Abstract
Summary
We evaluated the influence of two endogenous hormones on bone health in older women. Higher FSH was associated with bone disease, especially in lower estradiol environments. FSH attenuated the relationship between estradiol and bone. This may provide a mechanism through which future clinical research intervenes on bone loss.
Introduction/purpose
Despite preclinical evidence for an inverse association of follicle-stimulating hormone (FSH) and bone mineral density (BMD), no large epidemiologic studies have evaluated the separate and joint influences of FSH and estradiol on bone in postmenopausal women.
Methods
In a cross-sectional study of 675 postmenopausal women, we evaluated associations of serum FSH and dual X-ray absorptiometry (DXA)-classified areal BMD as well as low bone mass or osteoporosis (T-score < − 1.0) of the femoral neck and total hip. We stratified this analysis by serum estradiol (cut at the median). We tested whether FSH mediates the association of estradiol and BMD using the Sobel test.
Results
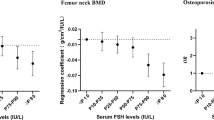
In linear regression models, there was a significant inverse association of serum FSH with both femoral neck and total hip BMD (both p < 0.01) when adjusted for age, hormone therapy (HT) use, and diabetes. In fully adjusted logistic regression models, women in the highest FSH tertile had higher odds of low bone mass/osteoporosis at the femoral neck (OR = 2.98; 95% CI = 1.86–4.77) and at the total hip (OR = 1.74; 95% CI = 1.06–2.84) compared to those in the lowest FSH tertile. We report evidence of effect modification by estradiol in stratified models and an interaction term. FSH met all criteria of a mediator, including an estimated 70% attenuation of the estradiol-BMD relationship (Sobel p value < 0.001).
Conclusions
FSH is associated with higher odds of having low bone mass/osteoporosis even after accounting for HT use. FSH is a mediator of the relationship between estradiol and BMD in healthy postmenopausal women. Larger, prospective studies of FSH concentrations and bone health are needed.


Similar content being viewed by others
References
Wright NC et al (2014) The recent prevalence of osteoporosis and low bone mass in the United States based on bone mineral density at the femoral neck or lumbar spine. J Bone Miner Res 29(11):2520–2526
Shaw ND et al (2010) Estrogen negative feedback on gonadotropin secretion: evidence for a direct pituitary effect in women. J Clin Endocrinol Metab 95(4):1955–1961
Faddy MJ et al (1992) Accelerated disappearance of ovarian follicles in mid-life: implications for forecasting menopause. Hum Reprod 7(10):1342–1346
Sun L et al (2010) Further evidence for direct pro-resorptive actions of FSH. Biochem Biophys Res Commun 394(1):6–11
Robinson LJ et al (2010) FSH-receptor isoforms and FSH-dependent gene transcription in human monocytes and osteoclasts. Biochem Biophys Res Commun 394(1):12–17
Ritter V et al (2008) Follicle-stimulating hormone does not impact male bone mass in vivo or human male osteoclasts in vitro. Calcif Tissue Int 82(5):383–391
Veldhuis-Vlug AG et al (2021) Serum FSH is associated with BMD, bone marrow adiposity, and body composition in the AGES-Reykjavik study of older adults. J Clin Endocrinol Metab 106(3):e1156–e1169
Wang J et al (2015) Follicle-stimulating hormone increases the risk of postmenopausal osteoporosis by stimulating osteoclast differentiation. PLoS ONE 10(8):e0134986
Gourlay ML et al (2011) Follicle-stimulating hormone and bioavailable estradiol are less important than weight and race in determining bone density in younger postmenopausal women. Osteoporos Int 22(10):2699–2708
Gourlay ML et al (2012) Follicle-stimulating hormone is independently associated with lean mass but not BMD in younger postmenopausal women. Bone 50(1):311–316
Wactawski-Wende J et al (2005) The association between osteoporosis and alveolar crestal height in postmenopausal women. J Periodontol 76(11 Suppl):2116–2124
Anderson GL et al (2003) Implementation of the Women’s Health Initiative study design. Ann Epidemiol 13(9):S5–S17
Looker AC et al (1995) Prevalence of low femoral bone density in older U.S. women from NHANES III. J Bone Miner Res 10(5):796–802
Preacher KJ, Leonardelli GJ (2022) Calculation for the Sobel test: An interactive calculation tool for mediation tests. Quantpsy. http://quantpsy.org/sobel/sobel.htm
McCauley LK, Tozum TF, Rosol TJ (2002) Estrogen receptors in skeletal metabolism: lessons from genetically modified models of receptor function. Crit Rev Eukaryot Gene Expr 12(2):89–100
Charlesworth MC, Schwartz NB (1986) Estrogen inhibition of LH and FSH secretion: effects of a GnRH antagonist. Am J Physiol 250(4 Pt 1):E341–E345
Windahl SH, Andersson G, Gustafsson JA (2002) Elucidation of estrogen receptor function in bone with the use of mouse models. Trends Endocrinol Metab 13(5):195–200
Kawai H, Furuhashi M, Suganuma N (2004) Serum follicle-stimulating hormone level is a predictor of bone mineral density in patients with hormone replacement therapy. Arch Gynecol Obstet 269(3):192–195
Randolph JF Jr et al (2004) Change in estradiol and follicle-stimulating hormone across the early menopausal transition: effects of ethnicity and age. J Clin Endocrinol Metab 89(4):1555–1561
Sowers MR et al (2006) Hormone predictors of bone mineral density changes during the menopausal transition. J Clin Endocrinol Metab 91(4):1261–1267
Ebeling PR et al (1996) Bone turnover markers and bone density across the menopausal transition. J Clin Endocrinol Metab 81(9):3366–3371
Iqbal J et al (2006) Follicle-stimulating hormone stimulates TNF production from immune cells to enhance osteoblast and osteoclast formation. Proc Natl Acad Sci USA 103(40):14925–14930
Cannon JG et al (2010) Follicle-stimulating hormone, interleukin-1, and bone density in adult women. Am J Physiol Regul Integr Comp Physiol 298(3):R790–R798
Zhu LL et al (2012) Blocking antibody to the beta-subunit of FSH prevents bone loss by inhibiting bone resorption and stimulating bone synthesis. Proc Natl Acad Sci USA 109(36):14574–14579
Drake MT et al (2010) Effects of suppression of follicle-stimulating hormone secretion on bone resorption markers in postmenopausal women. J Clin Endocrinol Metab 95(11):5063–5068
Rosen HN et al (1998) Utility of biochemical markers of bone turnover in the follow-up of patients treated with bisphosphonates. Calcif Tissue Int 63(5):363–368
Hannon R et al (1998) Response of biochemical markers of bone turnover to hormone replacement therapy: impact of biological variability. J Bone Miner Res 13(7):1124–1133
Crandall CJ, Vasan S, LaCroix A, LeBoff MS, Cauley JA, Robbins JA, Jackson RD, Bauer DC (2018) Bone turnover markers are not associated with hip fracture risk: a case-control study in the women’s health initiative. J Bone Miner Res 33(7):1199–1208. https://doi.org/10.1002/jbmr.3471
Eastell R, Hannon R (2007) Chapter 27—biochemical markers of bone turnover. Treatment of the postmenopausal woman, 3rd ed.; Lobo, RA, Ed, pp. 337–349.
Liu Z et al (2019) A meta-analysis of serum osteocalcin level in postmenopausal osteoporotic women compared to controls. BMC Musculoskelet Disord 20(1):532
Zaidi M et al (2016) Pituitary-bone connection in skeletal regulation. Horm Mol Biol Clin Investig 28(2):85–94
Shieh A et al (2019) Estradiol and follicle-stimulating hormone as predictors of onset of menopause transition-related bone loss in pre- and perimenopausal women. J Bone Miner Res 34(12):2246–2253
Crandall CJ et al (2013) Serum sex steroid levels and longitudinal changes in bone density in relation to the final menstrual period. J Clin Endocrinol Metab 98(4):E654–E663
Funding
This work was supported by the National Ruth L Kirschstein National Research Service Award Predoctoral Fellowship 1F31AG071241-01. Additional support was provided by the Breast Cancer Research and Education Fund through New York State Department of Health Contract #C34926GG. Additional grants include NHLBI-CSB-WH-2016–01-CM, DOD #OS950077, and NIH R01 DE13505. The WHI program is funded by the National Heart, Lung, and Blood Institute through contracts HHSN268201600018C, HHSN268201600001C, HHSN268201600002C, HHSN268201600003C, and HHSN268201600004C. The WHI program is also funded by the National Heart, Lung, and Blood Institute through 75N92021D00001, 75N92021D00002, 75N92021D00003, 75N92021D00004, and 75N92021D00005.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
None.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mattick, L.J., Bea, J.W., Hovey, K.M. et al. Follicle-stimulating hormone is associated with low bone mass in postmenopausal women. Osteoporos Int 34, 693–701 (2023). https://doi.org/10.1007/s00198-023-06676-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-023-06676-z