Abstract
Summary
We ascertained the fracture risk factors stratified by vertebral and non-vertebral sites in rheumatoid arthritis (RA) females. Bone/muscle features, but not disease activity, were the main markers for fractures in this long-standing RA population: low trabecular bone score (TBS) for vertebral fracture and decreased appendicular muscle mass for non-vertebral fracture.
Purpose
To assess risk factors for fractures, including clinical, laboratory and dual energy X-ray absorptiometry (DXA) parameters (bone mass, trabecular bone score—TBS, muscle mass) in women with established rheumatoid arthritis (RA).
Methods
Three hundred females with RA (ACR, 2010) were studied. Clinical data were obtained by questionnaire and disease activity by composite indices (DAS28, CDAI, SDAI), C-reactive protein (CRP) and erythrocyte sedimentation rate (ESR). Bone mineral density (BMD), TBS, body composition and Vertebral Fracture Assessment (VFA) were performed by DXA. Logistic regression models were constructed to identify factors independently associated with vertebral (VF) and non-vertebral fractures (NVF), separately.
Results
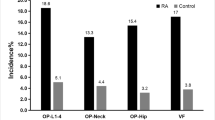
Through rigorous eligibility criteria, a total of 265 women were yielded for final data analysis (median age, 55 [22–86] years; mean disease duration, 16.2 years). Prevalence of VF and NVF were 30.6% and 17.4%, respectively. In multivariate analyzes, TBS (OR = 1.6, 95%CI = 1.09–2.36, p = 0.017), CRP (OR = 1.54, 95%CI = 1.15–2.08, p = 0.004), and parathormone (OR = 1.24, 95%CI = 1.05–1.45, p = 0.009) were risk factors for VF, whereas low appendicular muscle mass (OR = 2.71; 95%CI = 1.01–7,28; p = 0.048), body mass index (BMI) (OR = 0.90, 95%CI = 0.82–0.99; p = 0.025), ESR (OR = 1.18, 95%CI = 1.01–1,38, p = 0,038) and hip BMD (OR = 1.82, 95%CI = 1.10–3.03, p = 0.02) were associated with NVF.
Conclusion
In women with long-term RA, markers of fractures differed between distinct skeletal sites (vertebral and non-vertebral). The magnitude of association of bone/muscle parameters with fracture (TBS for VF and appendicular muscle mass for NVF) was greater than that of the association between RA activity and fracture. TBS seems to have greater discriminative power than BMD to identify subjects with VF in long-standing RA.



Similar content being viewed by others
Data Availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
References
Alarcón GS (1995) Epidemiology of rheumatoid arthritis. Rheum Dis Clin North Am 21:589–604
Conforti A, Cola ID, Pavlych V et al (2021) Beyond the joints, the extra-articular manifestations in rheumatoid arthritis. Autoimmun Rev 20:102735. https://doi.org/10.1016/j.autrev.2020.102735
Fardellone P, Salawati E, Monnier LL, Goëb V (2020) Bone loss, osteoporosis, and fractures in patients with rheumatoid arthritis: a review. J Clin Med 9:3361. https://doi.org/10.3390/jcm9103361
Xue AL, Wu SY, Jiang L, Feng AM, Guo HF, Zhao P (2017) Bone fracture risk in patients with rheumatoid arthritis: a meta-analysis. Medicine (Baltimore) 96:e6983. https://doi.org/10.1097/md.0000000000006983
Silva BC, Leslie WD, Resch H et al (2014) Trabecular bone score: a noninvasive analytical method based upon the DXA image. J Bone Miner Res 29:518–530. https://doi.org/10.1002/jbmr.2176
McCloskey EV, Odén A, Harvey NC et al (2016) A meta-analysis of trabecular bone score in fracture risk prediction and its relationship to FRAX. J Bone Miner Res 31:940–948. https://doi.org/10.1002/jbmr.2734
Choi YJ, Ock SY, Chung YS (2016) Trabecular bone score (TBS) and TBS-adjusted fracture risk assessment tool are potential supplementary tools for the discrimination of morphometric vertebral fractures in postmenopausal women with type 2 diabetes. J Clin Densitom 19:507–514. https://doi.org/10.1016/j.jocd.2016.04.001
Dos Santos LM, Ohe MN, Pallone SG et al (2021) Trabecular bone score (TBS) in primary hyperparathyroidism (PHPT): a useful tool? J Clin Densitom 24:563–570. https://doi.org/10.1016/j.jocd.2021.04.001
Cruz-Jentoft AJ, Bahat G, Bauer J, Boirie Y, Bruyère O, Cederholm T (2019) Sarcopenia: revised European consensus on definition and diagnosis. Age Ageing 48:16–31. https://doi.org/10.1093/ageing/afy169
Radkowski MJ, Slawinski P, Targowski T (2020) Osteosarcopenia in rheumatoid arthritis treated with glucocorticosteroids – essence, significance, consequences. Reumatologia 58:101–106. https://doi.org/10.5114/reum.2020.95363
Aletaha D, Neogi T, Silman AJ et al (2010) 2010 Rheumatoid arthritis classification criteria: an American College of Rheumatology/European League Against Rheumatism collaborative initiative. Arthritis Rheum 62:2569–2581. https://doi.org/10.1002/art.27584
Levey AS, Stevens LA, Schmid CH et al (2009) A new equation to estimate glomerular filtration rate. Ann Intern Med 150:604–612. https://doi.org/10.7326/0003-4819-150-9-200905050-00006
O’Neill TW, Cooper C, Cannata JB et al (1994) Reproducibility of a questionnaire on risk factors for osteoporosis in a multicentre prevalence survey: the European Vertebral Osteoporosis Study. Int J Epidemiol 23:559–565. https://doi.org/10.1093/ije/23.3.559
O’Neills TW, Felsenberg D, Varlow J, Cooper C, Kanis JA, Silman AJ (1996) The prevalence of vertebral deformity in European men and women: the European Vertebral Osteoporosis Study. J Bone Miner Res 11:1010–1018. https://doi.org/10.1002/jbmr.5650110719
Hagstromer M, Oja P, Sjostrom M (2006) The International Physical Activity Questionnaire (IPAQ): a study of concurrent and construct validity. Public Health Nutr 9:755–762. https://doi.org/10.1079/phn2005898
Craig CL, Marshall AL, Sjöström M et al (2003) International physical activity questionnaire: 12-country reliability and validity. Med Sci Sports Exerc 35:1381–95. https://doi.org/10.1249/01.mss.0000078924.61453.fb
Fries JF, Spitz P, Kraines RG, Holman HR (1980) Measurement of patient outcome in arthritis. Arthritis Rheum 23:137–145. https://doi.org/10.1002/art.1780230202
Ferraz MB, Oliveira LM, Araujo PM et al (1990) Crosscultural reliability of the physical ability dimension of the health assessment questionnaire. J Rheumatol 17:813–817
Baim S, Binkley N, Bilezikian JP et al (2008) Official positions of the International Society for Clinical Densitometry and executive summary of the 2007 ISCD Position Development Conference. J Clin Densitom 11:75–91. https://doi.org/10.1016/j.jocd.2007.12.007
Studenski SA, Peters KW, Alley DE et al (2014) The FNIH sarcopenia project: rationale, study description, conference recommendations, and final estimates. J Gerontol A Biol Sci Med Sci 69:547–558. https://doi.org/10.1093/gerona/glu010
Kanis JA, Johnell O, Oden A, Johansson H, McCloskey E (2008) FRAX and the assessment of fracture probability in men and women from the UK. Osteoporos Int 19:385–397. https://doi.org/10.1007/s00198-007-0543-5
McCloskey EV, Odén A, Harvey NC et al (2015) Adjusting fracture probability by trabecular bone score. Calcif Tissue Int 96:500–509. https://doi.org/10.1007/s00223-015-9980-x
Gregson CL, Armstrong DJ, Bowden J et al (2022) UK clinical guideline for the prevention and treatment of osteoporosis. Arch Osteoporos 17:58. https://doi.org/10.1007/s11657-022-01061-5
Kanis JA, Johansson H, Oden A, McCloskey EV (2011) Guidance for the adjustment of FRAX according to the dose of glucocorticoids. Osteoporos Int 22:809–816. https://doi.org/10.1007/s00198-010-1524-7
Genant HK, Wu CY, van Kujik C, Nevitt MC (1993) Vertebral fracture assessement using a semiquantitaive technique. J Bone Miner Res 8:1137–1148. https://doi.org/10.1002/jbmr.5650080915
Cooper C, Atkinson EJ, O’Fallon WM, Melton LJ 3rd (1992) Incidence of clinically diagnosed vertebral fractures: a population-based study in Rochester, Minnesota, 1985–1989. J Bone Miner Res 7:221–227. https://doi.org/10.1002/jbmr.5650070214
Schousboe JT, Shepherd JA, Bilezikian JP, Baim S (2013) Executive summary of the 2013 International Society for Clinical Densitometry Position Development Conference on bone densitometry. J Clin Densitom 16:455–66. https://doi.org/10.1016/j.jocd.2013.08.004
Liu Y, Jiang J, Mo M, Sun X, Yu K (2022) Incidence and risk factors for vertebral fracture in rheumatoid arthritis: an update meta-analysis. Clin Rheumatol 41:1313–1322. https://doi.org/10.1007/s10067-021-06046-2
Mohammad A, Lohan D, Bergin D et al (2014) The prevalence of vertebral fracture on vertebral fracture assessment imaging in a large cohort of patients with rheumatoid arthritis. Rheumatology (Oxford) 53:821–827. https://doi.org/10.1093/rheumatology/ket353
Bréban S, Briot K, Kolta S et al (2012) Identification of rheumatoid arthritis patients with vertebral fractures using bone mineral density and trabecular bone score. J Clin Densitom 15:260–266. https://doi.org/10.1016/j.jocd.2012.01.007
Kim D, Cho SK, Kim JY, Choi YY, Sung YK (2016) Association between trabecular bone score and risk factor for fractures in Korean female patients with rheumatoid arthritis. Mod Rheumatol 26:540–545. https://doi.org/10.3109/14397595.2015.1101212
Choi YJ, Chung YS, Suh CH, Jung JY, Kim HA (2017) Trabecular bone score as a supplementary tool for the discrimination of osteoporotic fractures in postmenopausal women with rheumatoid arthritis. Medicine (Baltimore) 96:e8661. https://doi.org/10.1097/md.0000000000008661
Del Rio LM, Winzenrieth R, Cormier C, Di Gregorio S (2013) Is bone microarchitecture status of the lumbar spine assessed by TBS related to femoral neck fracture? A Spanish case-control study. Osteoporos Int 24:991–998. https://doi.org/10.1007/s00198-012-2008-8
Li TH, Chang YS, Liu CW et al (2021) The prevalence and risk factors of sarcopenia in rheumatoid arthritis patients: a systematic review and meta-regression analysis. Semin Arthritis Rheum 51:236–245. https://doi.org/10.1016/j.semarthrit.2020.10.002
Carvalho GD, Bonfiglioli K, Caparbo VF, Takayama L, Pereira RMR, Domiciano DS (2020) Changes to body composition in women with long-standing established rheumatoid arthritis: differences by level of disease activity. J Clin Densitom 23:639–646. https://doi.org/10.1016/j.jocd.2019.06.002
Brance ML, Di Gregorio S, Pons-Estel BA et al (2021) Prevalence of sarcopenia and whole-body composition in rheumatoid arthritis. J Clin Rheumatol 27:S153–S160. https://doi.org/10.1097/rhu.0000000000001549
Chen YF, Zong HX, Xu SQ et al (2021) Synergistic effect of sarcopenia and poor balance on osteoporotic vertebral fracture in Chinese patients with rheumatoid arthritis. Clin Rheumatol 40:3627–3637. https://doi.org/10.1007/s10067-021-05703-w
Zhang M, Xu S, Zong H et al (2022) Effect of sarcopenia and poor balance on vertebral spinal osteoporotic fracture in female rheumatoid arthritis. Sci Rep 12:9477. https://doi.org/10.1038/s41598-022-13339-2
Bow CH, Cheung E, Cheung CL et al (2012) Ethnic difference of clinical vertebral fracture risk. Osteoporosis Int 23:879–885. https://doi.org/10.1007/s00198-011-1627-9
Lee YK, Jang S, Jang S et al (2012) Mortality after vertebral fracture in Korea: analysis of the National Claim Registry. Osteoporosis Int 23:1859–1865. https://doi.org/10.1007/s00198-011-1833-5
Furuya T, Kotake S, Inoue E et al (2007) Risk factors associated with incident clinical vertebral and nonvertebral fractures in Japanese women with rheumatoid arthritis: a prospective 54-month observational study. J Rheumatol 34:303–310
El Maghraoui A, Rezqi A, Mounach A, Achemlal L, Bezza A, Ghozlani I (2010) Prevalence and risk factors of vertebral fractures in women with rheumatoid arthritis using vertebral fracture assessment. Rheumatology (Oxford) 49:1303–1310. https://doi.org/10.1093/rheumatology/keq084
Tong JJ, Xu SQ, Zong HX, Pan MJ, Teng YZ, Xu JH (2020) Prevalence and risk factors associated with vertebral osteoporotic fractures in patients with rheumatoid arthritis. Clin Rheumatol 39:357–364. https://doi.org/10.1007/s10067-019-04787-9
Abtahi S, Driessen JHM, Burden AM et al (2022) Low-dose oral glucocorticoid therapy and risk of osteoporotic fractures in patients with rheumatoid arthritis: a cohort study using the clinical practice research datalink. Rheumatology (Oxford) 61:1448–1458. https://doi.org/10.1093/rheumatology/keab548
Leslie WD, Hans D, Silva BC (2023) Fracture prediction from trabecular bone score is unaffected by anti-resorptive treatment: a registry-based cohort study. J Clin Densitom 26:10–15. https://doi.org/10.1016/j.jocd.2023.01.001
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
This study was approved by the Local Ethics in Research Committee of the São Paulo University School of Medicine (CAPPesq: 51178115.1.0000.0068).
Consent to participate/publication
All participants gave written informed consent.
Conflicts of interest
FFS, GRM, ACMR, AYS, CPF, LMTG, VFC, RMRP have nothing to declare. KRB has received a speaker honorarium from Abbvie, Pfizer and Novartis. DSD has received a speaker honorarium from Amgen and Janssen.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
In memorian of Rosa M. R. Pereira.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Silva, F.F., Machado, G.R., Ribeiro, A.C.M. et al. Damaged bone microarchitecture by Trabecular Bone Score (TBS) and low appendicular muscle mass: main risk factors for vertebral and non-vertebral fractures in women with long-standing rheumatoid arthritis. Osteoporos Int 35, 819–830 (2024). https://doi.org/10.1007/s00198-024-07026-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-024-07026-3