Abstract
Summary
Hypoparathyroidism, a disorder characterized by low parathyroid hormone (PTH), is generally treated with oral calcium and vitamin D supplementation. We investigated the effects of PTH(1–84) treatment in 30 hypoparathyroid subjects for 24 months. PTH(1–84) treatment in hypoparathyroidism significantly reduced supplemental calcium and 1,25-dihydroxyvitamin D requirements without generally altering serum and urinary calcium levels.
Introduction
Hypoparathyroidism, a disorder characterized by low PTH, is associated with hypocalcemia, hypercalciuria, and increased bone mineral density (BMD). Conventional therapy with calcium and 1,25-dihydroxyvitamin D can maintain the serum calcium concentration, but doses are high, and control is variable. We investigated the effects of human PTH(1–84) treatment in hypoparathyroidism.
Methods
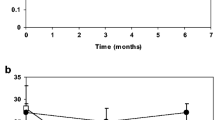
Thirty subjects with hypoparathyroidism were treated in an open-label study of PTH(1–84) 100 µg every other day by subcutaneous injection for 24 months, with monitoring of calcium and vitamin D supplementation requirements, serum and 24 h urinary calcium excretion, and BMD by dual energy X-ray absorptiometry.
Results
Requirements for supplemental calcium decreased significantly (3,030 ± 2,325 to 1,661 ± 1,267 mg/day (mean ± SD); p < 0.05), as did requirements for supplemental 1,25-dihydroxyvitamin D (0.68 ± 0.5 to 0.40 ± 0.5 µg/day; p < 0.05). Serum calcium levels and 24 h urinary calcium excretion were mostly unchanged at 24 months. BMD increased at the lumbar spine by 2.9 ± 4% from baseline (p < 0.05), while femoral neck BMD remained unchanged and distal one third radial BMD decreased by 2.4 ± 4% (p < 0.05).
Conclusion
PTH(1–84) treatment in hypoparathyroidism significantly reduces supplemental calcium and 1,25-dihydroxyvitamin D requirements without generally altering serum and urinary calcium levels.




Similar content being viewed by others
References
Marx S (2000) Hyperparathyroid and hypoparathyroid disorders. N Engl J Med 343:1863–1875
Thakker R (2004) Genetics of endocrine and metabolic disorders: parathyroid. Rev Endocr Metab Disord 5:37–51
Rubin M, Levine M (2008) Hypoparathyroidism. In: Primer of metabolic bone diseases. American Society of Bone and Mineral Research, Washington
Kao PC, van Heerden JA, Grant CS, Klee GG, Khosla S (1992) Clinical performance of parathyroid hormone immunometric assays. Mayo Clin Proc 67:637–645
Michelangeli VP, Heyma P, Colman PG, Ebeling PR (1997) Evaluation of a new, rapid and automated immunochemiluminometric assay for the measurement of serum intact parathyroid hormone. Ann Clin Biochem 34(Pt 1):97–103
Christiansen C, Rodbro P, Christensen MS, Hartnack B, Transbol I (1978) Deterioration of renal function during treatment of chronic renal failure with 1, 25-dihydroxycholecalciferol. Lancet 2:700–703
Kurokawa K (1987) Calcium-regulating hormones and the kidney. Kidney Int 32:760–771
Litvak J, Moldawer MP, Forbes AP, Henneman PH (1958) Hypocalcemic hypercalciuria during vitamin D and dihydrotachysterol therapy of hypoparathyroidism. J Clin Endocrinol Metab 18:246–252
Winer KK, Ko CW, Reynolds JC et al (2003) Long-term treatment of hypoparathyroidism: a randomized controlled study comparing parathyroid hormone-(1–34) versus calcitriol and calcium. J Clin Endocrinol Metab 88:4214–4220
Winer KK, Yanovski JA, Cutler GB Jr (1996) Synthetic human parathyroid hormone 1–34 vs calcitriol and calcium in the treatment of hypoparathyroidism. JAMA 276:631–636
Winer KK, Yanovski JA, Sarani B, Cutler GB Jr (1998) A randomized, cross-over trial of once-daily versus twice-daily parathyroid hormone 1–34 in treatment of hypoparathyroidism. J Clin Endocrinol Metab 83:3480–3486
Nussbaum SR, Zahradnik RJ, Lavigne JR et al (1987) Highly sensitive two-site immunoradiometric assay of parathyrin, and its clinical utility in evaluating patients with hypercalcemia. Clin Chem 33:1364–1367
Neer RM, Arnaud CD, Zanchetta JR et al (2001) Effect of parathyroid hormone (1–34) on fractures and bone mineral density in postmenopausal women with osteoporosis. N Engl J Med 344:1434–1441
Conflicts of interest
None.
Funding
DK067619, DK 069350, FD-R-02525, and NPS Pharmaceuticals.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Rubin, M.R., Sliney, J., McMahon, D.J. et al. Therapy of hypoparathyroidism with intact parathyroid hormone. Osteoporos Int 21, 1927–1934 (2010). https://doi.org/10.1007/s00198-009-1149-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-009-1149-x