Abstract
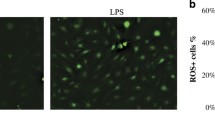
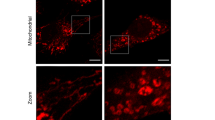
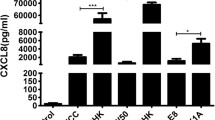
Periodontal diseases are infectious polymicrobial inflammatory diseases that lead to destruction of the periodontal ligament, gingiva, and alveolar bone. Sequential colonization of a broad range of bacteria, including Fusobacterium nucleatum and Porphyromonas gingivalis, is an important phenomenon in this disease model. F. nucleatum is a facultative anaerobic species thought to be a key mediator of dental plaque maturation due to its extensive coaggregation with other oral bacteria, while P. gingivalis is an obligate anaerobic species that induces gingival inflammation by secreting various virulence factors. The formation of a bacterial complex by these two species is central to the pathogenesis of periodontal disease. Reactive oxygen species (ROS) are produced during bacterial infections and are involved in intracellular signaling. However, the impact of oral bacteria-induced ROS on the ecology of F. nucleatum and P. gingivalis has yet to be clarified. In the present study, we investigated ROS production induced in primary human oral cells by F. nucleatum and P. gingivalis and its effect on the formation of their bacterial complexes and further host cell apoptosis. We found that in primary human gingival fibroblasts (GFs), two NADPH oxidase isoforms, NOX1 and NOX2, were activated in response to F. nucleatum infection but not P. gingivalis infection. Accordingly, increased NADPH oxidase activity and production of superoxide anion were observed in GFs after F. nucleatum infection, but not after P. gingivalis infection. Interestingly, in NOX1, NOX2, or NOX1/NOX2 knockdown cells, the number of P. gingivalis decreased when the cells were coinfected with F. nucleatum. A similar pattern of host cell apoptosis was observed. This implies that F. nucleatum contributes to attachment of P. gingivalis by triggering activation of NADPH oxidase in host cells, which may provide an environment more favorable to strict anaerobic bacteria and have a subsequent effect on apoptosis of host cells.





Similar content being viewed by others
References
Abid MR, Spokes KC, Shih SC, Aird WC (2007) NADPH oxidase activity selectively modulates vascular endothelial growth factor signaling pathways. J Biol Chem 282:35373–35385. doi:10.1074/jbc.M702175200
Ashida H, Mimuro H, Ogawa M, Kobayashi T, Sanada T, Kim M, Sasakawa C (2011) Cell death and infection: a double-edged sword for host and pathogen survival. J Cell Biol 195:931–942. doi:10.1083/jcb.201108081
Babior BM (1999) NADPH oxidase: an update. Blood 93:1464–1476
Banskota S, Regmi SC, Kim JA (2015) NOX1 to NOX2 switch deactivates AMPK and induces invasive phenotype in colon cancer cells through overexpression of MMP-7. Mol Cancer 14:123. doi:10.1186/s12943-015-0379-0
Bedard K, Lardy B, Krause KH (2007) NOX family NADPH oxidases: not just in mammals. Biochimie 89:1107–1112. doi:10.1016/j.biochi.2007.01.012
Chéret C, Gervais A, Lelli A, Colin C, Amar L, Ravassard P, Mallet J, Cumano A, Krause KH, Mallat M (2008) Neurotoxic activation of microglia is promoted by a nox1-dependent NADPH oxidase. J Neurosci 28:12039–12051. doi:10.1523/JNEUROSCI.3568-08.2008
Chose O, Sansilvestri-Morel P, Badier-Commander C, Bernhardt F, Fabiani JN, Rupin A, Verbeuren TJ (2008) Distinct role of nox1, nox2, and p47phox in unstimulated versus angiotensin II-induced NADPH oxidase activity in human venous smooth muscle cells. J Cardiovasc Pharmacol 51:131–139. doi:10.1097/FJC.0b013e31815d781d
Desta T, Graves DT (2007) Fibroblast apoptosis induced by Porphyromonas gingivalis is stimulated by a gingipain and caspase-independent pathway that involves apoptosis-inducing factor. Cell Microbiol 9:2667–2675. doi:10.1111/j.1462-5822.2007.00987.x
Diaz PI, Zilm PS, Rogers AH (2002) Fusobacterium nucleatum supports the growth of Porphyromonas gingivalis in oxygenated and carbon-dioxide-depleted environments. Microbiology 148:467–472
Edwards AM, Grossman TJ, Rudney JD (2006) Fusobacterium nucleatum transports noninvasive Streptococcus cristatus into human epithelial cells. Infect Immun 74:654–662. doi:10.1128/IAI.74.1.654-662.2006
Fu P, Mohan V, Mansoor S, Tiruppathi C, Sadikot RT, Natarajan V (2013) Role of nicotinamide adenine dinucleotide phosphate-reduced oxidase proteins in Pseudomonas aeruginosa-induced lung inflammation and permeability. Am J Respir Cell Mol Biol 48:477–488. doi:10.1165/rcmb.2012-0242OC
Golz L et al (2014) LPS from P. gingivalis and hypoxia increases oxidative stress in periodontal ligament fibroblasts and contributes to periodontitis. Mediat Inflamm 2014:1–13. doi:10.1155/2014/986264
Guo L, He X, Shi W (2014) Intercellular communications in multispecies oral microbial communities. Front Microbiol 5:328. doi:10.3389/fmicb.2014.00328
Hajishengallis G (2015) Periodontitis: from microbial immune subversion to systemic inflammation. Nat Rev Immunol 15:30–44. doi:10.1038/nri3785
Han YW, Shi W, Huang GT, Kinder Haake S, Park NH, Kuramitsu H, RJ Genco (2000) Interactions between periodontal bacteria and human oral epithelial cells: Fusobacterium nucleatum adheres to and invades epithelial cells. Infect Immun 68:3140–3146
Hordijk PL (2006) Regulation of NADPH oxidases: the role of Rac proteins. Circ Res 98:453–462. doi:10.1161/01.RES.0000204727.46710.5e
Jewett A, Hume WR, Le H, Huynh TN, Han YW, Cheng G, Shi W (2000) Induction of apoptotic cell death in peripheral blood mononuclear and polymorphonuclear cells by an oral bacterium, Fusobacterium nucleatum. Infect Immun 68:1893–1898
Jiang F, Zhang Y, Dusting GJ (2011) NADPH oxidase-mediated redox signaling: roles in cellular stress response, stress tolerance, and tissue repair. Pharmacol Rev 63:218–242. doi:10.1124/pr.110.002980
Kinane DF, Galicia JC, Gorr SU, Stathopoulou PG, Benakanakere M (2008) P. gingivalis interactions with epithelial cells. Front Biosci 13:966–984
Lambeth JD (2004) NOX enzymes and the biology of reactive oxygen. Nat Rev Immunol 4:181–189. doi:10.1038/nri1312
Loesche WJ, Grossman NS (2001) Periodontal disease as a specific, albeit chronic, infection: diagnosis and treatment. Clin Microbiol Rev 14:727–752. doi:10.1128/CMR.14.4.727-752.2001
Mantri CK, Chen C, Dong X, Goodwin JS, Xie H (2014) Porphyromonas gingivalis-mediated epithelial cell entry of HIV-1. J Dent Res 93:794–800. doi:10.1177/0022034514537647
Marcato LG et al (2008) The role of Toll-like receptors 2 and 4 on reactive oxygen species and nitric oxide production by macrophage cells stimulated with root canal pathogens. Oral Microbiol Immunol 23:353–359. doi:10.1111/j.1399-302X.2008.00432.x
Metzger Z, Blasbalg J, Dotan M, Weiss EI (2009a) Enhanced attachment of Porphyromonas gingivalis to human fibroblasts mediated by Fusobacterium nucleatum. J Endod 35:82–85. doi:10.1016/j.joen.2008.10.011
Metzger Z, Lin YY, Dimeo F, Ambrose WW, Trope M, Arnold RR (2009b) Synergistic pathogenicity of Porphyromonas gingivalis and Fusobacterium nucleatum in the mouse subcutaneous chamber model. J Endod 35:86–94. doi:10.1016/j.joen.2008.10.015
Min BM, Lee G, Kim SH, Nam YS, Lee TS, Park WH (2004) Electrospinning of silk fibroin nanofibers and its effect on the adhesion and spreading of normal human keratinocytes and fibroblasts in vitro. Biomaterials 25:1289–1297
Nakanishi A, Hie M, Iitsuka N, Tsukamoto I (2013) A crucial role for reactive oxygen species in macrophage colony-stimulating factor-induced RANK expression in osteoclastic differentiation. Int J Mol Med 31:874–880. doi:10.3892/ijmm.2013.1258
Opitz N, Drummond GR, Selemidis S, Meurer S, Schmidt HH (2007) The ‘A’s and ‘O’s of NADPH oxidase regulation: a commentary on “Subcellular localization and function of alternatively spliced Noxo1 isoforms”. Free Radic Biol Med 42:175–179. doi:10.1016/j.freeradbiomed.2006.11.003
Rampersad SN (2012) Multiple applications of Alamar Blue as an indicator of metabolic function and cellular health in cell viability bioassays. Sensors 12:12347–12360. doi:10.3390/s120912347
Reyes L, Herrera D, Kozarov E, Roldan S, Progulske-Fox A (2013) Periodontal bacterial invasion and infection: contribution to atherosclerotic pathology. J Clin Periodontol 40(Suppl 14):S30–S50. doi:10.1111/jcpe.12079
Sadok A, Bourgarel-Rey V, Gattacceca F, Penel C, Lehmann M, Kovacic H (2008) Nox1-dependent superoxide production controls colon adenocarcinoma cell migration. Biochim Biophys Acta 1783:23–33. doi:10.1016/j.bbamcr.2007.10.010
Saito A, Inagaki S, Kimizuka R, Okuda K, Hosaka Y, Nakagawa T, Ishihara K (2008) Fusobacterium nucleatum enhances invasion of human gingival epithelial and aortic endothelial cells by Porphyromonas gingivalis. FEMS Immunol Med Microbiol 54:349–355. doi:10.1111/j.1574-695X.2008.00481.x
Saito A, Inagaki S, Ishihara K (2009) Differential ability of periodontopathic bacteria to modulate invasion of human gingival epithelial cells by Porphyromonas gingivalis. Microb Pathog 47:329–333. doi:10.1016/j.micpath.2009.09.012
Saito A, Kokubu E, Inagaki S, Imamura K, Kita D, Lamont RJ, Ishihara K (2012) Porphyromonas gingivalis entry into gingival epithelial cells modulated by Fusobacterium nucleatum is dependent on lipid rafts. Microb Pathog 53:234–242. doi:10.1016/j.micpath.2012.08.005
Sheets SM, Potempa J, Travis J, Casiano CA, Fletcher HM (2005) Gingipains from Porphyromonas gingivalis W83 induce cell adhesion molecule cleavage and apoptosis in endothelial cells. Infect Immun 73:1543–1552. doi:10.1128/IAI.73.3.1543-1552.2005
Signat B, Roques C, Poulet P, Duffaut D (2011) Fusobacterium nucleatum in periodontal health and disease. Curr Issues Mol Biol 13:25–36
Stathopoulou PG, Galicia JC, Benakanakere MR, Garcia CA, Potempa J, Kinane DF (2009) Porphyromonas gingivalis induce apoptosis in human gingival epithelial cells through a gingipain-dependent mechanism. BMC Microbiol 9:107. doi:10.1186/1471-2180-9-107
Takeya R, Sumimoto H (2006) Regulation of novel superoxide-producing NAD(P)H oxidases. Antioxid Redox Signal 8:1523–1532. doi:10.1089/ars.2006.8.1523
Topcuoglu N, Paltura C, Kulekci M, Ustek D, Kulekci G (2013) Real-time polymerase chain reaction versus conventional Pcr: a comparison between two methods for the detection of Fusobacterium Nucleatum in Saliva, nasopharyngeal secretion and middle ear effusion samples. Biotechnol Biotechnol Equip 27:3825–3828. doi:10.5504/Bbeq.2013.0022
Torres MA, Jones JD, Dangl JL (2006) Reactive oxygen species signaling in response to pathogens. Plant Physiol 141:373–378. doi:10.1104/pp.106.079467
Wang H et al (2014) Porphyromonas gingivalis-induced reactive oxygen species activate JAK2 and regulate production of inflammatory cytokines through c-Jun. Infect Immun 82:4118–4126. doi:10.1128/IAI.02000-14
Yilmaz O, Verbeke P, Lamont RJ, Ojcius DM (2006) Intercellular spreading of Porphyromonas gingivalis infection in primary gingival epithelial cells. Infect Immun 74:703–710. doi:10.1128/IAI.74.1.703-710.2006
Acknowledgments
This work was supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry Science, ICT and Future Planning (2014R1A2A2A01005448), and by a National Research Foundation of Korea (NRF) grant funded by the Korean Government (MSIP) (2011-0030121). Sun Hee Ahn was also supported by the Basic Science Research Program through the National Research Foundation of Korea (NRF) funded by the Ministry of Education (2013R1A1A2059721).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interests to declare.
Additional information
Communicated by Erko Stackebrandt.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ahn, S.H., Song, JE., Kim, S. et al. NOX1/2 activation in human gingival fibroblasts by Fusobacterium nucleatum facilitates attachment of Porphyromonas gingivalis . Arch Microbiol 198, 573–583 (2016). https://doi.org/10.1007/s00203-016-1223-7
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-016-1223-7