Abstract
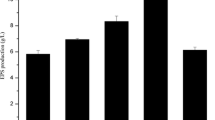
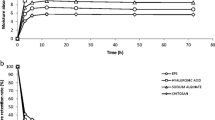
A heterofermentative Lactobacillus sp. CFR-2182 was isolated from dahi samples and it was found to produce 8.0 and 20.5 g/L heteropolysaccharide (HePS) in EPS medium (a simplified synthetic medium) and modified MRS broth, respectively, after 72 h at 30°C. The total carbohydrate, reducing sugar and moisture contents of the purified HePS were 74, 10.6 and 2 g, respectively, per 100 g on dry weight basis. The HePS produced in EPS medium had glucose and mannose in 17:1 ratio. The HePS was non-gelling and non-film forming type. It was completely soluble in water and 1 N sodium hydroxide solution. Gel permeation chromatography and HPLC analysis indicated considerable heterogeneity of the HePS, having three fractions with molecular weights ranging from 3.3 × 104 to 1.32 × 106 Da. The enzymatic hydrolysis of the HePS with pullulanase and α-amylase [with α(1→4) linkage] indicated the presence of α(1→6) and traces of α(1→4) linkages, respectively. NMR analysis of the EPS revealed unique chemical shifts.




Similar content being viewed by others
References
Ayala-Hernández A, Hassan AN, Goff HD, Corredig M (2008) Effect of protein supplementation on the rheological characteristics of milk permeates fermented with exopolysaccharide-producing Lactococcus lactis subsp. cremoris. Food Hydrocolloids http://dx.doi.org/10.1016/j.foodhyd.2008.11.004
Bernfeld P (1955) Amylases α and β. In: Colowck SP, Kaplon NO (eds) Methods in enzymology (vol 1). Academic Press, New York, pp 149–158
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Cerning J, Bouillanne C, Landon M, Desmazeaud M (1988) Exocellular polysaccharide production by Streptococcus thermophilus. Biotechnol Lett 10:255–260
Cobb BA, Kasper DL (2005) Coming age: carbohydrates and immunity. Eur J Immunol 35:352–356
Dahlquist A (1964) Methods for assay of intestinal disaccharides. Anal Biochem 7:19–25
Degeest B, Vaningelgem F, De Vuyst L (2001) Microbial physiology, fermentation kinetics and process engineering of heteropolysaccharides production by lactic acid bacteria. Int Dairy J 11:747–758
De Vuyst L, Degeest B (1999) Heteropolysaccharides from lactic acid bacteria. FEMS Microbiol Rev 23:153–177
De Vuyst L, De Vin F, Vaningelgem F, Degeest B (2001) Recent developments in the biosynthesis and applications of heteropolysaccharides from lactic acid bacteria. Int Dairy J 11:687–708
Dische Z (1947) A new specific colour reaction of hexuronic acid. J Biol Chem 167:189–198
Duboc P, Mollet B (2001) Applications of exopolysaccharides in dairy industry. Int Dairy J 11:759–768
Dubois M, Gilles KA, Hamilton JK, Rebers PA, Smith F (1956) Colorimetric method for the determination of sugars and related substances. Anal Chem 28:350–356
Dueñas-Chasco MT, Rodriguez-Carrvajal MA, Tejero-Mateo P, Espartero JL, Irastorza-Iribas A, Gil-Serrano AM (1988) Structural analysis of the exopolysaccharides produced by Lactobacillus spp. G-77. Carbo Res 307:125–133
Dueñas M, Munduate A, Perea A, Irastorza A (2003) Exopolysaccharide production by Pediococcus damnosus 2.6 in a semi defined medium under different growth conditions. Int J Food Microbiol 87:113–120
Grobben GJ, Sikkema J, Smith MR, de Bont JAM (1995) Production of extra cellular polysaccharide by Lactobacillus delbrueckii ssp. bulgaricus NCFB 2772 grown in a chemically defined medium. J Appl Bacteriol 79:103–107
Gruter M, Leeflang BR, Kuiper J, Kamerling JP, Vliegenthart FG (1993) Structural characterization of theexopolysaccharide produced by Lactobacillus delbrueckii subsp. bulgaricus IT grown in skimmed milk. Carb Res 239:209–226
Harding LP, Marshall VM, Hernandez Y, Gu Y, Maqsood M, McLay N, Laws AP (2005) Structural characterization of highly branched exopolysaccharide produced by Lactobacillus delbrueckii subsp. bulgaricus NCFB2074. Carbo Res 340:1107–1111
Holt GJ, Krieg NR, Sneath PHA, Staley JT, Williams ST (1994) Bergey’s manual of determinative bacteriology. Williams & Wilkins, Baltimore
JaganMohan Rao L, Krishna Kumari GN, Prakasa Rao NS (1982) Anisofolin-A, a new acylated flavone glucoside from Anisomeles ovata R Br. Heterocycles 19:1655–1661
Jaworska D, Waszkiewicz-Robak B, Kolanowski W, Swiderski F (2005) Relative importance of textural properties in the sensory quality and acceptance of natural yoghurts. Int Dairy Technol 58:39–46
Ketabi A, Soleimanian-Zad S, Kadivar M, Sheikh-Zeinoddin M (2008) Production of microbial exopolysaccharides in the sourdough and its effects on the rheological properties of dough. Food Res Int 41:948–951. doi:10.1016/j.foodres.2008.07.009
Laws AP, Marshall VM (2001) The relevance of exopolysaccharides to the rheological properties in milk fermented with ropy strains of lactic acid bacteria. Int Dairy J 11:709–721
Lin TY, Chang Chien MF (2007) Exopolysaccharides production as affected by lactic acid bacteria and fermentation time. Food Chem 100:1419–1423
Looijesteijn PJ, Hugenholtz J (1999) Uncoupling of growth and exopolysaccharide production by Lactococcus lactis subsp. cremoris NIZO B40 and optimization of its synthesis. J Biosci Bioeng 88:178–182
Makino S, Ikegami S, Kano H, Sashihara T, Sugano H, Horiuchi H, Saito T, Oda M (2006) Immunomodulatory effects of polysaccharides produced by Lactobacillus delbrueckii ssp. bulgaricus OLL1073R–1. J Dairy Sci 89:2873–2881
Mårtensson O, Öste R, Holst O (2000) Lactic acid bacteria in an oat-based nondairy milk substitute: fermentation characteristics and exopolysaccharide formation. Lwt Food Sci Technol 33:525–530
Mozzi F, Vaningelgem F, Hébert EM, Van der Meulen R, Moreno MRF, Font de Valdez G, De Vuyst L (2006) Diversity of heteropolysaccharide-producing lactic acid bacterium strains and their biopolymers. Appl Environ Microbiol 72:4431–4435
Petry S, Furlan S, Waghorne E, Saulnier L, Cerning J, Maguin E (2003) Comparison of the thickening properties of four Lactobacillus delbrueckii subsp bulgaricus strains and physicochemical characterization of their exopolysaccharides. FEMS Microbiol Lett 221:285–291
Pigeon RM, Cuesta EP, Gilliland SE (2002) Binding of free bile acids by cells of yoghurt starter culture bacteria. J Dairy Sci 85:2705–2710
Purwandari U, Shah NP, Vasiljevic T (2007) Effects of exopolysaccharide-producing strains of Streptococcus thermophilus on technological and rheological properties of set-type yoghurt. Int Dairy J 17:1344–1352
Ricciardi A, Parente E, Crudele MA, Zanetti F, Scolari G, Mannazzu I (2002) Exopolysaccharide production by Streptococcus thermophilus SY: production and preliminary characterization of the polymer. J Appl Microbiol 92:297–306
Ruas-Madiedo P, de los Reyes-Gavilán CG (2005) Methods for the screening, isolation and characterization of exopolysaccharides produced by lactic acid bacteria. J Dairy Sci 88:843–856
Sánchez J, Martínez B, Guillén R, Jiménez-Díaz R, Rodríguez A (2006) Culture conditions determine the balance between two different exopolysaccharides produced by Lactobacillus pentosus LPS26. Appl Environ Microbiol 72:7495–7502
Savadogo A, Ouattara CAT, Savadago PW, Barro N, Ouattara AS, Traoré AS (2004) Identification of exopolysaccharides-producing lactic acid bacteria from Burkina Faso fermented milk samples. Afr J Biotechnol 3:189–194
Semjonovs P, Zikmanis P (2008) Evaluation of novel lactose-positive and exopolysaccharide producing strain of Pediococcus pentosaceus for fermented foods. Eur Food Res Technol 227:851–856. doi:10.1007/s00217-007-0796-4
Şengül N, Aslím B, Uçar G, Yücel N, Işik S, Bozkurt H, Sakaoğullaŕı Z, Atalay F (2005) Effects of exopolysaccharide producing probiotic strains on experimental colitis in rats. Dis Colon Rectum 49:250–258
Sharpe ME (1979) Identification of lactic acid bacteria. In: Skinner FA, Lovelock DW (eds) Identification methods for microbiologists. Academic Press, New York, pp 233–259
Shivakumar S, Vijayendra SVN (2006) Production of exopolysaccharides by Agrobacterium sp. CFR-24 using coconut water—a byproduct of food industry. Lett Appl Microbiol 42:477–482
Schiraldi C, Valli V, Molinaro A, Carteni M, De Rosa M (2006) Exopolysaccharides production in Lactobacillus bulgaricus and Lactobacillus casei exploiting microfiltration. J Indus Microbiol Biotechnol 33:384–390
Smitinont T, Tansakul C, Tanasupawat S, Keeratipibul S, Navarini L, Bosco M, Cescutti P (1999) Exopolysaccharide producing lactic acid bacteria strains from traditional Thai fermented foods: isolation, identification and exopolysaccharide characterization. Int J Food Microbiol 51:105–111
Sutherland IW (1972) Bacterial exopolysaccharides. Adv Microbial Physiol 8:143–213
Tallon R, Bressollier P, Urdaci MC (2003) Isolation and characterization of two exopolysaccharides produced by Lactobacillus plantarum EP56. Res Microbiol 154:705–712
Tieking M, Korakli M, Ehrmann MA, Gänzle MG, Vogel RF (2003) In situ production of exopolysaccharides during sourdough fermentation by cereal and intestinal isolates of lactic acid bacteria. Appl Environ Microbiol 69:945–952
Torino MI, Mozzi F, Font de Valdez G (2005) Exopolysaccharide biosynthesis by Lactobacillus helveticus ATCC 15807. Appl Microbiol Biotechnol 68:259–265
van Calsteren MR, Pau-Roblot C, Begin A, Roy D (2002) Structure determination of the exopolysaccharide produced by Lactobacillus rhamnosus strains RW-9595 M and R. Biochem J 363:7–17
Vaningelgem F, Zamfir M, Mozzi F, Adriany T, Vancanney M, Swings J, De Vuyst L (2004) Biodiversity of exopolysaccharides produced by Streptococcus thermophilus strains is reflected in their production and their molecular and functional characteristics. Appl Environ Microbiol 70:900–912
Vasiljevic T, Shah NP (2007) Fermented milk—Health benefits beyond probiotic effect. In: Hui YH (ed) Handbook of food product manufacturing, vol 2. Wiley-Interscience, Hoboken, pp 99–116
Vijayendra SVN, Sharath Babu RS (2008) Optimization of a new heteropolysaccharide production by a native isolate of Leuconostoc sp CFR-2181. Lett Appl Microbiol 46:643–648. doi:10.1111/j.1472-765x.2008.02361.x
Vijayendra SVN, Palanivel G, Mahadevamma S, Tharanathan RN (2008) Physico-chemical characterization of an exopolysacchairde produced by a non-ropy strain of Leuconostoc sp. CFR 2181isolated from dahi, an Indian traditional lactic fermented milk product. Carbohydr Poly 72:300–307. doi:10.1016/j.carbpol.2007.08.016
Vinderola CG, Perdigón G, Duarte J, Farnworth E, Matar C (2006) Effects of the oral administration of the exopolysaccharide produced by Lactobacillus kefiranofaciens on the gut mucosal immunity. Cytokine 36:254–260
Welman AD, Maddox IS (2003) Exopolysaccharides from lactic acid bacteria: perspectives and challenges. Trends Biotechnol 21:269–274
Zisu B, Shah NP (2005) Textural and functional changes in low fat mozzarella cheeses in relation to proteolysis and microstructure as influenced by the use of fat replacers, pre-acidification and EPS starter. Int Dairy J 15:957–972
Acknowledgments
The research grant received from United Nations University, Tokyo, as a follow-up project of UNU-Kirin Fellowship to first author is highly acknowledged. Authors are thankful to Director, CFTRI, and Mysore for providing the facilities. The technical help received from the staff at Central Instrumentation Services & Facility, and NMR facility, CFTRI, Mysore, in analyzing the polymer is acknowledged.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Erko Stackebrandt.
Rights and permissions
About this article
Cite this article
Vijayendra, S.V.N., Palanivel, G., Mahadevamma, S. et al. Physico-chemical characterization of a new heteropolysaccharide produced by a native isolate of heterofermentative Lactobacillus sp. CFR-2182. Arch Microbiol 191, 303–310 (2009). https://doi.org/10.1007/s00203-008-0453-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-008-0453-8