Abstract
Summary
Little data exist on the frequency of fracture among oral glucocorticoid users. We examined the effect of oral glucocorticoids on fracture incidence using data from randomized controlled trials. Patients starting glucocorticoids had a higher probability of fracture and decline in bone mineral density compared to chronic glucocorticoid users.
Introduction
Oral glucocorticoids (GCs) are the leading cause of secondary osteoporosis. However, there have been few studies that quantify the rate of fracture among GC users. We sought to provide a pooled estimate of fracture risk from randomized controlled trials (RCTs) of GC-treated patients.
Methods
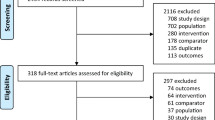
We updated a MEDLINE search published by the American College of Rheumatology through to March 2015 and identified RCTs of osteoporosis therapies that reported fracture and bone mineral density (BMD) among oral GC users. We restricted the analysis to placebo or control arms. RCT arms were stratified by GC exposure at enrolment to GC initiators (≤6 months) and chronic GC users (>6 months). Bayesian meta-regression was used to estimate the annual probability of vertebral fracture (primary), non-vertebral fracture and percentage change in lumbar spine and femoral neck BMD.
Results
The annual incidence of vertebral and non-vertebral fracture was 5.1 % (95 % CrI = 2.8–8.2) and 2.5 % (95 % CrI = 1.2–-4.2) among GC initiators, and 3.2 % (95 % CrI = 1.8–5.0) and 3.0 % (95 % CrI = 0.8–5.9) among chronic GC users. Our meta-regression identified a non-significant effect of group-level variables (mean age, mean BMD, mean GC daily dose, patients with previous vertebral fractures, proportion of women and adjuvant used) on vertebral fracture rate.
Conclusion
Our study found higher vertebral fracture incidence among GC initiators, yet a relative decline in fracture incidence with longer exposure. Our findings suggest that fracture incidence among oral GC users may be more common than previously estimated. Optimizing GC-induced osteoporosis management during early exposure to GC is essential to prevent fractures.



Similar content being viewed by others
Abbreviations
- GC:
-
Glucocorticoid
- BMD:
-
Bone mineral density
- RCT:
-
Randomized controlled trial
- MCMC:
-
Markov Chain Monte Carlo
- CrI:
-
Credible interval
References
Van Staa TP (2006) The pathogenesis, epidemiology and management of glucocorticoid-induced osteoporosis. Calcif Tissue Int 79:129–137
Grossman JM, Gordon R, Ranganath VK et al (2010) American College of Rheumatology 2010 recommendations for the prevention and treatment of glucocorticoid-induced osteoporosis. Arthritis Care Res 62:1515–1526
Nawata H, Soen S, Takayanagi R et al (2005) Guidelines on the management and treatment of glucocorticoid-induced osteoporosis of the Japanese Society for Bone and Mineral Research (2004). J Bone Miner Metab 23:105–109
Devogelaer J-P, Goemaere S, Boonen S et al (2006) Evidence-based guidelines for the prevention and treatment of glucocorticoid-induced osteoporosis: a consensus document of the Belgian bone club. Osteoporos Int 17:8–19
Gourlay M, Franceschini N, Sheyn Y (2007) Prevention and treatment strategies for glucocorticoid-induced osteoporotic fractures. Clin Rheumatol 26:144–153
Papaioannou A, Morin S, Cheung AM et al (2010) 2010 clinical practice guidelines for the diagnosis and management of osteoporosis in Canada: summary. CMAJ 182:1864–1873
Lekamwasam S, Adachi JD, Agnusdei D et al (2012) A framework for the development of guidelines for the management of glucocorticoid-induced osteoporosis. Osteoporos Int 23:2257–2276
de Nijs RNJ, Jacobs JWG, Lems WF et al (2006) Alendronate or alfacalcidol in glucocorticoid-induced osteoporosis. NEJM 355:675–684
Ismail AA, Pye SR, Cockerill WC et al (2002) Incidence of limb fracture across Europe: results from the European prospective osteoporosis study (Epos). Osteoporos Int 13:565–571
Adachi JD, Saag KG, Delmas PD et al (2001) Two-year effects of alendronate on bone mineral density and vertebral fracture in patients receiving glucocorticoids: a randomized, double-blind, placebo-controlled extension trial. Arthritis Rheum 44:202–211
Jenkins EA, Walker-Bone KE, Wood A et al (1999) The prevention of corticosteroid-induced bone loss with intermittent cyclical etidronate. Scand J Rheumatol 28:152–156
Cortet B, Hachulla E, Barton I, Bonvoisin B, Roux C (1999) Evaluation of the efficacy of etidronate therapy in preventing glucocorticoid-induced bone loss in patients with inflammatory rheumatic diseases. A randomized study. Rev Rhum Engl Ed 66:214–219
Cohen S, Levy RM, Keller M et al (1999) Risedronate therapy prevents corticosteroid-induced bone loss: a twelve-month, multicenter, randomized, double-blind, placebo-controlled, parallel-group study. Arthritis Rheum 42:2309–2318
Roux C, Oriente P, Laan R et al (1998) Randomized trial of effect of cyclical etidronate in the prevention of corticosteroid-induced bone loss. Ciblos study group. J Clin Endocrinol Metab 83:1128–1133
van Staa TP, Leufkens HG, Cooper C (2002) The epidemiology of corticosteroid-induced osteoporosis: a meta-analysis. Osteoporos Int 13:777–787
Kanis JA, Johansson H, Oden A et al (2004) A meta-analysis of prior corticosteroid use and fracture risk. J Bone Miner Res 19:893–899
Canalis E, Mazziotti G, Giustina A, Bilezikian JP (2007) Glucocorticoid-induced osteoporosis: pathophysiology and therapy. Osteoporos Int 18:1319–1328
den Uyl D, Bultink IE, Lems WF (2011) Advances in glucocorticoid-induced osteoporosis. Curr Rheumatol Rep 13:233–240
Dore RK (2013) Long-term safety, efficacy, and patient acceptability of teriparatide in the management of glucocorticoid-induced osteoporosis. Patient Prefer Adherence 7:435–446
Panday K, Gona A, Humphrey MB (2014) Medication-induced osteoporosis: screening and treatment strategies. Ther Adv Musculoskelet Disord 6:185–202
Angeli A, Guglielmi G, Dovio A et al (2006) High prevalence of asymptomatic vertebral fractures in post-menopausal women receiving chronic glucocorticoid therapy: a cross-sectional outpatient study. Bone 39:253–259
Amin S, Lavalley MP, Simms RW, Felson DT (2002) The comparative efficacy of drug therapies used for the management of corticosteroid-induced osteoporosis: a meta-regression. J Bone Miner Res 17:1512–1526
Homik JE, Cranney A, Shea B et al (1999) A meta-analysis on the use of bisphosphonates in corticosteroid induced osteoporosis. J Rheumatol 26:1148–1157
Rizzoli R, Biver E (2015) Glucocorticoid-induced osteoporosis: who to treat with what agent? Nat Rev Rheumatol 11:98–109
Seibel MJ, Cooper MS, Zhou H (2013) Glucocorticoid-induced osteoporosis: mechanisms, management, and future perspectives. Lancet Diabetes Endocrinol 1:59–70
Sutton AJ, Abrams KR (2001) Bayesian methods in meta-analysis and evidence synthesis. Stat Methods Med Res 10:277–303
Dias S, Welton NJ, Sutton AJ, Ades AE (2013) Evidence synthesis for decision making 5: the baseline natural history model. Med Decis Mak 33:657–670
Hamra G, MacLehose R, Richardson D (2013) Markov chain Monte Carlo: an introduction for epidemiologists. Int J Epidemiol 42:627–634
Turner RM, Davey J, Clarke MJ, Thompson SG, Higgins JP (2012) Predicting the extent of heterogeneity in meta-analysis, using empirical data from the Cochrane database of systematic reviews. Int J Epidemiol 41:818–827
van Staa TP, Leufkens HG, Abenhaim L, Zhang B, Cooper C (2000) Use of oral corticosteroids and risk of fractures. J Bone Miner Res 15:993–1000
Kalpakcioglu BB, Engelke K, Genant HK (2011) Advanced imaging assessment of bone fragility in glucocorticoid-induced osteoporosis. Bone 48:1221–1231
Rüegsegger P, Medici TC, Anliker M (1983) Corticosteroid-induced bone loss. A longitudinal study of alternate day therapy in patients with bronchial asthma using quantitative computed tomography. Eur J Clin Pharmacol 25:615–620
Albaum JM, Lévesque LE, Gershon AS, Liu G, Cadarette SM (2015) Glucocorticoid-induced osteoporosis management among seniors, by year, sex, and indication, 1996–2012. Osteoporos Int 26:2845–2852
Majumdar SR, Lix LM, Morin SN et al (2013) The disconnect between better quality of glucocorticoid-induced osteoporosis preventive care and better outcomes: a population-based cohort study. J Rheumatol 40:1736–1741
LoCascio V, Bonucci E, Imbimbo B et al (1990) Bone loss in response to long-term glucocorticoid therapy. Bone Miner 8:39–51
Reid DM, Devogelaer JP, Saag K et al (2009) Zoledronic acid and risedronate in the prevention and treatment of glucocorticoid-induced osteoporosis (horizon): a multicentre, double-blind, double-dummy, randomised controlled trial. Lancet 373:1253–1263
Ringe JD, Dorst A, Faber H, Schacht E, Rahlfs VW (2004) Superiority of alfacalcidol over plain vitamin D in the treatment of glucocorticoid-induced osteoporosis. Rheumatol Int 24:63–70
Teramukai S, Matsuyama Y, Mizuno S, Sakamoto J (2004) Individual patient-level and study-level meta-analysis for investigating modifiers of treatment effect. Jpn J Clin Oncol 34:717–721
Dias S, Sutton AJ, Welton NJ, Ades AE (2013) Evidence synthesis for decision making 3: heterogeneity--subgroups, meta-regression, bias, and bias-adjustment. Med Decis Mak 33:618–640
van Houwelingen HC, Arends LR, Stijnen T (2002) Advanced methods in meta-analysis: multivariate approach and meta-regression. Stat Med 21:589–624
Acknowledgments
MAA is supported by the CAnadian Network for Advanced Interdisciplinary Methods for Comparative Effectiveness Research (CAN-AIM) training scholarship and the Leslie Dan Faculty of Pharmacy Dean’s Entrance Scholarship and was supported by the Drug Safety and Effectiveness Cross-Disciplinary Training scholarship. The funding agencies did not have any role in the design and conduct of the study; analysis, or interpretation of the data; or preparation, review, or approval of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
JDA reports having participated in clinical trials sponsored by Procter & Gamble, Amgen and by Merck for the prevention and treatment of glucocorticoid-induced osteoporosis. JDA also reports having received consulting fees and research grants from Actavis, Amgen, Eli Lilly, Merck, and Novartis. MAA, JMA, MT, PP, LEL, and SMC state that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM 1
(DOCX 79 kb)
Rights and permissions
About this article
Cite this article
Amiche, M.A., Albaum, J.M., Tadrous, M. et al. Fracture risk in oral glucocorticoid users: a Bayesian meta-regression leveraging control arms of osteoporosis clinical trials. Osteoporos Int 27, 1709–1718 (2016). https://doi.org/10.1007/s00198-015-3455-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-015-3455-9