Abstract.
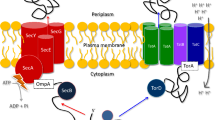
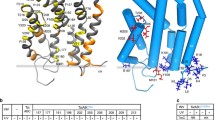
The Tat protein-export system serves to translocate folded proteins, often containing redox cofactors, across the bacterial inner membrane. Substrate proteins are directed to the Tat apparatus by distinctive N-terminal signal peptides containing a consensus SRRxFLK 'twin-arginine' motif. Here we review recent studies of the Tat system with particular emphasis on the assembly of membrane-bound respiratory complexes. We discuss the connection between Tat targeting and topological organisation of the complexes and consider the role of chaperone proteins in cofactor insertion and Tat targeting. The crystal structure of Escherichia coli formate dehydrogenase-N demonstrates that some Tat substrates are integral membrane proteins. Sequence analysis suggests that one-quarter of all traffic on the E. coli Tat pathway is inner-membrane proteins.
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Sargent, F., Berks, B.C. & Palmer, T. Assembly of membrane-bound respiratory complexes by the Tat protein-transport system. Arch Microbiol 178, 77–84 (2002). https://doi.org/10.1007/s00203-002-0434-2
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/s00203-002-0434-2