Abstract
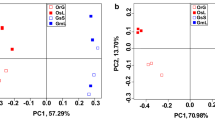
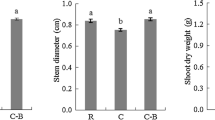
The fungicides used to control diseases in cereal production can have adverse effects on non-target microbial communities, with possible consequences for plant health and productivity. Although we know that fungicides affect microbial community structure and soil activities, it is unclear how crop cultivars have altered the impact of fungicides on rhizomicrobiomes. In this study, the rhizosphere bacterial and fungal communities and structures of cultivated crops and their wild relatives were studied by Illumina MiSeq sequencing analysis. The results indicated that the rhizomicrobiome communities of wild crops reacted more strongly to fungicides than that of their cultivated relatives. Furthermore, fungal community composition was more affected by fungicides than bacterial community composition. Remarkably, the same trend was observed in both soybean and rice with regard to the influence of crop cultivar on the response of the rhizomicrobiome to fungicide application, although the level of the response was not similar. We report for the first time that the rhizomicrobiomes of wild crops reacted more strongly to fungicides than the rhizomicrobiomes of cultivated crops.




Similar content being viewed by others
References
A-Be D (2007) The ade4 package: implementing the duality diagram for ecologists. J Stat Softw 22:1–20
Altschul SF et al (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402. https://doi.org/10.1093/nar/25.17.3389
Amaral D, Pinto N, Souza V, Aragao F, Santos M (2017) Control of Fusarium oxysporum infection in transgenic tobacco carrying oxalate descarboxilase gene. J Appl Biol Biotechnol 5:079–083. https://doi.org/10.7324/jabb.2017.50114
Beauregard PB, Chai Y, Vlamakis H, Losick R, Kolter R (2013) Bacillus subtilis biofilm induction by plant polysaccharides. Proc Natl Acad Sci USA 110:E1621–E1630. https://doi.org/10.1073/pnas.1218984110
Benjamini Yoav HY (1995) Controlling the false discovery rate: a practical and powerful approach to multiple testing. J R Stat Soc Ser B (Methodol) 57:289–300
Bischoff M, Lee LS, Turco RF (2005) Accelerated degradation of N, N′-dibutylurea (DBU) upon repeated application. Biodegradation 16:265–273. https://doi.org/10.1007/s10532-004-1361-5
Bouffaud M-L, Poirier M-A, Muller D, Moënne-Loccoz Y (2014) Root microbiome relates to plant host evolution in maize and other Poaceae. Environ Microbiol 16:2804–2814. https://doi.org/10.1111/1462-2920.12442
Bulgarelli D et al (2015) Structure and function of the bacterial root microbiota in wild and domesticated barley. Cell Host Microbe 17:392–403. https://doi.org/10.1016/j.chom.2015.01.011
Caporaso JG et al (2010) QIIME allows analysis of high-throughput community sequencing data. Nat Methods 7:335–336. https://doi.org/10.1038/nmeth.f.303
Cipriano MA et al (2016) Lettuce and rhizosphere microbiome responses to growth promoting Pseudomonas species under field conditions. FEMS Microbiol Ecol. https://doi.org/10.1093/femsec/fiw197
Cretoiu MS, Korthals GW, Visser JH, van Elsas JD (2013) Chitin amendment increases soil suppressiveness toward plant pathogens and modulates the actinobacterial and oxalobacteraceal communities in an experimental agricultural field. Appl Environ Microbiol 79:5291–5301. https://doi.org/10.1128/aem.01361-13
Edgar RC (2013) UPARSE: highly accurate OTU sequences from microbial amplicon reads. Nat Methods 10:996–998. https://doi.org/10.1038/nmeth.2604
Edgar RC, Haas BJ, Clemente JC, Quince C, Knight R (2011) UCHIME improves sensitivity and speed of chimera detection. Bioinformatics 27:2194–2200. https://doi.org/10.1093/bioinformatics/btr381
Fox JE, Gulledge J, Engelhaupt E, Burow ME, McLachlan JA (2007) Pesticides reduce symbiotic efficiency of nitrogen-fixing rhizobia and host plants. Proc Natl Acad Sci USA 104:10282–10287. https://doi.org/10.1073/pnas.0611710104
Garcia-Cazorla J, Xirau-Vayreda M (1998) Monitoring degradation of dicarboximidic fungicide residues in soils. J Agric Food Chem 46:2845–2850. https://doi.org/10.1021/jf971025t
Gonzalez Montoro AM, Cao R, Espinosa N, Cudeiro J, Marino J (2014) Functional two-way analysis of variance and bootstrap methods for neural synchrony analysis. BMC Neurosci 15:96. https://doi.org/10.1186/1471-2202-15-96
Gu L et al (2010) Assessing the impact of fungicide enostroburin application on bacterial community in wheat phyllosphere. J Environ Sci China 22:134–141. https://doi.org/10.1016/S1001-0742(09)60084-X
Haack FS et al (2016) Molecular keys to the Janthinobacterium and Duganella spp. interaction with the plant pathogen Fusarium graminearum. Front Microbiol 7:1668. https://doi.org/10.3389/fmicb.2016.01668
Halpin AL et al (2016) Intestinal microbiome disruption in patients in a long-term acute care hospital: a case for development of microbiome disruption indices to improve infection prevention. Am J Infect Control 44:830–836. https://doi.org/10.1016/j.ajic.2016.01.003
Husson F, Josse J, Le S (2008) FactoMineR: an R package for multivariate analysis. J Stat Softw 25:1–18
Karlsson I, Friberg H, Steinberg C, Persson P (2014) Fungicide effects on fungal community composition in the wheat phyllosphere. PLoS One 9:e111786. https://doi.org/10.1371/journal.pone.0111786
Koljalg U et al (2013) Towards a unified paradigm for sequence-based identification of fungi. Mol Ecol 22:5271–5277. https://doi.org/10.1111/mec.12481
Lambers H, Mougel C, Jaillard B, Hinsinger P (2009) Plant-microbe-soil interactions in the rhizosphere: an evolutionary perspective. Plant Soil 321:83–115.https://doi.org/10.1007/s11104-009-0042-x
Magoc T, Salzberg SL (2011) FLASH: fast length adjustment of short reads to improve genome assemblies. Bioinformatics 27:2957–2963. https://doi.org/10.1093/bioinformatics/btr507
Matson PA, Parton WJ, Power AG, Swift MJ (1997) Agricultural intensification and ecosystem properties. Science 277:504–509. https://doi.org/10.1126/science.277.5325.504
Mendes LW, Kuramae EE, Navarrete AA, van Veen JA, Tsai SM (2014) Taxonomical and functional microbial community selection in soybean rhizosphere. ISME J 8:1577–1587. https://doi.org/10.1038/ismej.2014.17
Miller AW, Dearing D (2013) The metabolic and ecological interactions of oxalate-degrading bacteria in the mammalian gut. Pathogens 2:636–652. https://doi.org/10.3390/pathogens2040636
Moulas C, Petsoulas C, Rousidou K, Perruchon C, Karas P, Karpouzas DG (2013) Effects of systemic pesticides imidacloprid and metalaxyl on the phyllosphere of pepper plants. Biomed Res Int 2013:969750. https://doi.org/10.1155/2013/969750
Newton AC, Gravouil C, Fountaine JM (2010) Managing the ecology of foliar pathogens: ecological tolerance in crops. Annal Appl Biol 157:343–359. https://doi.org/10.1111/j.1744-7348.2010.00437.x
Ofek M, Voronov-Goldman M, Hadar Y, Minz D (2014) Host signature effect on plant root-associated microbiomes revealed through analyses of resident vs. active communities. Environ Microb 16:2157–2167. https://doi.org/10.1111/1462-2920.12228
Oksanen JRK, Legendre P, O’Hara RB, Stevens MH (2013) Community ecology package. R package version 2.5-2. http://CRAN.R-project.org/package=vegan
Padovani L, Capri E, Padovani C, Puglisi E, Trevisan M (2006) Monitoring tricyclazole residues in rice paddy watersheds. Chemosphere 62:303–314 https://doi.org/10.1016/j.chemosphere.2005.05.025
Parks DH, Tyson GW, Hugenholtz P, Beiko RG (2014) STAMP: statistical analysis of taxonomic and functional profiles. Bioinformatics 30:3123–3124. https://doi.org/10.1093/bioinformatics/btu494
Perez-Jaramillo JE, Mendes R, Raaijmakers JM (2016) Impact of plant domestication on rhizosphere microbiome assembly and functions. Plant Mol Biol 90:635–644. https://doi.org/10.1007/s11103-015-0337-7
Pinto C, Pinho D, Sousa S, Pinheiro M, Egas C, Gomes AC (2014) Unravelling the diversity of grapevine microbiome. PLoS One 9:e85622. https://doi.org/10.1371/journal.pone.0085622
Prober SM et al (2015) Plant diversity predicts beta but not alpha diversity of soil microbes across grasslands worldwide. Ecol Lett 18:85–95. https://doi.org/10.1111/ele.12381
Pruesse E et al (2007) SILVA: a comprehensive online resource for quality checked and aligned ribosomal RNA sequence data compatible with ARB. Nucleic Acids Res 35:7188–7196. https://doi.org/10.1093/nar/gkm864
Satya P et al (2016) Insight into genetic relation and diversity of cultivated and semi-domesticated under-utilized Crotalaria species gained using start codon targeted (SCoT) markers. Biochem Syst Ecol 66:24–32. https://doi.org/10.1016/j.bse.2016.02.032
Schlemper TR, van Veen JA, Kuramae EE (2017) Co-variation of bacterial and fungal communities in different sorghum cultivars and growth stages is soil dependent. Microb Ecol. https://doi.org/10.1007/s00248-017-1108-6
Schloss PD et al (2009) Introducing mothur: open-source, platform-independent, community-supported software for describing and comparing microbial communities. Appl Environ Microbiol 75:7537–7541. https://doi.org/10.1128/aem.01541-09
Soni R, Kumar V, Suyal DC, Jain L, Goel R (2017) Metagenomics of plant rhizosphere microbiome. In: Singh RP, Kothari R, Koringa PG, Singh SP (eds) Understanding host-microbiome interactions—an omics approach: omics of host-microbiome association. Springer, Singapore, pp 193–205
Thijs S et al (2017) Comparative evaluation of four bacteria-specific primer pairs for 16S rRNA gene surveys. Front Microbiol https://doi.org/10.3389/fmicb.2017.00494
van der Heijden MG, Bardgett RD, van Straalen NM (2008) The unseen majority: soil microbes as drivers of plant diversity and productivity in terrestrial ecosystems. Ecol Lett 11:296–310. https://doi.org/10.1111/j.1461-0248.2007.01139.x
Verma SK, Kingsley K, Irizarry I, Bergen M, Kharwar RN, White JF Jr (2017) Seed-vectored endophytic bacteria modulate development of rice seedlings. J Appl Microbiol 122:1680–1691. https://doi.org/10.1111/jam.13463
Weiss S et al (2017) Normalization and microbial differential abundance strategies depend upon data characteristics. Microbiome 5:27. https://doi.org/10.1186/s40168-017-0237-y
Yu Y, Chu X, Pang G, Xiang Y, Fang H (2009) Effects of repeated applications of fungicide carbendazim on its persistence and microbial community in soil. J Environ Sci (China) 21:179–185. https://doi.org/10.1016/S1001-0742(08)62248-2
Zhang R, Vivanco JM, Shen Q (2017a) The unseen rhizosphere root-soil-microbe interactions for crop production. Curr Opin Microbiol 37:8–14. https://doi.org/10.1016/j.mib.2017.03.008
Zhang Y, Xu J, Riera N, Jin T, Li J, Wang N (2017b) Huanglongbing impairs the rhizosphere-to-rhizoplane enrichment process of the citrus root-associated microbiome. Microbiome 5:97. https://doi.org/10.1186/s40168-017-0304-4
Zhou Y, Yang P, Cui F, Zhang F, Luo X, Xie J (2016) Transcriptome analysis of salt stress responsiveness in the seedlings of Dongxiang wild rice (Oryza rufipogon Griff.). PLoS One 11:e0146242. https://doi.org/10.1371/journal.pone.0146242
Zhou X, Tian L, Zhang J, Ma L, Li X, Tian C (2017) Rhizospheric fungi and their link with the nitrogen-fixing Frankia harbored in host plant Hippophae rhamnoides L. J Basic Microbiol 57:1055–1064. https://doi.org/10.1002/jobm.201700312
Acknowledgements
This research was financially supported by the Science Foundation of Chinese Academy of Sciences (XDB15030103), the National Key Research and Development Program of China (2016YFC0501202), the Key Deployment Project of the Chinese Academy of Sciences(KFZD-SW-112),the National Natural Science Foundation of China (41571255), Key Laboratory Foundation of Mollisols Agroecology (2016ZKHT-05), the Key Technology Research and Development Program of CAS during the “13th Five-Year Plan” (Y6H2043001), and the Excellent Researcher Award Program from Jilin province of China (20180520052JH). We thank Dr. Jun Rong for the supply of the seeds of wild rice and thank Dr. Xinhou Zhang for the supply of the seeds of wild soybean. We thank Johannes A. van Veen for improving manuscript and thank Eiko E Kuramae for improving figures.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All the authors declare no conflict of interest.
Additional information
Communicated by Yusuf Akhter.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Shi, S., Tian, L., Xu, S. et al. The rhizomicrobiomes of wild and cultivated crops react differently to fungicides. Arch Microbiol 201, 477–486 (2019). https://doi.org/10.1007/s00203-018-1586-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-018-1586-z