Abstract
Summary
Calcitonin may relieve pain by modulating central serotonin activity. Calcitonin partly reversed the hypersensitivity to pain induced by ovariectomy. This suggests that the anti-nociceptive effects of calcitonin in the treatment of osteoporosis may be mediated by alterations in neural serotonin transporter (SERT) activity.
Introduction
This study used a rat model of osteoporosis to evaluate the role of the cerebral serotonin system in the anti-nociceptive effect of calcitonin, a drug used to treat post-menopausal osteoporosis.
Methods
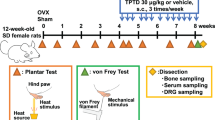
Osteoporosis was induced in rats by ovariectomy (OVX). Rats were then randomized to the following four groups: sham operation, OVX, OVX plus calcitonin, or OVX plus alendronate.
Results
OVX led to alterations in bone micro-architecture; alendronate strongly reversed this effect, and calcitonin moderately reversed this effect. OVX increased hyperalgesia (determined as the time for hind paw withdrawal from a heat source); calcitonin reduced this effect, but alendronate had no effect. OVX increased the expression of c-Fos (a neuronal marker of pain) in the thalamus; calcitonin strongly reversed this effect, and alendronate moderately reversed this effect. OVX also reduced SERT but increased 5-HT1A receptor expression and activity; calcitonin aggravated this effect, but alendronate had no effect on recovery of SERT/5-HT1A activity and expression.
Conclusions
Our study of a rat model of osteoporosis suggests that OVX-induced enhancement of the serotonergic system may protect against hyperalgesia. However, the anti-nociceptive effects of calcitonin in osteoporosis may be mediated by decreased neural SERT activity and increased activation of 5-HT1 receptors in the thalamus.







Similar content being viewed by others
References
Diab DL, Watts NB (2013) Postmenopausal osteoporosis. Curr Opin Endocrinol Diabetes Obes 20:501–509
Tella SH, Gallagher JC (2014) Prevention and treatment of postmenopausal osteoporosis. J Steroid Biochem Mol Biol 142:155–170
Karsdal MA, Henriksen K, Arnold M, Christiansen C (2008) Calcitonin: a drug of the past or for the future? Physiologic inhibition of bone resorption while sustaining osteoclast numbers improves bone quality. BioDrugs 22:137–144
Takayama B, Kikuchi S, Konno S, Sekiguchi M (2008) An immunohistochemical study of the antinociceptive effect of calcitonin in ovariectomized rats. BMC Musculoskelet Disord 9:164
Iwamoto J, Makita K, Sato Y, Takeda T, Matsumoto H (2011) Alendronate is more effective than elcatonin in improving pain and quality of life in postmenopausal women with osteoporosis. Osteoporos Int 22:2735–2742
Fujita T, Ohue M, Nakajima M, Fujii Y, Miyauchi A, Takagi Y (2011) Comparison of the effects of elcatonin and risedronate on back and knee pain by electroalgometry using fall of skin impedance and quality of life assessment using SF-36. J Bone Miner Metab 29:588–597
Gennari C (2002) Analgesic effect of calcitonin in osteoporosis. Bone 30:67S–70S
Lyritis GP, Trovas G (2002) Analgesic effects of calcitonin. Bone 30:71S–74S
Knopp-Sihota JA, Newburn-Cook CV, Homik J, Cummings GG, Voaklander D (2012) Calcitonin for treating acute and chronic pain of recent and remote osteoporotic vertebral compression fractures: a systematic review and meta-analysis. Osteoporos Int 23:17–38
Knopp JA, Diner BM, Blitz M, Lyritis GP, Rowe BH (2005) Calcitonin for treating acute pain of osteoporotic vertebral compression fractures: a systematic review of randomized, controlled trials. Osteoporos Int 16:1281–1290
Gobelet C, Meier JL, Schaffner W, Bischof-Delaloye A, Gerster JC, Burckhardt P (1986) Calcitonin and reflex sympathetic dystrophy syndrome. Clin Rheumatol 5:382–388
Hamamci N, Dursun E, Ural C, Cakci A (1996) Calcitonin treatment in reflex sympathetic dystrophy: a preliminary study. Br J Clin Pract 50:373–375
Matayoshi S, Shimodozono M, Hirata Y, Ueda T, Horio S, Kawahira K (2009) Use of calcitonin to prevent complex regional pain syndrome type I in severe hemiplegic patients after stroke. Disabil Rehabil 31:1773–1779
Perez RS, Kwakkel G, Zuurmond WW, de Lange JJ (2001) Treatment of reflex sympathetic dystrophy (CRPS type 1): a research synthesis of 21 randomized clinical trials. J Pain Symptom Manag 21:511–526
Quatraro A, Minei A, De Rosa N, Giugliano D (1992) Calcitonin in painful diabetic neuropathy. Lancet 339:746–747
Visser EJ, Kwei PL (2006) Salmon calcitonin in the treatment of post herpetic neuralgia. Anaesth Intensive Care 34:668–671
Zieleniewski W (1990) Calcitonin nasal spray for painful diabetic neuropathy. Lancet 336:449
Azria M (2002) Possible mechanisms of the analgesic action of calcitonin. Bone 30:80S–83S
Davey RA, Findlay DM (2013) Calcitonin: physiology or fantasy? J Bone Miner Res 28:973–979
Guidobono F, Netti C, Sibilia V, Olgiati VR, Pecile A (1985) Role of catecholamines in calcitonin-induced analgesia. Pharmacology 31:342–348
Maeda Y, Yamada K, Hasegawa T, Nabeshima T (1995) Neuronal mechanism of the inhibitory effect of calcitonin on N-methyl-D-aspartate-induced aversive behavior. Eur J Pharmacol 275:163–170
Meisenberg G, Simmons WH (1983) Minireview. Peptides and the blood–brain barrier. Life Sci 32:2611–23
Pecile A (1992) Calcitonin and relief of pain. Bone Miner 16:187–189
Sibilia VF, Pagani N, Lattuada D, Rapetti F, Guidobono NC (2000) Amylin compared with calcitonin: competitive binding studies in rat brain and antinociceptive activity. Brain Res 854:79–84
Yoshimura M (2000) Analgesic mechanism of calcitonin. J Bone Miner Metab 18:230–233
Viguier F, Michot B, Hamon M, Bourgoin S (2013) Multiple roles of serotonin in pain control mechanisms—implications of 5-HT(7) and other 5-HT receptor types. Eur J Pharmacol 716:8–16
Craft RM (2007) Modulation of pain by estrogens. Pain 132(Suppl 1):S3–12
Martin VT (2009) Ovarian hormones and pain response: a review of clinical and basic science studies. Gend Med 6(Suppl 2):168–192
Ito A, Takeda M, Yoshimura T, Komatsu T, Ohno T, Kuriyama H, Matsuda A, Yoshimura M (2012) Anti-hyperalgesic effects of calcitonin on neuropathic pain interacting with its peripheral receptors. Mol Pain 8:42
Kharode YP, Sharp MC, Bodine PV (2008) Utility of the ovariectomized rat as a model for human osteoporosis in drug discovery. Methods Mol Biol 455:111–124
Zheng XF, Li B, Zhang YH, Yang YH, Meng XY, Jiang SD, Jiang LS (2013) Blockade of substance P receptor attenuates osteoporotic pain, but not bone loss, in ovariectomized mice. Menopause 20:1074–1083
Egermann M, Goldhahn J, Schneider E (2005) Animal models for fracture treatment in osteoporosis. Osteoporos Int 16(Suppl 2):S129–138
Lelovas PP, Xanthos TT, Thoma SE, Lyritis GP, Dontas IA (2008) The laboratory rat as an animal model for osteoporosis research. Comp Med 58:424–430
Kuo YJ, Tsuang FY, Sun JS, Lin CH, Chen CH, Li JY, Huang YC, Chen WY, Yeh CB, Shyu JF (2012) Calcitonin inhibits SDCP-induced osteoclast apoptosis and increases its efficacy in a rat model of osteoporosis. PLoS One 7:e40272
Guy JA, Shea M, Peter CP, Morrissey R, Hayes WC (1993) Continuous alendronate treatment throughout growth, maturation, and aging in the rat results in increases in bone mass and mechanical properties. Calcif Tissue Int 53:283–8
Coderre TJ, Fundytus ME, McKenna JE, Dalal S, Melzack R (1993) The formalin test: a validation of the weighted-scores method of behavioural pain rating. Pain 54:43–50
Hargreaves K, Dubner R, Brown F, Flores C, Joris J (1988) A new and sensitive method for measuring thermal nociception in cutaneous hyperalgesia. Pain 32:77–88
Paxinos G, Watson C (2007) The rat brain in stereotaxic coordinates, 6th edn. Academic Press, Burlington, MA, USA, p 208
Muller R, Van Campenhout H, Van Damme B, Van Der Perre G, Dequeker J, Hildebrand T, Ruegsegger P (1998) Morphometric analysis of human bone biopsies: a quantitative structural comparison of histological sections and micro-computed tomography. Bone 23:59–66
Abate G, Taormina F, Brillante C, Fraccalaglio L (1994) The effects of the carbocalcitonin + arginine-lysine-lactose combination in senile involutional osteoporosis. Minerva Med 85:253–259
Descarries L, Riad M (2012) Effects of the antidepressant fluoxetine on the subcellular localization of 5-HT1A receptors and SERT. Philos Trans R Soc Lond B Biol Sci 367:2416–2425
Hirsch PF, Lester GE, Talmage RV (2001) Calcitonin, an enigmatic hormone: does it have a function? J Musculoskelet Nueronal Interact 1:299–305
Overman RA, Borse M, Gourlay ML (2013) Salmon calcitonin use and associated cancer risk. Ann Pharmacother 47:1675–1684
Purdue BW, Tilakaratne N, Sexton PM (2002) Molecular pharmacology of the calcitonin receptor. Recept Channels 8:243–255
Acknowledgments
This research was partly supported by research grants to J.-F. Shyu from the National Defense Medical Center (MAB-102-56), the Tri-Service General Hospital (TSGH-C102-124), and the Ministry of Science and Technology (MOST 104-2320-B-016-008) of Taiwan.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
None.
Rights and permissions
About this article
Cite this article
Yeh, CB., Weng, SJ., Chang, KW. et al. Calcitonin alleviates hyperalgesia in osteoporotic rats by modulating serotonin transporter activity. Osteoporos Int 27, 3355–3364 (2016). https://doi.org/10.1007/s00198-016-3652-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-016-3652-1