–76
Butcher LD, den Hartog G, Ernst PB, Crowe SE (2017) Oxidative stress resulting from Helicobacter pylori infection contributes to gastric carcinogenesis. Cell Mol Gastroenterol Hepatol 3:316–322
Chakraborty S, Srivastava A, Jha MK, Nair A, Pandey SP, Srivastava N, Kumari S, Singh S, Krishnasastry MV, Saha B (2015) Inhibition of CD40-induced N-Ras activation reduces leishmania major infection. J Immunol 194:3852–3860
Clutton SM, Townsend KMS, Walker C, Ansell JD, Wright EG (1996) Radiation-induced genomic instability and persisting oxidative stress in primary bone marrow cultures. Carcinogenesis 17:1633–1639
Da Silva ALG, Bresciani MJ, Karnopp TE, Weber AF, Ellwanger JH, Henriques JAP, Valim ARDM, Possuelo LG (2015) DNA damage and cellular abnormalities in tuberculosis, lung cancer and chronic obstructive pulmonary disease. Multidiscip Respir Med 10:38
Das A, Jawed JJ, Das MC, Sandhu P, De UC, Dinda B, Akhter Y, Bhattacharjee S (2017) Antileishmanial and immunomodulatory activities of lupeol, a triterpene compound isolated from Sterculia villosa. Int J Antimicrob Agents 50:512–522
Dil N, Qureshi MA (2002) Differential expression of inducible nitric oxide synthase is associated with differential Toll-like receptor-4 expression in chicken macrophages from different genetic backgrounds. Vet Immunol Immunopathol 84:191–207
Dye C, Williams BG (2010) The population dynamics and control of tuberculosis. Science 328:856–861
Fang FC (2011) Antimicrobial actions of reactive oxygen species. MBio 2:e00141–e00111
Fenech M (2007) Cytokinesis-block micronucleus cytome assay. Nat Protoc 2:1084
Fenech M, Kirsch-Volders M, Natarajan AT, Surralles J, Crott JW, Parry J, Norppa H, Eastmond DA, Tucker JD, Thomas P (2011) Molecular mechanisms of micronucleus, nucleoplasmic bridge and nuclear bud formation in mammalian and human cells. Mutagenesis 26:125–132
Forman HJ, Torres M (2002) Reactive oxygen species and cell signaling: respiratory burst in macrophage signaling. Am J Respir Crit Care Med 166:S4–S8
Guirado E, Mbawuike U, Keiser TL, Arcos J, Azad AK, Wang SH, Schlesinger LS (2015) Characterization of host and microbial determinants in individuals with latent tuberculosis infection using a human granuloma model. MBio 6:e02537–e02514
Hemnani T, Parihar MS (1998) Reactive oxygen species and oxidative DNA damage. Indian J Physiol Pharmacol 42:440–452
Hibbs JB Jr, Taintor RR, Vavrin Z, Rachlin EM (1988) Nitric oxide: a cytotoxic activated macrophage effector molecule. Biochem Biophys Res Commun 157:87–94
Kuo LJ, Yang LX (2008) γ-H2AX-a novel biomarker for DNA double-strand breaks. In vivo 22:305–309
Limoli CL, Giedzinski E (2003) Induction of chromosomal instability by chronic oxidative stress. Neoplasia 5:339–346
Lopez-Revuelta A, Sanchez-Gallego JI, Hernandez-Hernandez A, Sanchez-Yague J, Llanillo M (2005) Increase in vulnerability to oxidative damage in cholesterolmodified erythrocytes exposed to t-BuOOH. Biochim Biophys Acta 1734:74–85
MacMicking J, Xie QW, Nathan C (1997) Nitric oxide and macrophage function. Annu Rev Immunol 15:323–350
Majumder N, Dey R, Mathur RK, Datta S, Maitra M, Ghosh S, Saha B, Majumdar S (2006) An unusual pro-inflammatory role of interleukin-10 induced by arabinosylated lipoarabinomannan in murine peritoneal macrophages. Glycoconj J 23:675–686
Mohanty S, Jena P, Mehta R, Pati R, Banerjee B, Patil S, Sonawane A (2013) Cationic antimicrobial peptides and biogenic silver nanoparticles kill mycobacteria without eliciting DNA damage and cytotoxicity in mouse macrophages. Antimicrob Agents Chemother 57:3688–3698
Mohanty S, Molin MD, Ganguli G, Padhi A, Jena P, Selchow M, Sengupta S, Meuli M, Sander P, Sonawane A (2016) Mycobacterium tuberculosis EsxO (Rv2346c) promotes bacillary survival by inducing oxidative stress mediated genomic instabilityin macrophages. Tuberculosis 96:44–57
Naik SK, Mohanty S, Padhi A, Pati R, Sonawane A (2014) Evaluation of antibacterial and cytotoxic activity of Artemisia nilagirica and Murraya koenigii leaf extracts against mycobacteria and macrophages. BMC Complement Altern Med 14:87
Nita M, Grzybowski A (2016) The role of the reactive oxygen species and oxidative stress in the pathomechanism of the age-related ocular diseases and other pathologies of the anterior and posterior eye segments in adults. Oxid Med Cell Longev 2016:3164734
Qin L, Li G, Qian X, Liu Y, Wu X, Liu B, Hong JS, Block ML (2005) Interactive role of the toll-like receptor 4 and reactive oxygen species in LPS-induced microglia activation. Glia 52:78–84
Reiling N, Blumenthal A, Flad HD, Ernst M, Ehlers S (2001) Mycobacteria-induced TNF-α and IL-10 formation by human macrophages is differentially regulated at the level of mitogen-activated protein kinase activity. J Immunol 167:3339–3345
Rothschild BM, Martin LD, Lev G, Bercovier H, Bar-Gal GK, Greenblatt C, Donoghue H, Spigelman M, Brittain D (2001) Mycobacterium tuberculosis complex DNA from an extinct bison dated 17,000 years before the present. Clin Infect Dis 33:305–311
Roy S, Kar M, Roy S, Saha A, Padhi S, Banerjee B (2018) Role of β-catenin in cisplatin resistance, relapse and prognosis of head and neck squamous cell carcinoma. Cell Oncol 41:185–200
Rutkowski M, Grzegorczyk K (2007) Modifications of spectrophotometric methods for antioxidative vitamins determination convenient in analytic practice. Acta Sci Pol Technol Aliment 6:17–28
Ryan KA, Smith MF, Sanders MK, Ernst PB (2004) Reactive oxygen and nitrogen species differentially regulate Toll-like receptor 4-mediated activation of NF-κB and interleukin-8 expression. Infect Immun 72:2123–2130
Singh NP, McCoy MT, Tice RR, Schneider EL (1988) A simple technique for quantitation of low levels of DNA damage in individual cells. Exp Cell Res 175:184–191
Sonawane A, Santos JC, Mishra BB, Jena P, Progida C, Sorensen OE, Gallo R, Appelberg R, Griffiths G (2011) Cathelicidin is involved in the intracellular killing of mycobacteria in macrophages. Cell Microbiol 13:1601–1617
Weiss G, Schaible UE (2015) Macrophage defense mechanisms against intracellular bacteria. Immunol Rev 264:182–203
World Health Organization (2017) Global tuberculosis report 2017. In: Global tuberculosis report
Yang Y, Zhao S, Song J (2004) Caspase-dependent apoptosis and-independent poly (ADP-ribose) polymerase cleavage induced by transforming growth factor β1. Int J Biochem Cell Biol 36:223–234
Acknowledgements
Research in Dr. S. Bhattacharjee (Tripura University) and Dr. B. N. Banerjee’s (KIIT University) labs are supported by Govt. of India extramural research funds from DBT—Twinning (DBT sanction no.—BT/519/NE/TBP/2013) and DST-SERB NPDF, Govt. of India (Sanction Order No.—PDF/2016/004121).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Erko Stackebrandt.
Rights and permissions
About this article
Cite this article
Ghosh, C., Sarkar, A., Anuja, K. et al. Free radical stress induces DNA damage response in RAW264.7 macrophages during Mycobacterium smegmatis infection. Arch Microbiol 201, 487–498 (2019). https://doi.org/10.1007/s00203-018-1587-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-018-1587-y