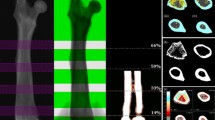
Abstract
Summary
The aim of this study is to demonstrate the deficiencies of dual-energy X-ray absorptiometry (DXA), compared with quantitative computed tomography, to reflect and differentiate between changes in bone mineral density and microstructure that contribute to a well-defined finding of altered skeletal state for both osteoporosis and renal osteodystrophy induced by chronic renal insufficiency.
Introduction
The aim of this study is to demonstrate the deficiencies of dual-energy X-ray absorptiometry (DXA), compared with quantitative CT, to reflect and differentiate between changes in bone mineral density and microstructure that contribute to a well-defined finding of altered skeletal state for both osteoporosis and renal osteodystrophy induced by chronic renal insufficiency.
Methods
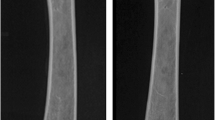
Forty-five female Sprague–Dawley rats were divided into three equal groups: control, ovariectomy, and nephrectomy. Following euthanasia, femurs were excised, divided into diaphyseal and distal metaphyseal sections, and subjected to DXA and micro-CT imaging and mechanical testing.
Results
Ovariectomy does not affect the structural and mechanical properties of cortical bone material, but partial nephrectomy does adversely affect these properties. Both are verified by DXA and micro-CT imaging and mechanical testing. Meanwhile, nephrectomy does not affect trabecular bone microstructure or equivalent density, yet ovariectomy affects the trabecular microstructure. DXA is unable to detect changes in trabecular bone microstructure in relation to changes in their mechanical properties.
Discussion
Dual energy X-ray absorptiometry measures the average bone mineral content in a 2D projected area and cannot differentiate whether the changes occur in the bone microstructure or equivalent bone tissue density. In contrast, micro-CT provides an accurate measurement of the changes in both equivalent bone tissue mineral density and microstructure for cancellous and cortical bone.




Similar content being viewed by others
References
Riggs BL, Melton LJ III (1995) The worldwide problem of osteoporosis: insights afforded by epidemiology. Bone 17:505S–511S
Chrischilles EA, Butler CD, Davis CS, Wallace RB (1991) A model of lifetime osteoporosis impact. Arch Intern Med 151:2026–2032
Roche JJ, Wenn RT, Sahota O, Moran CG (2005) Effect of comorbidities and postoperative complications on mortality after hip fracture in elderly people: prospective observational cohort study. BMJ 331:1374
Consensus Development Conference (1993) Diagnosis, prophylaxis, and treatment of osteoporosis. Am J Med 94:646–650
Schuit SC, van der Klift M, Weel AE et al (2004) Fracture incidence and association with bone mineral density in elderly men and women: the Rotterdam Study. Bone 34:195–202
Cummings SR, Karpf DB, Harris F et al (2002) Improvement in spine bone density and reduction in risk of vertebral fractures during treatment with antiresorptive drugs. Am J Med 112:281–289
Heaney RP (2003) Is the paradigm shifting? Bone 33:457–465
Bauer DC, Black DM, Garnero P et al (2004) Change in bone turnover and hip, non-spine, and vertebral fracture in alendronate-treated women: the Fracture Intervention Trial. J Bone Miner Res 19:1250–1258
Riggs BL, Melton LJ III (2002) Bone turnover matters: the raloxifene treatment paradox of dramatic decreases in vertebral fractures without commensurate increases in bone density. J Bone Miner Res 17:11–14
Hayes WC, Bouxsein ML (1997) Biomechanics of cortical and trabecular bone: implication for assessment of fracture risk. In: Mow VC, Hayes WC (eds) Basic orthopaedic biomechanics. Lippincott-Raven, Philadelphia, pp 69–112
Rice JC, Cowin SC, Bowman JA (1988) On the dependence of the elasticity and strength of cancellous bone on apparent density. J Biomech 21:155–168
Snyder SM, Schneider E (1991) Estimation of mechanical properties of cortical bone by computed tomography. J Orthop Res 9:422–431
Favus MJ (ed) (2006) Primer on the metabolic bone diseases and disorders of mineral metabolism. American Society for Bone and Mineral Research, Washington, DC
LeBoff MS, Kohlmeier L, Hurwitz S, Franklin J, Wright J, Glowacki J (1999) Occult vitamin D deficiency in postmenopausal US women with acute hip fracture. JAMA 281:1505–1511
Glowacki J, Hurwitz S, Thornhill TS, Kelly M, LeBoff MS (2003) Osteoporosis and vitamin-D deficiency among postmenopausal women with osteoarthritis undergoing total hip arthroplasty. J Bone Joint Surg Am 85-A:2371–2377
Aaron J, Gallagher JC, Nordin BE (1974) Osteomalacia [letter]. Lancet 1:939
Aaron J, Gallagher JC, Nordin BE (1974) Osteomalacia and femoral fractures [letter]. Lancet 1:572
Chalmers J, Barclay A, Davison AM, Macleod DA, Williams DA (1969) Quantitative measurements of osteoid in health and disease. Clin Orthop Relat Res 63:196–209
Sokoloff L (1978) Occult osteomalacia in American (U.S.A.) patients with fracture of the hip. Am J Surg Pathol 2:21–30
Lips P, Netelenbos JC, Jongen MJ et al (1982) Histomorphometric profile and vitamin D status in patients with femoral neck fracture. Metab Bone Dis Relat Res 4:85–93
Kanis JA, Melton LJ III, Christiansen C, Johnston CC, Khaltaev N (1994) The diagnosis of osteoporosis. J Bone Miner Res 9:1137–1141
Miller SC, Wronski TJ (1993) Long-term osteopenic changes in cancellous bone structure in ovariectomized rats. Anat Rec 236:433–441
Reddy Nagareddy P, Lakshmana M (2005) Assessment of experimental osteoporosis using CT-scanning, quantitative X-ray analysis and impact test in calcium deficient ovariectomized rats. J Pharmacol Toxicol Methods 52:350–355
Yao W, Hadi T, Jiang Y, Lotz J, Wronski TJ, Lane NE (2005) Basic fibroblast growth factor improves trabecular bone connectivity and bone strength in the lumbar vertebral body of osteopenic rats. Osteoporos Int 16:1939–1947
Ito M, Nishida A, Aoyagi K, Uetani M, Hayashi K, Kawase M (2005) Effects of risedronate on trabecular microstructure and biomechanical properties in ovariectomized rat tibia. Osteoporos Int 16:1042–1048
Hornby SB, Evans GP, Hornby SL, Pataki A, Glatt M, Green JR (2003) Long-term zoledronic acid treatment increases bone structure and mechanical strength of long bones of ovariectomized adult rats. Calcif Tissue Int 72:519–527
Ogawa K, Hori M, Takao R, Sakurada T (2005) Effects of combined elcatonin and alendronate treatment on the architecture and strength of bone in ovariectomized rats. J Bone Miner Metab 23:351–358
Kaczmarczyk-Sedlak I, Janiec W, Pytlik M et al (2005) Effect of administration of etidronate and retinol on bone mechanical properties in ovariectomized rats. Pharmacol Rep 57:203–211
Guo XE, Goldstein SA (2000) Vertebral trabecular bone microscopic tissue elastic modulus and hardness do not change in ovariectomized rats. J Orthop Res 18:333–336
Wang L, Orhii PB, Banu J, Kalu DN (2001) Effects of separate and combined therapy with growth hormone and parathyroid hormone on lumbar vertebral bone in aged ovariectomized osteopenic rats. Bone 28:202–207
Feng JQ, Ward LM, Liu S et al (2006) Loss of DMP1 causes rickets and osteomalacia and identifies a role for osteocytes in mineral metabolism. Nat Genet 38:1310–1315
Turner CH, Owan I, Brizendine EJ, Zhang W, Wilson ME, Dunipace AJ (1996) High fluoride intakes cause osteomalacia and diminished bone strength in rats with renal deficiency. Bone 19:595–601
Jokihaara J, Jarvinen TL, Jolma P et al (2006) Renal insufficiency-induced bone loss is associated with an increase in bone size and preservation of strength in rat proximal femur. Bone 39:353–360
Miller MA, Chin J, Miller SC, Fox J (1998) Disparate effects of mild, moderate, and severe secondary hyperparathyroidism on cancellous and cortical bone in rats with chronic renal insufficiency. Bone 23:257–266
Kazama JJ, Iwasaki Y, Yamato H et al (2003) Microfocus computed tomography analysis of early changes in bone microstructure in rats with chronic renal failure. Nephron 95:e152–e157
Brkovic D, Linke J, Jakse G, Bauss F (2005) Changes in bone structure after augmentation cystoplasty in chronic uraemic rats. BJU Int 95:1066–1070
Freesmeyer MG, Abendroth K, Faldum A, Krauss C, Stein G (2001) Comparison of peripheral bone and body axis skeleton in a rat model of mild-to-moderate renal failure in the presence of physiological serum levels of calcitropic hormones. Bone 29:258–264
Melhus G, Solberg LB, Dimmen S, Madsen JE, Nordsletten L, Reinholt FP (2007) Experimental osteoporosis induced by ovariectomy and vitamin D deficiency does not markedly affect fracture healing in rats. Acta Orthop 78:393–403
Keaveny TM, Borchers RE, Gibson LJ, Hayes WC (1993) Theoretical analysis of the experimental artifact in trabecular bone compressive modulus. J Biomech 26:599–607
Hildebrand T, Laib A, Muller R, Dequeker J, Ruegsegger P (1999) Direct three-dimensional morphometric analysis of human cancellous bone: microstructural data from spine, femur, iliac crest, and calcaneus. J Bone Miner Res 14:1167–1174
Hildebrand T, Ruegsegger P (1997) Quantification of bone microarchitecture with the structure model index. Comput Methods Biomech Biomed Eng 1:15–23
Odgaard A, Gundersen HJ (1993) Quantification of connectivity in cancellous bone, with special emphasis on 3-D reconstructions. Bone 14:173–182
Laib A, Kumer JL, Majumdar S, Lane NE (2001) The temporal changes of trabecular architecture in ovariectomized rats assessed by MicroCT. Osteoporos Int 12:936–941
Ocarino NM, Marubayashi U, Cardoso TG et al (2007) Physical activity in osteopenia treatment improved the mass of bones directly and indirectly submitted to mechanical impact. J Musculoskelet Neuronal Interact 7:84–93
Jiang SD, Shen C, Jiang LS, Dai LY (2007) Differences of bone mass and bone structure in osteopenic rat models caused by spinal cord injury and ovariectomy. Osteoporos Int 18:743–750
Iwaniec UT, Moore K, Rivera MF, Myers SE, Vanegas SM, Wronski TJ (2007) A comparative study of the bone-restorative efficacy of anabolic agents in aged ovariectomized rats. Osteoporos Int 18:351–362
Acknowledgements
The authors would like to acknowledge the Komen Foundation for providing financial support for this project (BDS Grant No: BCTR0403271). Additionally, they would like to acknowledge the efforts of Michael Bohanske, John Muller, Laura Gould, and Jeffrey O’Connell from the Orthopedic Biomechanics Laboratory with specimen preparation and testing.
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Additional information
A. Nazarian and E. Cory have contributed equally to this work.
Rights and permissions
About this article
Cite this article
Nazarian, A., Cory, E., Müller, R. et al. Shortcomings of DXA to assess changes in bone tissue density and microstructure induced by metabolic bone diseases in rat models. Osteoporos Int 20, 123–132 (2009). https://doi.org/10.1007/s00198-008-0632-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-008-0632-0