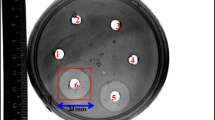
Abstract
The aim of this study was to isolate a novel bacterial strain with strong and broad spectrum antibacterial activity from a livestock feed prebiotic supplement. A novel strain, termed Paenibacillus polymyxa JB05-01-1, was isolated using traditional microbiological methods and identified on the basis of its phenotypic and biochemical properties as well as its 16S rRNA gene sequence. This strain was able to inhibit growth of gram-negative bacteria including Escherichia coli RR1, Pseudomonas fluorescens R73, Pantoea agglomerans BC1, Butyrivibrio fibrisolvens OR85, and Fibrobacter succinogenes. The above antagonism against the aforementioned bacteria was attributed to production of an antimicrobial substance(s) termed “JB05-01-1.” Its production was optimal during the stationary phase. JB05-01-1 has a molecular weight of 2.5 KDa, its mode of action is bactericidal, and the divalent cations, Ca2+ and Mn2+, reduced its lethality. The antibacterial activity was heat-stable and was effective at a pH range of 2–9. Enzymes like trypsin, α-chymotrypsin, and proteinase K have abolished the antibacterial activity of JB05-01-1 indicating a proteinaceous motif. This type of naturally occurring bacteria and inhibitory substance(s) could represent an additional value in livestock feed supplements. The natural presence of antibacterial activity indicates an opportunity to decrease the addition of antibiotics.





Similar content being viewed by others
References
Abad AR, Cease KR, Blanchette RA (1987) A rapid technique using epoxy resin Quetol 651 to prepare woody plant tissues for ultrastructural study. Can J Bot 66:677–682
Altschul SF, Gish W, Miller W, Myers EW, Lipman DJ (1990) Basic local alignment search tool. J Mol Biol 215:403–410
Ash C, Priest FG, Collins MD (1993) Molecular identification of rRNA group 3 bacilli (Ash, Farrow, Wallbanks and Collins) using a PCR probe test. Proposal for the creation of a new genus Paenibacillus. Antonie van Leeuwenhoek 64:253–260
Bhunia AK, Johnson MC, Ray B (1987) Direct detection of an antimicrobial peptide of Pediococcus acidilactici in sodium dodecyl sulfate-polyacrylamide gel electrophoresis. J Ind Microbiol 2:319–322
Budde B, Hornbæk T, Jacobsen T, Barkholt V, Koch AG (2003) Leuconostoc carnosum 4010 has the potential for use a protective culture for vacuum-packed meats: culture isolation, bacteriocin identification, and meat application experiments. Int J Food Microbiol 83:171–184
Caldwell DR, Bryant MP (1966) Medium without rumen fluid for non selective enumeration and isolation of rumen bacteria. Appl Microbiol 14:794–801
Choi SK, Park SY, Kim R, Kim SB, Lee CH, Kim JF, Park SH (2009) Identification of Polymyxin Synthetase Gene Cluster of Paenibacillus polymyxa and Heterologous Expression of the Gene in Bacillus subtilis. J Bacteriol 191:3350–3358
Daba H, Lacroix C, Huang J, Simard RE, Lemieux L (1994) Simple method of purification and sequencing of a bacteriocin produced by Pediococcus acidilactici UL5. J Appl Bacteriol 77:682–688
de Man JC, Rogosa M, Sharpe ME (1960) A medium for the cultivation of lactobacilli. J Appl Bacteriol 23:130–135
Groisman EA, Kayser J, Soncini FC (1997) Regulation of polymyxin resistance and adaptation to low-Mg2+ environments. J Bacteriol 179:7040–7045
Hamer DH (2000) Treatment of nosocomial pneumonia and tracheobronchitis caused by multidrug-resistant Pseudomonas aeruginosa with aerosolized colistin. Am J Respir Crit Care Med 162:328–330
Kane MD, Poulsen LK, Stahl DA (1993) Monitoring the enrichment and isolation of sulfate-reducing bacteria by using oligonucleotide hybridization probes designed from environmentally derived 16S rRNA sequences. Appl Environ Microbiol 59:682–686
Kleinkauf H, von Dorhren H (1990) Nonribosomal biosynthesis of peptide antibiotics. Eur J Biochem 192:1–15
Labbé N, Juteau P, Parent S, Villemur R (2003) Bacterial diversity in a marine methanol-fed denitrification reactor at the Montreal Biodome, Canada. Microb Ecol 46:12–21
Liu WT, Marsh TL, Cheng H, Forney LJ (1997) Characterization of microbial diversity by determining terminal restriction fragment length polymorphisms of genes encoding 16S rRNA. Appl Environ Microbiol 63:4516–4522
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin-Phenol reagents. J Biol Chem 193:265–275
Martin NI, Hu H, Moake MM, Churey JJ, Whittal R, Worobo RW, Vederas JC (2003) Isolation, structural characterization, and properties of mattacin (polymyxin M), a cyclic peptide antibiotic produced by Paenibacillus kobensis M. J Biol Chem 278:13124–13132
Menichetti F (2005) Current and emerging serious gram-positive infections. Clin Microbiol Infect 11:22–28
Messens W, De Vuyst L (2002) Inhibitory substances produced by Lactobacilli isolated from sourdoughs. Int J Food Microbiol 72:31–43
Motta AS, Lorenzini DM, Brandelli A (2007) Purification and partial characterization of an antimicrobial peptide produced by a novel Bacillus sp. Isolated from the amazon basin. Curr Microbiol 54:282–286
Naghmouchi K, Drider D, Kheadr E, Lacroix C, Prévost H, Fliss I (2006) Multiple characterizations of Listeria monocytogenes sensitive and insensitive variants to divergicin M35, a new pediocin-like bacteriocin. J Appl Microbiol 100:29–39
Naghmouchi K, Drider D, Hammami R, Fliss I (2008) Effect of Antimicrobial Peptides Divergicin M35 and Nisin A on Listeria monocytogenes LSD530 Potassium Channels. Current Microbiol 56:609–612
Ongena M, Henry G, Thonart P (2010) The roles of cyclic lipopeptides in the biocontrol activity of Bacillus. Plant Pathol 1:59–69
Orwa JA, Govaerts K, Gevers E, Roets A, Schepdael V, Hoogmartens J (2002) Study of the stability of polymyxins B(1), E(1) and E(2) in aqueous solution using liquid chromatography and mass spectrometry. J Pharma Biomed Anal 29:203–212
Pirri G, Giuliani A, Nicoletto SF, Pizzuto L, Rinaldi AC (2009) Lipopeptides as anti-infectives: a practical perspective. Cent Eur J Biol 4:258–273
Selim S, Negrel J, Govaerts C, Gianinazzi S, van Tuinen D (2005) Isolation and partial characterization of antagonistic peptides produced by Paenibacillus sp. Strain B2 Isolated from the Sorghum Mycorrhizosphere. Appl Environ Microbiol 71:501–6507
Tagg JR, Dajani AS, Wannamaker LW (1976) Bacteriocins of Gram-positive bacteria. Bacteriol Rev 40:722–756
Tamra MA, Forrest GN, Messmer KJ (2007) Polymyxin antibiotics for gram-negative infections. Am J Health Syst Pharm 64:819–826
White DG, Zhao S, Singh R, McDermott PF (2004) Antimicrobial resistance among gram-negative foodborne bacterial pathogens associated with foods of animal origin. Foodborne Patholog Dis 1:137–152
Wolf CE, Gibbons WR (1996) Improved method for quantification of the bacteriocin nisin. J Appl Bacteriol 80:453–457
Zavascki AP, Goldani LZ, Li J, Nation RL (2007) Polymyxin B for the treatment of multidrug-resistant pathogens: a critical review. J Antimicrob Chemother 60:1206–1215
Zengguo He, Duygu K, Liwen Z, Chunhua Y, Kari GC, Ahmed EY (2007) Isolation and identification of a Paenibacillus polymyxa strain that coproduces a novel lantibiotic and polymyxin. App Environ Microbiol 73:168–178
Acknowledgments
This research was supported by a grant from Basic Environmental Systems & Technology Inc. Edmonton, AB, Canada, with matching funds provided by Agriculture and Agri-food Canada. Karim Naghmouchi was a recipient of a Post-doctoral fellowship from NSERC. The authors would like to thank Gert Moll for critical reading of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Erko Stackebrandt.
Rights and permissions
About this article
Cite this article
Naghmouchi, K., Paterson, L., Forster, B. et al. Paenibacillus polymyxa JB05-01-1 and its perspectives for food conservation and medical applications. Arch Microbiol 193, 169–177 (2011). https://doi.org/10.1007/s00203-010-0654-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-010-0654-9