Abstract
Summary
We have analysed the association of single-nucleotide polymorphisms (SNPs) in CD40 and CD40L genes with bone mineral density (BMD) in our women. Results showed that women with TT genotype for rs1883832 (CD40) and for rs1126535 (CD40L) SNPs displayed reduced BMD and increased risk for osteopenia/osteoporosis. Our data notwithstanding, the results need to be replicated.
Introduction
Recent data have revealed that the CD40/CD40L system can be implicated in bone metabolism regulation. Moreover, we previously demonstrated that rs1883832 in the CD40 gene was significantly associated with BMD and osteoporosis risk. The objective of the present work was to determine whether polymorphisms in CD40 and CD40L genes are associated with BMD and osteoporosis risk.
Methods
We conducted an association study of BMD values with SNPs in CD40 and CD40L genes in a population of 811 women of which 693 and 711 had femoral neck (FN) and lumbar spine (LS) densitometric studies, respectively.
Results
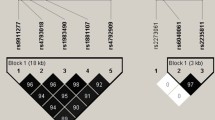
Women with the TT genotype for rs1883832 (CD40) showed a reduction in FN-BMD (P = 0.005) and LS-BMD (P = 0.020) when compared with women with the CC/CT genotype. Moreover, we found that rs1126535 (CD40L) was significantly associated with LS-BMD so that women with the TT genotype displayed lower BMD (P = 0.014) than did women with the CC/CT genotype. Interestingly, we have found a strong interaction between polymorphisms in these genes. Thus, women with the TT genotype for both rs1883832 and rs1126535 SNPs (TT + TT women) showed a lower age-adjusted BMD (Z-score) for FN (P = 0.0007) and LS (0.007) after adjusting by years since menopause, body mass index, smoking and menopausal status, densitometer type, hormone replacement therapy (HRT) use and HRT duration and after making the Bonferroni adjustment for multiple comparisons than did the remaining women. Logistic regression analysis adjusted by these covariates showed that TT + TT women had increased risk for FN (odds ratio (OR) = 2.76; P = 0.006) and LS (OR = 2.39; P = 0.020) osteopenia or osteoporosis than did the other women.
Conclusions
Our results suggest that interaction between genetic variants in the CD40 and CD40L genes exerts a role on BMD regulation. Further studies, which we welcome, are needed to replicate these data in other populations.

Similar content being viewed by others
References
Kanis JA, Burlet N, Cooper C et al (2008) European guidance for the diagnosis and management of osteoporosis in postmenopausal women. Osteoporos Int 19(4):399–428
Ralston SH, de Crombrugghe B (2006) Genetic regulation of bone mass and susceptibility to osteoporosis. Genes Dev 20(18):2492–2506
Ralston SH (2002) Genetic control of susceptibility to osteoporosis. J Clin Endocrinol Metab 87(6):2460–2466
Duncan EL, Brown MA (2008) Genetic studies in osteoporosis—the end of the beginning. Arthritis Res Ther 10(5):214
Videman T, Levalahti E, Battie MC, Simonen R, Vanninen E, Kaprio J (2007) Heritability of BMD of femoral neck and lumbar spine: a multivariate twin study of Finnish men. J Bone Miner Res 22(9):1455–1462
Ralston SH (2005) Genetic determinants of osteoporosis. Curr Opin Rheumatol 17(4):475–479
Weitzmann MN, Pacifici R (2006) Estrogen deficiency and bone loss: an inflammatory tale. J Clin Invest 116(5):1186–1194
Lorenzo J, Horowitz M, Choi Y (2008) Osteoimmunology: interactions of the bone and immune system. Endocr Rev 29(4):403–440
Arron JR, Choi Y (2000) Bone versus immune system. Nature 408(6812):535–536
Walsh MC, Kim N, Kadono Y et al (2006) Osteoimmunology: interplay between the immune system and bone metabolism. Annu Rev Immunol 24:33–63
Elgueta R, Benson MJ, de Vries V, Wasiuk A, Guo Y, Noelle RJ (2009) Molecular mechanism and function of CD40/CD40L engagement in the immune system. Immunol Rev 229(1):152–172
Rizvi M, Pathak D, Freedman JE, Chakrabarti S (2008) CD40–CD40 ligand interactions in oxidative stress, inflammation and vascular disease. Trends Mol Med 14(12):530–538
Lopez-Granados E, Temmerman ST, Wu L et al (2007) Osteopenia in X-linked hyper-IgM syndrome reveals a regulatory role for CD40 ligand in osteoclastogenesis. Proc Natl Acad Sci U S A 104(12):5056–5061
Li Y, Toraldo G, Li A et al (2007) B cells and T cells are critical for the preservation of bone homeostasis and attainment of peak bone mass in vivo. Blood 109(9):3839–3848
Pineda B, Laporta P, Hermenegildo C, Cano A, Garcia-Perez MA (2008) A C > T polymorphism located at position −1 of the Kozak sequence of CD40 gene is associated with low bone mass in Spanish postmenopausal women. Osteoporos Int 19(8):1147–1152
Cauley JA, Hochberg MC, Lui LY et al (2007) Long-term risk of incident vertebral fractures. JAMA 298(23):2761–2767
Kanis JA, Oden A, Johnell O et al (2007) The use of clinical risk factors enhances the performance of BMD in the prediction of hip and osteoporotic fractures in men and women. Osteoporos Int 18(8):1033–1046
Pineda B, Laporta P, Cano A, Garcia-Perez MA (2008) The Asn19Lys substitution in the osteoclast inhibitory lectin (OCIL) gene is associated with a reduction of bone mineral density in postmenopausal women. Calcif Tissue Int 82(5):348–353
Lu Y, Fuerst T, Hui S, Genant HK (2001) Standardization of bone mineral density at femoral neck, trochanter and Ward's triangle. Osteoporos Int 12(6):438–444
Hui SL, Gao S, Zhou XH et al (1997) Universal standardization of bone density measurements: a method with optimal properties for calibration among several instruments. J Bone Miner Res 12(9):1463–1470
Jacobson EM, Concepcion E, Oashi T, Tomer Y (2005) A Graves' disease-associated Kozak sequence single-nucleotide polymorphism enhances the efficiency of CD40 gene translation: a case for translational pathophysiology. Endocrinology 146(6):2684–2691
Conde L, Vaquerizas JM, Dopazo H et al (2006) PupaSuite: finding functional single nucleotide polymorphisms for large-scale genotyping purposes. Nucleic Acids Res 34(Web Server issue):W621–W625
Yuan HY, Chiou JJ, Tseng WH et al (2006) FASTSNP: an always up-to-date and extendable service for SNP function analysis and prioritization. Nucleic Acids Res 34(Web Server issue):W635–W641
Malarstig A, Lindahl B, Wallentin L, Siegbahn A (2006) Soluble CD40L levels are regulated by the −3459 A > G polymorphism and predict myocardial infarction and the efficacy of antithrombotic treatment in non-ST elevation acute coronary syndrome. Arterioscler Thromb Vasc Biol 26(7):1667–1673
Sole X, Guino E, Valls J, Iniesta R, Moreno V (2006) SNPStats: a web tool for the analysis of association studies. Bioinformatics 22(15):1928–1929
Gauderman WJ (2002) Sample size requirements for association studies of gene–gene interaction. Am J Epidemiol 155(5):478–484
Ahuja SS, Zhao S, Bellido T, Plotkin LI, Jimenez F, Bonewald LF (2003) CD40 ligand blocks apoptosis induced by tumor necrosis factor alpha, glucocorticoids, and etoposide in osteoblasts and the osteocyte-like cell line murine long bone osteocyte-Y4. Endocrinology 144(5):1761–1769
Schrum LW, Marriott I, Butler BR, Thomas EK, Hudson MC, Bost KL (2003) Functional CD40 expression induced following bacterial infection of mouse and human osteoblasts. Infect Immun 71(3):1209–1216
Acknowledgments
The authors are indebted to Mrs. R. Aliaga and Mrs. E. Calap for their excellent technical assistance. MAG-P is a recipient of a research contract from Fondo de Investigación Sanitaria-Consellería de Sanitat (Generalitat Valenciana).
Grants
This work was supported by grants PI06/154 and PS09/00184 from Fondo de Investigación Sanitaria (FIS, Madrid, Spain), AP-024/07, EVES 045-2007 and AP-077/08 from Conselleria de Sanitat (Generalitat Valenciana), Red HERACLES RD06/0009/0005 from Instituto Carlos III (Ministerio de Ciencia e Innovación; Madrid, Spain) and Fundación Santiago Dexeus Font (Barcelona, Spain).
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pineda, B., Tarín, J.J., Hermenegildo, C. et al. Gene–gene interaction between CD40 and CD40L reduces bone mineral density and increases osteoporosis risk in women. Osteoporos Int 22, 1451–1458 (2011). https://doi.org/10.1007/s00198-010-1324-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-010-1324-0