Abstract
Summary
In clinical practice, it was found that some patients experienced short-term recurrent pain (SRP) in the original site after PVP treatment. This study was designed to develop and validate a nomogram for predicting the potential risks of SRP after PVP, which may help to provide a painless postoperative experience and personalized health management for patients with OVCF.
Introduction
With the aging of China’s population, the incidence of osteoporotic vertebral compression fractures (OVCF) has increased significantly. Percutaneous vertebroplasty (PVP) has been widely accepted due to its minimally invasive, rapid, and effective characteristics. However, it has been found that some patients have short-term recurrent pain (SRP) in the original site after surgery in practical clinical work.
Methods
We retrospectively reviewed the clinical data of OVCF patients who were treated with PVP in our center from January 1st, 2019, to December 30th, 2020. A total of 296 patients were enrolled in the study cohort, and patients were randomly divided into the training set (70%) and validation set (30%). Univariate and multivariate logistic regression analyses were used to determine the risk factors of SRP, and a nomogram predictive model was established accordingly. The model was evaluated by calibration curve, receiver operation characteristic (ROC) curve, and decision curve analysis (DCA).
Results
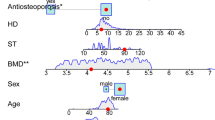
Among the 296 patients, 83 (27.85%) patients experienced SRP after surgery. The independent risk factors included fracture segments (OR: 14.148, 95%CI: 1.532–130.661; p < 0.019), number of surgical vertebrae (OR: 7.896, 95%CI: 3.007–20.729; p < 0.001; (OR: 12.563, 95%CI: 2.223–70.993; p = 0.004), and smoking (OR: 3.833, 95%CI: 1.219–12.052; p = 0.022). The AUC of the prediction model was 0.819 in the training set and 0.794 in the validation set. The calibration curve and DCA indicated the good performance of this nomogram.
Conclusion
The nomogram prediction model had satisfactory accuracy and clinical utility, which may benefit clinical decision-making for the treatment of OVCF and strengthen patient education.





Similar content being viewed by others
References
Alejandro P, Constantinescu F (2018) A review of osteoporosis in the older adult: an update. Rheum Dis Clin North Am 44(3):437–451
Hernlund E et al (2013) Osteoporosis in the European Union: medical management, epidemiology and economic burden. A report prepared in collaboration with the International Osteoporosis Foundation (IOF) and the European Federation of Pharmaceutical Industry Associations (EFPIA). Arch Osteoporos 8(1):136
Zeng Q et al (2019) The prevalence of osteoporosis in China, a nationwide, multicenter DXA survey. J Bone Miner Res 34(10):1789–1797
Lamy O, Uebelhart B, Aubry-Rozier B (2014) Risks and benefits of percutaneous vertebroplasty or kyphoplasty in the management of osteoporotic vertebral fractures. Osteoporos Int 25(3):807–819
Van der Klift M et al (2002) The incidence of vertebral fractures in men and women: the Rotterdam study. J Bone Miner Res 17(6):1051–1056
Klotzbuecher CM et al (2000) Patients with prior fractures have an increased risk of future fractures: a summary of the literature and statistical synthesis. J Bone Miner Res 15(4):721–739
Galibert P et al (1987) Preliminary note on the treatment of vertebral angioma by percutaneous acrylic vertebroplasty. Neurochirurgie 33(2):166–168
Clark W et al (2016) Safety and efficacy of vertebroplasty for acute painful osteoporotic fractures (VAPOUR): a multicentre, randomised, double-blind, placebo-controlled trial. Lancet 388(10052):1408–1416
Klazen CA et al (2010) Vertebroplasty versus conservative treatment in acute osteoporotic vertebral compression fractures (Vertos II): an open-label randomised trial. Lancet 376(9746):1085–1092
Yuan WH, Hsu HC, Lai KL (2016) Vertebroplasty and balloon kyphoplasty versus conservative treatment for osteoporotic vertebral compression fractures: a meta-analysis. Medicine (Baltimore) 95(31):e4491
Manchikanti L et al (2009) Comprehensive review of epidemiology, scope, and impact of spinal pain. Pain Physician 12(4):E35-70
Akça O et al (1999) Postoperative pain and subcutaneous oxygen tension. Lancet 354(9172):41–42
Beattie WS, Buckley DN, Forrest JB (1993) Epidural morphine reduces the risk of postoperative myocardial ischaemia in patients with cardiac risk factors. Can J Anaesth 40(6):532–541
McCarthy J, Davis A (2016) Diagnosis and management of vertebral compression fractures. Am Fam Physician 94(1):44–50
Balachandran VP et al (2015) Nomograms in oncology: more than meets the eye. Lancet Oncol 16(4):e173–e180
Sternberg CN (2006) Are nomograms better than currently available stage groupings for bladder cancer? J Clin Oncol 24(24):3819–3820
Choi SH et al (2020) Incidence and management trends of osteoporotic vertebral compression fractures in South Korea: a nationwide population-based study. Asian Spine J 14(2):220–228
Burge R et al (2007) Incidence and economic burden of osteoporosis-related fractures in the United States, 2005–2025. J Bone Miner Res 22(3):465–475
Lange A, Zeidler J, Braun S (2014) One-year disease-related health care costs of incident vertebral fractures in osteoporotic patients. Osteoporos Int 25(10):2435–2443
Hopkins RB et al (2016) The current economic burden of illness of osteoporosis in Canada. Osteoporos Int 27(10):3023–3032
Qu B et al (2014) The economic burden of fracture patients with osteoporosis in western China. Osteoporos Int 25(7):1853–1860
Liu R et al (2018) Incidence and risk factors of medical complications and direct medical costs after osteoporotic fracture among patients in China. Arch Osteoporos 13(1):12
Belkoff SM et al (2001) The biomechanics of vertebroplasty. The effect of cement volume on mechanical behavior. Spine (Phila Pa 1976) 26(14):1537–41
Chen Z et al (2019) Risk factors of secondary vertebral compression fracture after percutaneous vertebroplasty or kyphoplasty: a retrospective study of 650 patients. Med Sci Monit 25:9255–9261
Hsieh MK et al (2019) Risk factors of neurological deficit and pulmonary cement embolism after percutaneous vertebroplasty. J Orthop Surg Res 14(1):406
van Ransbeeck A et al (2018) Pain assessment discrepancies: a cross-sectional study highlights the amount of underrated pain. Pain Pract 18(3):360–367
Brinck EC et al (2018) Perioperative intravenous ketamine for acute postoperative pain in adults. Cochrane Database Syst Rev 12(12):Cd012033
Zhang T-Y, Zhang P-X, Xue F, Zhang D-Y, Jiang B-G (2020) Risk factors for cement leakage and nomogram for predicting the intradiscal cement leakage after the vertebra augmented surgery. BMC Musculoskelet Disord 21(1):792. https://doi.org/10.1186/s12891-020-03810-4
Layton KF et al (2007) Vertebroplasty, first 1000 levels of a single center: evaluation of the outcomes and complications. AJNR Am J Neuroradiol 28(4):683–689
Lin CC et al (2009) The clinical influence of fluid sign in treated vertebral bodies after percutaneous vertebroplasty. Radiology 251(3):866–872
Grados F et al (2000) Long-term observations of vertebral osteoporotic fractures treated by percutaneous vertebroplasty. Rheumatology (Oxford) 39(12):1410–1414
Lane JI et al (2002) Intravertebral clefts opacified during vertebroplasty: pathogenesis, technical implications, and prognostic significance. AJNR Am J Neuroradiol 23(10):1642–1646
Yang W, Yang J, Liang M (2019) Percutaneous vertebroplasty does not increase the incidence of new fractures in adjacent and nonadjacent vertebral bodies. Clin Spine Surg 32(2):E99-e106
Lindsay R et al (2001) Risk of new vertebral fracture in the year following a fracture. JAMA 285(3):320–323
Ross PD et al (1991) Pre-existing fractures and bone mass predict vertebral fracture incidence in women. Ann Intern Med 114(11):919–923
Kim SH et al (2004) Risk factors of new compression fractures in adjacent vertebrae after percutaneous vertebroplasty. Acta Radiol 45(4):440–445
Tanigawa N et al (2006) Radiological follow-up of new compression fractures following percutaneous vertebroplasty. Cardiovasc Intervent Radiol 29(1):92–96
Mathis JM (2003) Percutaneous vertebroplasty: complication avoidance and technique optimization. AJNR Am J Neuroradiol 24(8):1697–1706
Hoffmann RT et al (2007) Vertebroplasty in the treatment of osteoporotic vertebral body fracture. Eur Radiol 17(10):2656–2662
Abbasi Fard S et al (2017) Instability in thoracolumbar trauma: is a new definition warranted? Clin Spine Surg 30(8):E1046-e1049
Schroeder GD et al (2015) Can a thoracolumbar injury severity score be uniformly applied from T1 to L5 or are modifications necessary? Global Spine J 5(4):339–345
Cauley JA et al (2007) Long-term risk of incident vertebral fractures. JAMA 298(23):2761–2767
Ditre JW et al (2011) Pain, nicotine, and smoking: research findings and mechanistic considerations. Psychol Bull 137(6):1065–1093
Khor S et al (2018) Development and validation of a prediction model for pain and functional outcomes after lumbar spine surgery. JAMA Surg 153(7):634–642
Shen L et al (2018) Decreased pain tolerance before surgery and increased postoperative narcotic requirements in abstinent tobacco smokers. Addict Behav 78:9–14
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Zongtai Liu, Xiaohui Zhang, Haiyan Liu, and Dalin Wang declare that they have no conflict of interest.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Liu, Z., Zhang, X., Liu, H. et al. A nomogram for short-term recurrent pain after percutaneous vertebroplasty for osteoporotic vertebral compression fractures. Osteoporos Int 33, 851–860 (2022). https://doi.org/10.1007/s00198-021-06232-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-021-06232-7