Abstract
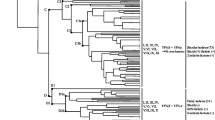
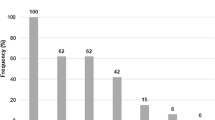
Producers of cereulide, the emetic toxin of Bacillus cereus, are known to constitute a specific subset within this species. We investigated physiological and genetic properties of 24 strains of B. cereus including two high cereulide producers (600–1,800 ng cereulide mg−1 wet weight biomass), seven average producers (180–600 ng cereulide mg−1 wet weight biomass), four low cereulide producers (20–160 ng cereulide mg−1 wet weight biomass) and 11 non-producers representing isolates from food, food poisoning, human gut and environment. The 13 cereulide producers possessed 16S rRNA gene sequences identical to each other and identical to that of B. anthracis strains Ames, Sterne from GenBank and strain NC 08234–02, but showed diversity in the adk gene (two sequence types), in ribopatterns obtained with EcoRI and PvuII (three types of patterns), in tyrosin decomposition, haemolysis and lecithin hydrolysis (two phenotypes). The cereulide-producing isolates from the human gut represented two ribopatterns of which one was novel to cereulide-producing B. cereus and two phenotypes. We conclude that the cereulide-producing B. cereus are genetically and biochemically more diverse than hitherto thought.

Similar content being viewed by others
References
Ackermann H-W, Du Bow MS (1987) Viruses of Prokaryotes, vol 1. CRC Press, Boca Raton, pp 103–108
Agata N, Mori M, Ohta M, Suwan S, Ohtani I, Isobe M (1994) A novel dodecadepsipeptide, cereulide, isolated from Bacillus cereus causes vacuole formation in HEp-2 cells. FEMS Microbiol Lett 121:32–34
Agata N, Ohta M, Mori M, Isobe M (1995) A novel dodecadepsipeptide, cereulide, is an emetic toxin of Bacillus cereus. FEMS Microbiol Lett 129:17–20
Agata N, Ohta M, Mori M (1996) Production of an emetic toxin, cereulide, is associated with a specific class of Bacillus cereus. Curr Microbiol 33:67–69
Andersson MA, Jääskeläinen E, Shaheen R, Pirhonen T, Wijnands LM, Salkinoja-Salonen MS (2004) Sperm bioassay for rapid detection of cereulide-producing Bacillus cereus in food and related environments. Int J Food Microbiol 94:175–183. DOI 10.1016/j.ijfoodmicro.2004.01.018
AOAC (1995) Bacillus cereus in foods. Enumeration and confirmation. Microbiological methods, AOAC Official Method 980.3. In: Cunniff P (ed) Official methods of analysis of AOAC international, 16th edn, vol 1. AOAC International, Arlington
Ash C, Collins MD (1992) Comparative analysis of 23S ribosomal RNA gene sequences of Bacillus anthracis and emetic Bacillus cereus determined by PCR-direct sequencing. FEMS Microbiol Lett 94:75–80
Ash C, Farrow JAE, Dorsch M, Stackebrandt E, Collins MD (1991) Comparative analysis of Bacillus anthracis,Bacillus cereus, and related species on the basis of reverse transcriptase sequencing of 16S rRNA. Int J Syst Bacteriol 41:343–346
Ausubel F, Brent R, Kingston RE, Moore DD, Seidman JG, Smith JA, Struhl K (1994) Current protocols in molecular biology, 3rd edn. A compendium of methods from current protocols in molecular biology, vol 1, Unit 2.4. Wiley, New York, pp 2.4.1–2.4.2
Brown ER, Cherry WB (1955) Specific identification of Bacillus anthracis by means of a variant bacteriophage. J Infect Dis 96:34–39
Drobniewski F (1993) Bacillus cereus and related species. Clin Microbiol Rev 6:324–338
Edwards U, Rogall T, Blöcker H, Emde M, Böttger EC (1998) Isolation and direct complete nucleotide determination of entire genes. Characterization of a gene coding for 16S ribosomal RNA. Nucleic Acids Res l 17(19):7843–7853
Ehling-Schulz M, Fricker M, Scherer S (2004) Identification of emetic toxin producing Bacillus cereus strains by a novel molecular assay. FEMS Microbiol Lett 232(2):180–195
Ehling-Schulz M, Vukov N, Schulz A, Shaheen R, Andersson M, Märtlbauer E, Scherer S (2005a) Identification and partial characterization of the nonribosomal peptide synthetase gene responsible for cereulide production in emetic Bacillus cereus. Appl Environ Microbiol 71:105–113. DOI: 10.1128/AEM.71.1.105–113.2005
Ehling-Schulz M, Svensson B, Guinebretiere M-H, Lindbäck T, Andersson M, Schulz A, Fricker M, Christiansson A, Granum PE, Märtlbauer E, Nguyen-The C, Salkinoja-Salonen M, Scherer S (2005b) Emetic toxin formation of Bacillus cereus is restricted to a single evolutionary lineage of closely related strains. Microbiology 151:183–197. DOI: 10.1099/mic.0.27607–0
Gerhardt P, Murray RGE, Wood WA, Krieg NR (1994) Methods for general and molecular bacteriology, Chap 25. American Society for Microbiology, Washington, pp 630
Häggblom MM, Apetroaie C, Andersson MA, Salkinoja-Salonen MS (2002) Quantitative analysis of cereulide, the emetic toxin of Bacillus cereus, produced under various conditions. Appl Environ Microbiol 68:2479–2483
Hall TA (1999) BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucl Acids Symp Ser 41:95–98
Hallaksella A-M, Väisänen O, Salkinoja-Salonen M (1991) Identification of Bacillus species isolated from Picea abies by physiological tests, phage typing and fatty acid analysis. Scand J For Res 6:365–377
Helgason E, Økstad OE, Caugant DA, Johansen HA, Fouet A, Mock M, Hegna I, Kolstø A-B (2000) Bacillus anthracis,Bacillus cereus, and Bacillus thuringiensis—one species on the basis of genetic evidence. Appl Environ Microbiol 66:2627–2630
Helgason E, Tourasse NJ, Meisal R, Caugant DA, Kolstø A-B (2004) Multilocus sequence typing scheme for bacteria of the Bacillus cereus Group. Appl Environ Microbiol 70:191–201. DOI: 10.1128/AEM.70.1.191–201.2004
Hoffmaster AR, Ravel J, Rasko DA, Chapman GD, Chute MD, Marston CK, De BK, Sacchi CT, Fitzgerald C, Mayer LW, Maiden MCJ, Priest F, Barker M, Jiang L, Cer RZ, Rilstone J, Peterson SN, Weyant RS, Galloway DR, Read TD, Popovic T, Fraser CM (2004) Identification of anthrax toxin genes in a Bacillus cereus associated with an illness resembling inhalation anthrax. Proc Natl Acad Sci USA 101(22):8449–8454. http://www.pnas.org/cgi/doi/10.1073/pnas.0402414101
Horwood PF, Burgess GW, Oakey HJ (2004) Evidence for non-ribososmal peptide synthetase production of cereulide (the emetic toxin) in Bacillus cereus. FEMS Microbiol Lett 236:319–324
IDF (1998) Dried milk products. Enumeration of Bacillus cereus. Most probable number technique. Standard 181:1998, International Dairy Federation, Brussels, Belgium
ISO (1993) Microbiology, general guidance for the enumeration of Bacillus cereus, colony count technique at 30°C. Standard 7932, 2nd edn. International Organisation for Standardisation, Geneva
Jääskeläinen EL, Häggblom MM, Andersson MA, Vanne L, Salkinoja-Salonen MS (2003) Potential of Bacillus cereus for producing an emetic toxin, cereulide, in bakery products: quantitative analysis by chemical and biological methods. J Food Prot 66:1047–1054
Jukes TH, Cantor CR (1969) Evolution of protein molecules. In: Munro HN (eds) Mammalian protein metabolism, vol 3. Academic, New York, pp 21–132
Kotiranta AK, Ito H, Haapasalo MPP, Lounatmaa K (1999) Radiation sensitivity of Bacillus cereus with and without a crystalline surface protein layer. FEMS Microbiol Lett 179:275–280
Mac Faddin JF (1985) Media for isolation-cultivation-identification-maintenance of medical bacteria, vol 1. Williams and Wilkins, Baltimore, p 861
Mac Faddin JF (2000) Biochemical Tests for identification of medical bacteria. Lippincott & Wilkins, USA, pp 173; 328
Maidak BL, Cole JR, Parker CT, Garrity Jr GM, Larsen N, Li B, Lilburn TG, McCaughey MJ, Olsen J, Overbeek R, Pramanik S, Schmidt TM, Tiedje JM, Woese CR (1999) A new version of the RDP (Ribosomal Database Project). Nucleic Acids Res 27:171–173
NMKL (1997) Bacillus cereus Determination in foods, NMKL Method No. 67, 4th edn. The Nordic Committee on Food Analysis, Oslo, Norway
Parry JM, Turnbull PCB, Gibson JR (1983) A colour atlas of Bacillus species. Wolfe Medical Atlases-19, Wolfe Medical Publications Ltd, UK, p 19
Pirttijärvi TSM, Graeffe TH, Salkinoja-Salonen MS (1996) Bacterial contaminants in liquid packaging boards: assessment of potential food spoilage. J Appl Bacteriol 81:445–458
Pirttijärvi TSM, Andersson MA, Scoging AC, Salkinoja-Salonen MS (1999) Evaluation of methods for recognising strains of the Bacillus cereus Group with food poisoning potential among industrial and environmental contaminants. Syst Appl Microbiol 22:133–144
Priest FG, Barker M, Baillie LWJ, Holmes EC, Maiden MCJ (2004) Population structure and evolution of the Bacillus cereus group. J Bacteriol 186 23:7959–7970. DOI: 10.1128/JB.186.23.7959–7970.2004
Qi Y, Patra G, Liang X, Williams LE, Rose S, Redkar RJ, Del Vecchio V (2001) Utilization of the rpoB gene as a specific chromosomal marker for real-time PCR detection of Bacillus anthracis. Appl Environ Microbiol 67:3720–3727
Rainey FA, Ward-Rainey N, Kroppenstedt RM, Stackebrandt E (1996) The genus Nocardiopsis represents a phylogenetically coherent taxon and a distinct actinomycete lineage: proposal of Nocardiopsaceae fam. nov. Int J Syst Bacteriol 46:1088–1092
Rhodehamel EJ, Harmon SM (1998) Bacillus cereus, food and drug administration, bacteriological analytical manual, 8th edn, revision A. AOAC International, Gaithersburg, pp 14.01–14.08
Sacchi CT, Whitney AM, Mayer LW, Morey R, Steigerwalt A, Boras A, Weyant RS, Popovic T (2002) Sequencing of 16S rRNA gene: a rapid tool for identification of Bacillus anthracis. Emerg Infect Dis 8(10):1117–1123
Shinagawa K (1993) Serology and characterisation of toxigenic Bacillus cereus. Neth Milk Dairy J 47:89–102
Slamti L, Perchat S, Gominet M, Vilas-Bôas G, Fouet A, Mock M, Sanchis V, Chaufaux J, Gohar M, Lereclus D (2004) Distinct Mutations in PlcR Explain Why Some Strains of the Bacillus cereus Group Are Nonhemolytic. J Bacteriol 186(11):3531–3538. DOI: 10.1128/JB.186.11.3531–3538.2004
Turnbull PCB, Kramer JM (1985) Intestinal carriage of Bacillus cereus: faecal isolation studies in three population groups. J Hyg 95:629–638
Turnbull PCB, Kramer JM, Jørgensen K, Gilbert RJ, Melling J (1979) Properties and production characteristics of vomiting, diarrhoeal, and necrotizing toxins of Bacillus cereus. Am J Clin Nutr 32:219–228
Turnbull PCB, Böhm R, Cosivi O, Doganay M, Hugh-Jones ME, Joshi DD, Lalitha MK, De Vos V (1998) Guideliness for the surveillance and control of anthrax in humans and animals. DOI: WHO/EMC/ZDI/98.6 (online accessed 12.1.2005)
Wijnands LM, Dufrenne JB, Leusden FM (2002) National Institute of Public Health and the Environment (RIVM) Report 250912002 DOI: 250912002 (online accessed 7.2.2005)
Acknowledgements
This study was supported by the Academy of Finland, grant to the Centre of Excellence “Microbial Resources” (grant 53305) and the European Commission, Quality of Life Programme, Key action 1 (Health, Food & Nutrition), contract QLK1-CT-2001-00854. We want to thank to Prof. Dr H-W. Ackermann and Dr Anja Siitonen for their valuable co-operation. We also thank Douwe Hoornstra for the help with the Bionumerics program and Ilona Oksanen for helpful discussions and advice. We thank Viikki Science Library for the excellent information service, the Faculty Instrument Centre for technical services, and Leena Steininger, Hannele Tukiainen and Tuula Suortti for many kinds of help.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Apetroaie, C., Andersson, M.A., Spröer, C. et al. Cereulide-producing strains of Bacillus cereus show diversity. Arch Microbiol 184, 141–151 (2005). https://doi.org/10.1007/s00203-005-0032-1
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00203-005-0032-1