Abstract
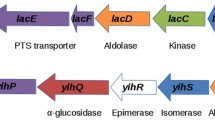
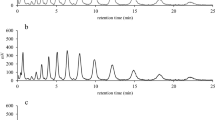
Physiological, biochemical and macroarray analyses of Lactococcus lactis IL1403 and its ccpA and bglR single and double mutants engaged in lactose and β-glucosides catabolism were performed. The kinetic analysis indicated the presence of different transport systems for salicin and cellobiose. The control of salicin catabolism was found to be mediated by the transcriptional regulator BglR and the CcpA protein. The transcriptional analysis by macroarray technology of genes from the PEP:PTS regions showed that several genes, like ybhE, celB, ptcB and ptcA, were expressed at higher levels both in wild type cells exposed to cellobiose and in the ccpA mutant. We also demonstrated that in L. lactis IL1403 cultured on medium with cellobiose and lactose as carbon sources, after the first phase of cellobiose consumption and then co-metabolism of the two sugars, when cellobiose is exhausted the strain uses lactose as the only carbon source. These data could indicate that lactose and cellobiose are transported by a unique system—a PTS carrier induced by the presence of cellobiose, and negatively controlled by the CcpA regulator.





Similar content being viewed by others
Abbreviations
- LAB:
-
Lactic acid bacteria
- CDM:
-
Chemically defined medium
References
Aleksandrzak T, Kowalczyk M, Kok J, Bardowski J (2000) Regulation of carbon catabolism in Lactococcus lactis. In: Bielecki S, Tramper J, Polak J (eds) Food biotechnology, vol 17. Elsevier Science BV, Amsterdam, The Netherlands, pp 61–66
Aleksandrzak-Piekarczyk T, Kok J, Renault P, Bardowski J (2005) Alternative lactose catabolic pathway in Lactococcus lactis. Appl Environ Microbiol 71(10):6060–6069
Aung-Hilbrich LM, Seidel G, Wagner A, Hillen W (2002) Quantification of the influence of HprSer46P on CcpA-cre interaction. J Mol Biol 319:77–85
Bardowski J, Ehrlich SD, Chopin A (1994) BglR protein, which belongs to the BglG family of transcriptional antiterminators, is involved in β-glucoside utilization in Lactococcus lactis. J Bacteriol 176:5681–5685
Bardowski J, Ehrlich SD, Chopin A (1995) A protein, belonging to a family of RNA-binding transcriptional antiterminators, controls β-glucoside assimilation in Lactococcus lactis. Dev Biol Stand Basel Karger 85:555–558
Bolotin A, Wincker P, Mauger S, Jaillon O, Malarme K, Weissenbach J, Ehrlich SD, Sorokin A (2001) The complete genome sequence of the lactic acid bacterium Lactococcus lactis ssp lactis IL1403. Genome Res 11:731–753
Cocaign-Bousquet M, Garriques C, Loubiere P, Lindley ND (1996) Physiology of pyruvate metabolism in Lactococcus lactis. Antonie van Leeuwenhoek 70:253–267
Cocaign-Bousquet M, Even S, Loubiere P, Lindley ND (2002) Anaerobic sugar catabolism in Lactococcus lactis: genetic regulation and enzyme control over pathway flux. Appl Microbiol Biotechnol 60:24–32
Curtis SJ, Epstein W (1975) Phosphorylation of d-glucose in Escherichia coli mutants defective in glucose phosphotransferase, mannose phosphotransferase, and glucokinase. J Bacteriol 122:1189–1199
Dalet K, Arous S, Cenatiempo Y, Hechard Y (2003) Characterization of a unique sigma54-dependent PTS operon of the lactose family in Listeria monocytogenes. Biochimie 85(7):633–638
Deutscher J, Küster E, Bergstedt U, Charrier V, Hillen W (1995) Protein kinase-dependent HPr/CcpA interaction links glycolytic activity to carbon catabolite repression in Gram-positive bacteria. Mol Microbiol 15:1049–1053
Even S, Lindley ND, Cocaign-Bousquet M (2001) Molecular physiology of sugar catabolism in Lactococcus lactis IL1403. J Bacteriol 183:3817–3824
Fontaine L, Even S, Soucaille P, Lindley ND, Cocaign-Bousquet M (2001) Transcript quantification based on chemical labeling of RNA associated with fluorescent detection. Anal Biochem 298:246–252
Garriques C, Loubiere P, Lindley ND, Cocaign-Bousquet M (1997) Control of the shift from homolactic acid to mixed-acid fermentation in Lactococcus lactis: predominant role of the NADH/NAD+ ratio. J Bacteriol 179:5282–5287
Hueck CJ, Kraus A, Schmiedel D, Hillen W (1995) Cloning, expression and functional analyses of the catabolite control protein CcpA from Bacillus subtilis. Mol Microbiol 16(5):855–864
Jones BE, Dossonnet V, Küster E, Hillen W, Deutscher J, Klevit RE (1997) Binding of the catabolite repressor protein CcpA to its DNA target is regulated by phosphorylation of its corepressor HPr. J Biol Chem 272:26530–26535
Kowalczyk M, Bardowski J (2007) Regulation of sugar catabolism in Lactococcus lactis. Crit Rev Microbiol 33:1–13
Kowalczyk M, Borcz B, Plochocka D, Bardowski J (2007) In vitro DNA binding of purified CcpA protein from Lactococcus lactis IL1403. Acta Biochim Pol 54(1):71–78
Küster E, Hilbich T, Dahl MK, Hillen W (1999) Mutations in catabolite control protein CcpA separating growth effects from catabolite repression. J Bacteriol 181(13):4125–4128
Luesink EJ, van Herpen RE, Grossiord BP, Kuipers OP, de Vos WM (1998) Transcriptional activation of the glycolytic las operon and catabolite repression of the gal operon in Lactococcus lactis are mediated by the catabolite control protein CcpA. Mol Microbiol 30(4):789–798
Otto R, ten Brink T, Veldkamp H, Konings WN (1983) The relation between growth rate and electrochemical proton gradient of Streptococcus cremoris. FEMS Microbiol Lett 16:69–74
Poolman B, Konings WN (1988) Relation of growth of Streptococcus lactis and Streptococcus cremoris to amino acid transport. J Bacteriol 170:700–707
Ramseier TM, Reizer J, Küster E, Hillen W, Saier MH Jr (1995) In vitro binding of the CcpA protein of Bacillus megaterium to cis-acting catabolite responsive elements (CREs) of Gram-positive bacteria. FEMS Microbiol Lett 129:207–214
Saier MH Jr, Chauvaux S, Cook GM, Deutscher J, Paulsen IT, Reizer J, Ye JJ (1996) Catabolite repression and inducer control in gram-positive bacteria. Microbiology 142:217–230
Simons G, Nijhuis M, de Vos WM (1993) Integration and gene replacement in the Lactococcus lactis lac operon: induction of a cryptic phospho-beta-glucosidase in LacG-deficient strains. J Bacteriol 175(16):5168–5175
Sinha RP (1991) Genetic characterization of partial lactose-fermenting revertants from lactose-negative mutants of lactococci. Can J Microbiol 37:281–286
Titgemeyer F, Hillen W (2002) Global control of sugar metabolism: a Gram-positive solution. Antonie van Leeuwenhoek 82:59–71
Wray LV Jr, Pettengill FK, Fisher SH (1994) Catabolite repression of the Bacillus subtilis hut operon requires a cis-acting site located downstream of the transcription initiation site. J Bacteriol 176(7):1894–1902
Acknowledgments
This work was supported in part by KBN grant 6 P06G 055 20, the Polish–French scientific program Polonium and Marie Curie Host fellowship.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Erko Stackebrandt.
Rights and permissions
About this article
Cite this article
Kowalczyk, M., Cocaign-Bousquet, M., Loubiere, P. et al. Identification and functional characterisation of cellobiose and lactose transport systems in Lactococcus lactis IL1403. Arch Microbiol 189, 187–196 (2008). https://doi.org/10.1007/s00203-007-0308-8
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-007-0308-8