Abstract
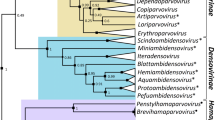
Bacteriophages rapidly diversify their genes through co-evolution with their hosts. We hypothesize that gene diversification of phages leads to locality in phages genome. To test this hypothesis, we investigated the genetic diversity and composition of Microcystis cyanophages using 104 sequences of Ma-LMM01-type cyanophages from two geographically distant sampling sites. The intergenetic region between the ribonucleotide reductase genes nrdA and nrdB was used as the genetic marker. This region contains the host-derived auxiliary metabolic genes nblA, an unknown function gene g04, and RNA ligase gene g03. The sequences obtained were conserved in the Ma-LMM01 gene order and contents. Although the genetic diversity of the sequences was high, it varied by gene. The genetic diversity of nblA was the lowest, suggesting that nblA is a highly significant gene that does not allow mutation. In contrast, g03 sequences had many point mutations. RNA ligase is involved in the counter-host’s phage defense mechanism, suggesting that phage defense also plays an important role for rapid gene diversification. The maximum parsimony network and phylogenic analysis showed the sequences from the two sampling sites were distinct. These findings suggest Ma-LMM01-type phages rapidly diversify their genomes through co-evolution with hosts in each location and eventually provided locality of their genomes.





Similar content being viewed by others
References
Bellec L, Grimsley N, Derelle E, Moreau H, Desdevises Y (2010) Abundance, spatial distribution and genetic diversity of Ostreococcus tauri viruses in two different environments. Environ Microbiol Rep 2:313–321
Brockhurst MA, Morgan AD, Fenton A, Buckling A (2007) Experimental coevolution with bacteria and phage. The Pseudomonas fluorescens: phi 2 model system. Infec Genet Evol 7:547–552
Carstens EB (2010) Ratification vote on taxonomic proposals to the International Committee on Taxonomy of Viruses (2009). Arch Virol 155:133–146
Clement M, Posada D, Crandall KA (2000) TCS: a computer program to estimate gene genealogies. Mol Ecol 9:1657–1659
Codd G, Lindsay J, Young F, Morrison L, Metcalf J (2005) Harmful cyanobacteria. In: Huisman J, Matthijis HCP, Visser PM (eds) Cyanobacterial toxins. Springer, Dordrecht, pp 1–23
Colwell RK (2004) Estimates: statistical estimation of species richness and shared species from samples. Version 8. http://viceroy.eeb.uconn.edu/estimates
Dwivedi B, Xue B, Lundin D, Edwards RA, Breitbart M (2013) A bioinformatic analysis of ribonucleotide reductase genes in phage genomes and metagenomes. BMC Evol Biol 13:33
El Omari K, Ren J, Bird LE, Bona MK, Klarmann G, LeGrice SFJ, Stammers DK (2006) Molecular architecture and ligand recognition determinants for T4 RNA ligase. J Biol Chem 281:1573–1579
Gaevsky NA, Kolmakov VI, Belykh OI, Tikhonova IV, Joung Y, Ahn TS, Nabatova VA, Gladkikh AS (2011) Ecological development and genetic diversity of Microcystis aeruginosa from artificial reservoir in Russia. J Microbiol 49:714–720
Gao EB, Gui J-F, Zhang Q-Y (2012) A novel cyanophage with a cyanobacterial nonbleaching protein a gene in the genome. J Virol 86:236–245
Hargreaves KR, Anderson NJ, Clokie MR (2013) Recovery of viable cyanophages from the sediments of a eutrophic lake at decadal timescales. FEMS Microbiol Ecol 83:450–456
Ho CK, Wang LK, Lima CD, Shuman S (2004) Structure and mechanism of RNA ligase. Structure 12:327–339
Honjo M, Matsui K, Ueki M, Nakamura R, Fuhrman JA, Kawabata Z (2006) Diversity of virus-like agents killing Microcystis aeruginosa in a hyper-eutrophic pond. J Plankton Res 28:407–412
Ignacio-Espinoza JC, Sullivan MB (2012) Phylogenomics of T4 cyanophages: lateral gene transfer in the ‘core’ and origins of host genes. Environ Microbiol 14:2113–2126
Iranzo J, Lobkovsky AE, Wolf YI, Koonin EV (2013) Evolutionary dynamics of archaeal and bacterial adaptive immunity systems, CRISPR–Cas, in an explicit ecological context. J Bacteriol. doi:10.1128/JB.000412-13
Iritani N, Vennema H, Siebenga JJ, Siezen RJ, Renckens B, Seto Y, Kaida A, Koopmans M (2008) Genetic analysis of the capsid gene of genotype GII. 2 noroviruses. J Virol 82:7336–7345
Jochimsen EM, Carmichael WW, An JS et al (1998) Liver failure and death after exposure to microcystins at a hemodialysis center in Brazil. N Engl J Med 338:873–878
Kimura S, Sako Y, Yoshida T (2013) Rapid gene diversification of Microcystis cyanophages revealed by long-and short-term genetic analysis of the tail sheath gene in a natural pond. Appl Environ Microbiol 79:2789–2795
Koskella B, Thompson JN, Preston GM, Buckling A (2011) Local biotic environment shapes the spatial scale of bacteriophage adaptation to bacteria. Am Nat 177:440–451
Kuno S, Yoshida T, Kaneko T, Sako Y (2012) Intricate interactions between the bloom-forming cyanobacterium Microcystis aeruginosa and foreign genetic elements, revealed by diversified clustered regularly interspaced short palindromic repeat (CRISPR) signatures. Appl Environ Microbiol 78:5353–5360
Labrie SJ, Samson JE, Moineau S (2010) Bacteriophage resistance mechanisms. Nat Rev Microbiol 8:317–327
Lavigne R, Darius P, Summer EJ, Seto D, Mahadevan P, Nilsson AS, Ackermann HW, Kropinski AM (2009) Classification of Myoviridae bacteriophages using protein sequence similarity. BMC Microbiol 9:224
Lindell D, Sullivan MB, Johnson ZI, Tolonen AC, Rohwer F, Chisholm SW (2004) Transfer of photosynthesis genes to and from Prochlorococcus viruses. Proc Natl Acad Sci USA 101:11013–11018
Makarova KS, Wolf YI, Snir S, Koonin EV (2011) Defense Islands in bacterial and archaeal genomes and prediction of novel defense systems. J Bacteriol 193:6039–6056
Marston MF, Pierciey FJ, Shepard A, Gearin G, Qi J, Yandava C, Schuster SC, Henn MR, Martiny JBH (2012) Rapid diversification of coevolving marine Synechococcus and a virus. Proc Natl Acad Sci USA 109:4544–4549
Moisander PH, Lehman PW, Ochiai M, Corum S (2009) Diversity of Microcystis aeruginosa in the Klamath River and San Francisco Bay delta, California, USA. Aquat Microb Ecol 57:19–31
Ou T, Li S, Liao X, Zhang Q (2013) Cultivation and characterization of the MaMV-DC cyanophage that infects bloom-forming cyanobacterium Microcystis aeruginosa. Virol Sin 28:266–271
Raymond A, Shuman S (2007) Deinococcus radiodurans RNA ligase exemplifies a novel ligase clade with a distinctive N-terminal module that is important for 5′-PO4 nick sealing and ligase adenylylation but dispensable for phosphodiester formation at an adenylylated nick. Nucleic Acids Res 35:839–849
Rodriguez-Valera F, Martin-Cuadrado A-B, Beltran Rodriguez-Brito LP, Thingstad TM, Forest Rohwer AM (2009) Explaining microbial population genomics through phage predation. Nat Rev Microbiol 7:828–836
Sabart M, Pobel D, Latour D, Robin J, Salencon MJ, Humbert JF (2009) Spatiotemporal changes in the genetic diversity in French bloom-forming populations of the toxic cyanobacterium, Microcystis aeruginosa. Environ Microbiol Rep 1:263–272
Sanmukh SG, Rahman M, Paunikar WN (2012) Comparative genomic studies of hypothetical proteins in cyanophages. Evolution 20:33
Sullivan MB, Coleman ML, Weigele P, Rohwer F, Chisholm SW (2005) Three Prochlorococcus cyanophage genomes: signature features and ecological interpretations. PLoS Biol 3:790–806
Suttle CA (2005) Viruses in the sea. Nature 437:356–361
Suttle CA (2007) Marine viruses: major players in the global ecosystem. Nat Rev Microbiol 5:801–812
Takashima Y, Yoshida T, Yoshida M, Shirai Y, Tomaru Y, Takao Y, Hiroishi S, Nagasaki K (2007) Development and application of quantitative detection of cyanophages phylogenetically related to cyanophage Ma-LMM01 infecting Microcystis aeruginosa in fresh water. Microbes Environ 22:207–213
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S (2011) MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol 28:2731–2739
Tucker S, Pollard P (2005) Identification of cyanophage Ma-LBP and infection of the cyanobacterium Microcystis aeruginosa from an Australian subtropical lake by the virus. Appl Environ Microbiol 71:629–635
Vos M, Birkett PJ, Birch E, Griffiths RI, Buckling A (2009) Local adaptation of bacteriophages to their bacterial hosts in soil. Science 325:833–833
Yamamoto Y, Nakahara H (2009) Seasonal variations in the morphology of bloom-forming cyanobacteria in a eutrophic pond. Limnology 10:185–193
Yoshida T, Takashima Y, Tomaru Y, Shirai Y, Takao Y, Hiroishi S, Nagasaki K (2006) Isolation and characterization of a cyanophage infecting the toxic cyanobacterium Microcystis aeruginosa. Appl Environ Microbiol 72:1239–1247
Yoshida M, Yoshida T, Kashima A, Takashima Y, Hosoda N, Nagasaki K, Hiroishi S (2008a) Ecological dynamics of the toxic bloom-forming cyanobacterium Microcystis aeruginosa and its cyanophages in freshwater. Appl Environ Microbiol 74:3269–3273
Yoshida T, Nagasaki K, Takashima Y, Shirai Y, Tomaru Y, Takao Y, Sakamoto S, Hiroishi S, Ogata H (2008b) Ma-LMM01 infecting toxic Microcystis aeruginosa illuminates diverse cyanophage genome strategies. J Bacteriol 190:1762–1772
Yoshida-Takashima Y, Yoshida M, Ogata H, Nagasaki K, Hiroishi S, Yoshida T (2012) Cyanophage infection in the bloom-forming cyanobacteria Microcystis aeruginosa in surface freshwater. Microbes Environ 27:350–355
Acknowledgments
This study was partially supported by Grant-in-Aid for Scientific Research (B) (No. 20310045). We thank Takahiro Miyazako for sampling help.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Erko Stackebrandt.
Rights and permissions
About this article
Cite this article
Nakamura, G., Kimura, S., Sako, Y. et al. Genetic diversity of Microcystis cyanophages in two different freshwater environments. Arch Microbiol 196, 401–409 (2014). https://doi.org/10.1007/s00203-014-0980-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-014-0980-4