Abstract
Summary
This population-based case–control analysis investigated the association between osteoporosis and prior urinary calculus (UC) in Taiwan. We succeeded in detecting an association between osteoporosis and prior UC (adjusted odds ratio = 1.66). This association was consistent and significant regardless of stone location.
Introduction
UC has been demonstrated to be a risk factor for osteoporotic fractures, but no studies to date have directly investigated the association between UC and osteoporosis. This case–control analysis aimed to investigate the association of osteoporosis with prior UC using a population-based dataset in Taiwan.
Methods
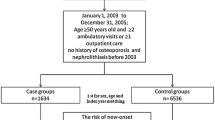
We first identified 39,840 cases ≥40 years who received their first-time diagnosis of osteoporosis between 2002 and 2009 and then randomly selected 79,680 controls. We used conditional logistic regression analyses to compute the odds ratio (OR) and the corresponding 95 % confidence interval (CI) for having been previously diagnosed with UC between cases and controls.
Results
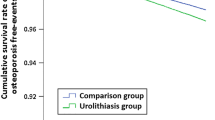
The OR of having been previously diagnosed with UC for patients with osteoporosis was 1.66 (95 % CI = 1.59–1.73) when compared to controls after adjusting for geographic location, urbanization level, type I diabetes mellitus, coronary heart disease, hyperlipidemia, rheumatoid arthritis, stroke, renal disease, Parkinson's disease, hyperthyroidism, chronic hepatopathy, Cushing's syndrome, malabsorption, gastrectomy, obesity, and alcohol abuse/alcohol dependence syndrome. The results consistently showed that osteoporosis was significantly associated with a previous diagnosis of UC regardless of stone location; the adjusted ORs of prior kidney calculus, ureter calculus, bladder calculus, and unspecified calculus when compared to controls were 1.71 (95 % CI = 1.61–1.81), 1.60 (95 % CI = 1.47–1.74), 1.59 (95 % CI = 1.23–2.04), and 1.69 (95 % CI = 1.59–1.80), respectively.
Conclusions
This study succeeded in detecting an association between osteoporosis and prior UC. In addition, our findings were consistent and significant regardless of stone location.
Similar content being viewed by others
References
Long LO, Park S (2007) Update on nephrolithiasis management. Minerva Urol Nefrol 59:317–25
Goldfarb DS (2003) Increasing prevalence of kidney stones in the United States. Kidney Int 63:1951–2
Stamatelou KK, Francis ME, Jones CA et al (2003) Time trends in reported prevalence of kidney stones in the United States: 1976–1994. Kidney Int 63:1817–23
López M, Hoppe B (2010) History, epidemiology and regional diversities of urolithiasis. Pediatr Nephrol 25:49–59
NIH Consensus Development Panel on Osteoporosis Prevention, Diagnosis, and Therapy (2001) Osteoporosis Prevention, Diagnosis, and Therapy. JAMA 285:785–95
Wang Y, Tao Y, Hymen ME, Li J, Chen Y (2009) Osteoporosis in China. Osteoporos Int 20:1651–62
Pande KC (2002) Prevalence of low bone mass in healthy Indian population. J Indian Med Assoc 100:598–602
Tenenhouse A, Joseph L, Kreiger N, Poliquin S, Murray TM, Blondeau L, Berger C, Hanley DA, Prior JC; Canadian Multicentre Osteoporosis Study (CaMos) Research Group (2000) Estimation of the prevalence of low bone density in Canadian women and men using a population-specific DXA reference standard: the Canadian Multicentre Osteoporosis Study (CaMos). Osteoporos Int 11:897–904
Buduneli N, Saygan BH, Karaduman U, Saraç F, Karaduman M, Ayçelik N (2008) Calcium, vitamin D supplements with or without alendronate and supragingival calculus formation in osteoporotic women: a preliminary study. Expert Opin Pharmacother 9:2015–20
World Health Organization (2004) WHO Scientific Group on the assessment of osteoporosis at primary health care level: summary meeting report May 2004. http://who.int/chp/topics/Osteoporosis.pdf
Martínez Pérez JA, Palacios S, García FC, Pérez M (2011) Assessing osteoporosis risk factors in Spanish menopausal women. Gynecol Endocrinol 27:807–13
Sakhaee K (2008) Nephrolithiasis as a systemic disorder. Curr Opin Nephrol Hypertens 17:304–9
Iba A, Kohjimoto Y, Mori T et al (2010) Insulin resistance increases the risk of urinary stone formation in a rat model of metabolic syndrome. BJU Int 106:1550–4
Sakhaee K, Adams-Huet B, Moe OW et al (2002) Pathophysiologic basis for normouricosuric uric acid nephrolithiasis. Kidney Int 62:971–9
Rule AD, Roger VL, Melton LJ 3rd et al (2010) Kidney stones associate with increased risk for myocardial infarction. J Am Soc Nephrol 21:1641–4
Taylor EN, Stampfer MJ, Curhan GC (2005) Diabetes mellitus and the risk of nephrolithiasis. Kidney Int 68:1230–5
Srivastava T, Alon US (2005) Urolithiasis in adolescent children. Adolesc Med Clin 16:87–109
Pacifici R, Rothstein M, Rifas L, Lau KH, Baylink DJ, Avioli LV, Hruska K (1990) Increased monocyte interleukin-1 activity and decreased vertebral bone density in patients with fasting idiopathic hypercalciuria. J Clin Endocrinol Metab 71:138–45
Bilić-Curcić I, Milas-Ahić J, Smolić M, Smolić R, Mihaljević I, Tucak-Zorić S (2009) Urolithiasis and osteoporosis: clinical relevance and therapeutic implications. Coll Antropol 33(Suppl 2):189–92
Sakhaee K, Maalouf NM, Kumar R, Pasch A, Moe OW (2011) Nephrolithiasis-associated bone disease: pathogenesis and treatment options. Kidney Int 79:393–403
Senzaki H, Yasui T, Okada A, Ito Y, Tozawa K, Kohri K (2004) Alendronate inhibits urinary calcium microlith formation in a three-dimensional culture model. Urol Res 32:223–8
Lauderdale DS, Thisted RA, Wen M, Favus MJ (2001) Bone mineral density and fracture among prevalent kidney stone cases in the third National Health and Nutrition Examination Survey. J Bone Miner Res 16:1893–8
Melton LJ 3rd, Crowson CS, Khosla S, Wilson DM, O'Fallon WM (1998) Fracture risk among patients with urolithiasis: a population-based cohort study. Kidney Int 53:459–64
Galsworthy TD (1990) The mechanism of an osteoporosis center. Orthop Clin North Am 21:163–9
Park KJ, Jeon SS, Han DH, Lee SY (2011) Clinical and metabolic evaluation of Korean patients with urolithiasis. Scand J Clin Lab Invest 71:481–5
Wilson DM (1989) Clinical and laboratory approaches for evaluation of nephrolithiasis. J Urol 141:770–4
Incel NA, Gökoğlu F, Nacir B, Incel N (2006) Bone and stone in ankylosing spondylitis: osteoporosis and urolithiasis. Clin Rheumatol 25:667–70
Wu XP, Liao EY, Huang G, Dai RC, Zhang HA (2003) Comparison study of the reference curves of bone mineral density at different skeletal sites in native Chinese, Japanese, and American Caucasian women. Calcif Tissue Int 73:122–32
Levine BS, Rodman JS, Wienerman S, Bockman RS, Lane JM, Chapman DS (1994) Effect of calcium citrate supplementation on urinary calcium oxalate saturation in female stone formers: implications for prevention of osteoporosis. Am J Clin Nutr 60:592–6
Acknowledgments
This study is based in part on data from the National Health Insurance Research Database provided by the Bureau of National Health Insurance, Department of Health, Taiwan and managed by the National Health Research Institutes. The interpretations and conclusions contained herein do not represent those of the Bureau of National Health Insurance, Department of Health, or the National Health Research Institutes.
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
Joseph Keller and Ching-Chun Lin have equal contribution to this manuscript.
Rights and permissions
About this article
Cite this article
Keller, J.J., Lin, CC., Kang, JH. et al. Association between osteoporosis and urinary calculus: evidence from a population-based study. Osteoporos Int 24, 651–657 (2013). https://doi.org/10.1007/s00198-012-2019-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-012-2019-5