Abstract
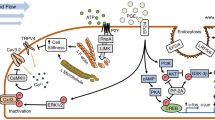
Bone loss during skeletal unloading, whether due to neurotrauma resulting in paralysis or prolonged immobilization due to a variety of medical illnesses, accelerates bone loss. In this review the evidence that skeletal unloading leads to bone loss, at least in part, due to disrupted insulin like growth factor (IGF) signaling, resulting in reduced osteoblast proliferation and differentiation, will be examined. The mechanism underlying this disruption in IGF signaling appears to involve integrins, the expression of which is reduced during skeletal unloading. Integrins play an important, albeit not well defined, role in facilitating signaling not only by IGF but also by other growth factors. However, the interaction between selected integrins such as αυβ3 and β1 integrins and the IGF receptor are of especial importance with respect to the ability of bone to respond to mechanical load. Disruption of this interaction blocks IGF signaling and results in bone loss.




Similar content being viewed by others
Abbreviations
- BMSC:
-
Bone marrow stromal cells
- FAK:
-
Focal adhesion kinase
- grb2:
-
Growth receptor binding protein-2
- GEF:
-
Guanine nucleotide exchange factor
- IGF-R:
-
Insulin like growth factor (IGF) and its receptor
- IRS:
-
Insulin receptor substrate
- MAPK:
-
Mitogen activated protein kinase
- NO:
-
Nitric oxide
- PI3K:
-
Phosphatidyl inositol 3 kinase
- PIP2 and PIP3 :
-
Phosphatidyl inositol bis- and tris-phosphate
- PDK:
-
Phosphoinositide dependent kinase
- PTB:
-
Phosphotyrosine binding protein
- Pyk:
-
Phosphotyrosine kinase
- PGE2 :
-
Prostaglandin E2
- PKB/Akt:
-
Protein kinase B
- SOS:
-
Son of sevenless
- SH2:
-
src homology 2
- SHPS:
-
SH2 domain containing protein tyrosine phosphatase (SHP) and its substrate
References
Robling AG, Castillo AB, Turner CH (2006) Biomechanical and molecular regulation of bone remodeling. Ann Rev Biomed Eng 8:455–498
Robling AG, Hinant FM, Burr DB, Turner CH (2002) Improved bone structure and strength after long-term mechanical loading is greatest if loading is separated into short bouts. J Bone Miner Res 17:1545–1554
Lanyon LE, Rubin CT (1984) Static vs dynamic loads as an influence on bone remodelling. J Biomech 17:897–905
Turner CH, Owan I, Takano Y (1995) Mechanotransduction in bone: role of strain rate. Am J Physiol 269:E438–E442
Weinbaum S, Cowin SC, Zeng Y (1994) A model for the excitation of osteocytes by mechanical loading-induced bone fluid shear stresses. J Biomech 27:339–360
Li J, Duncan RL, Burr DB, Gattone VH, Turner CH (2003) Parathyroid hormone enhances mechanically induced bone formation, possibly involving L-type voltage-sensitive calcium channels. Endocrinology 144:1226–1233
Rawlinson SC, Pitsillides AA, Lanyon LE (1996) Involvement of different ion channels in osteoblasts’ and osteocytes’ early responses to mechanical strain. Bone 19:609–614
Genetos DC, Geist DJ, Liu D, Donahue HJ, Duncan RL (2005) Fluid shear-induced ATP secretion mediates prostaglandin release in MC3T3-E1 osteoblasts. J Bone Miner Res 20:41–49
Jorgensen NR, Geist ST, Civitelli R, Steinberg TH (1997) ATP- and gap junction-dependent intercellular calcium signaling in osteoblastic cells. J Cell Biol 139:497–506
Bakker AD, Soejima K, Klein-Nulend J, Burger EH (2001) The production of nitric oxide and prostaglandin E(2) by primary bone cells is shear stress dependent. J Biomech 34:671–677
Lean JM, Jagger CJ, Chambers TJ, Chow JW (1995) Increased insulin-like growth factor I mRNA expression in rat osteocytes in response to mechanical stimulation. Am J Physiol 268:E318–E327
Reijnders CM, Bravenboer N, Tromp AM, Blankenstein MA, Lips P (2007) Effect of mechanical loading on insulin-like growth factor-I gene expression in rat tibia. J Endocrinol 192:131–140
Kapur S, Mohan S, Baylink DJ, Lau KH (2005) Fluid shear stress synergizes with insulin-like growth factor-I (IGF-I) on osteoblast proliferation through integrin-dependent activation of IGF-I mitogenic signaling pathway. J Biol Chem 280:20163–20170
Morey ER, Baylink DJ (1978) Inhibition of bone formation during space flight. Science 201:1138–1141
Wronski TJ, Morey ER (1983) Effect of spaceflight on periosteal bone formation in rats. Am J Physiol 244:R305–R309
Vico L, Novikov VE, Very JM, Alexandre C (1991) Bone histomorphometric comparison of rat tibial metaphysis after 7-day tail suspension vs. 7-day spaceflight. Aviat Space Environ Med 62:26–31
Oganov VS, Rakhmanov AS, Novikov VE, Zatsepin ST, Rodionova SS, Cann C (1991) The state of human bone tissue during space flight. Acta Astronaut 23:129–133
Vico L, Alexandre C (1992) Microgravity and bone adaptation at the tissue level. J Bone Miner Res 7(Suppl 2):S445–S447
Zerath E, Holy X, Malouvier A, Caissard JC, Nogues C (1991) Rat and monkey bone study in the Biocosmos 2044 space experiment. Physiologist 34:S194–S195
Jee WS, Wronski TJ, Morey ER, Kimmel DB (1983) Effects of spaceflight on trabecular bone in rats. Am J Physiol 244:R310–R314
Yagodovsky VS, Triftanidi LA, Gorokhova GP (1976) Space flight effects on skeletal bones of rats (light and electron microscopic examination). Aviat Space Environ Med 47:734–738
Vico L, Chappard D, Palle S, Bakulin AV, Novikov VE, Alexandre C (1988) Trabecular bone remodeling after seven days of weightlessness exposure (BIOCOSMOS 1667). Am J Physiol 255:R243–R247
Turner RT, Evans GL, Wakley GK (1995) Spaceflight results in depressed cancellous bone formation in rat humeri. Aviat Space Environ Med 66:770–774
Garetto LP, Gonsalves MR, Morey ER, Durnova G, Roberts WE (1990) Preosteoblast production 55 hours after a 12.5-day spaceflight on Cosmos 1887. Faseb J 4:24–28
Globus RK, Bikle DD, Morey-Holton E (1984) Effects of simulated weightlessness on bone mineral metabolism. Endocrinology 114:2264–2270
Halloran BP, Bikle DD, Cone CM, Morey-Holton E (1988) Glucocorticoids and inhibition of bone formation induced by skeletal unloading. Am J Physiol 255:E875–E879
Globus RK, Bikle DD, Morey-Holton E (1986) The temporal response of bone to unloading. Endocrinology 118:733–742
Abram AC, Keller TS, Spengler DM (1988) The effects of simulated weightlessness on bone biomechanical and biochemical properties in the maturing rat. J Biomech 21:755–767
Bikle DD, Halloran BP, Cone CM, Globus RK, Morey-Holton E (1987) The effects of simulated weightlessness on bone maturation. Endocrinology 120:678–684
Kidder LS, Klein GL, Stuart CA, Lee TC, Gundberg CM, Alcock N, Cooper CW, Simmons DJ (1990) Skeletal effects of sodium fluoride during hypokinesia. Bone Miner 11:305–318
LeBlanc A, Marsh C, Evans H, Johnson P, Schneider V, Jhingran S (1985) Bone and muscle atrophy with suspension of the rat. J Appl Physiol 58:1669–1675
Halloran BP, Bikle DD, Wronski TJ, Globus RK, Levens MJ, Morey-Holton E (1986) The role of 1,25-dihydroxyvitamin D in the inhibition of bone formation induced by skeletal unloading. Endocrinology 118:948–954
Patterson-Buckendahl P, Globus RK, Bikle DD, Cann CE, Morey-Holton E (1989) Effects of simulated weightlessness on rat osteocalcin and bone calcium. Am J Physiol 257:R1103–R1109
Shaw SR, Zernicke RF, Vailas AC, DeLuna D, Thomason DB, Baldwin KM (1987) Mechanical, morphological and biochemical adaptations of bone and muscle to hindlimb suspension and exercise. J Biomech 20:225–234
Machwate M, Zerath E, Holy X, Hott M, Modrowski D, Malouvier A, Marie PJ (1993) Skeletal unloading in rat decreases proliferation of rat bone and marrow-derived osteoblastic cells. Am J Physiol 264:E790–E799
Le Roith D, Bondy C, Yakar S, Liu JL, Butler A (2001) The somatomedin hypothesis: 2001. Endocr Rev 22:53–74
Favelyukis S, Till JH, Hubbard SR, Miller WT (2001) Structure and autoregulation of the insulin-like growth factor 1 receptor kinase. Nat Struct Biol 8:1058–1063
Kiely PA, Leahy M, O’ Gorman D, O’ Connor R (2005) RACK1-mediated integration of adhesion and insulin-like growth factor I (IGF-I) signaling and cell migration are defective in cells expressing an IGF-I receptor mutated at tyrosines 1250 and 1251. J Biol Chem 280:7624–7633
Bikle DD, Harris J, Halloran BP, Morey-Holton E (1994) Altered skeletal pattern of gene expression in response to spaceflight and hindlimb elevation. Am J Physiol 267:E822–E827
Canalis E (1985) Effect of growth factors on bone cell replication and differentiation. Clin Orthop 193:246–263
Baker J, Liu JP, Robertson EJ, Efstratiadis A (1993) Role of insulin-like growth factors in embryonic and postnatal growth. Cell 75:73–82
Powell-Braxton L, Hollingshead P, Warburton C, Dowd M, Pitts-Meek S, Dalton D, Gillett N, Stewart TA (1993) IGF-I is required for normal embryonic growth in mice. Genes Dev 7:2609–2617
Bikle D, Majumdar S, Laib A, Powell-Braxton L, Rosen C, Beamer W, Nauman E, Leary C, Halloran B (2001) The skeletal structure of insulin-like growth factor I-deficient mice. J Bone Miner Res 16:2320–2329
Zhao G, Monier-Faugere MC, Langub MC, Geng Z, Nakayama T, Pike JW, Chernausek SD, Rosen CJ, Donahue LR, Malluche HH, Fagin JA, Clemens TL (2000) Targeted overexpression of insulin-like growth factor I to osteoblasts of transgenic mice: increased trabecular bone volume without increased osteoblast proliferation. Endocrinology 141:2674–2682
Jiang J, Lichtler AC, Gronowicz GA, Adams DJ, Clark SH, Rosen CJ, Kream BE (2006) Transgenic mice with osteoblast-targeted insulin-like growth factor-I show increased bone remodeling. Bone 39:494–504
Zhang M, Xuan S, Bouxsein ML, von Stechow D, Akeno N, Faugere MC, Malluche H, Zhao G, Rosen CJ, Efstratiadis A, Clemens TL (2002) Osteoblast-specific knockout of the insulin-like growth factor (IGF) receptor gene reveals an essential role of IGF signaling in bone matrix mineralization. J Biol Chem 277:44005–44012
Wang Y, Nishida S, Boudignon BM, Burghardt A, Elalieh HZ, Hamilton MM, Majumdar S, Halloran BP, Clemens TL, Bikle DD (2007) IGF-I receptor is required for the anabolic actions of parathyroid hormone on bone. J Bone Miner Res 22:1329–1337
Wang Y, Nishida S, Elalieh HZ, Long RK, Halloran BP, Bikle DD (2006) Role of IGF-I signaling in regulating osteoclastogenesis. J Bone Miner Res 21:1350–1358
Boudignon BM, Bikle DD, Kurimoto P, Elalieh H, Nishida S, Wang Y, Burghardt A, Majumdar S, Orwoll BE, Rosen C, Halloran BP (2007) Insulin-like Growth Factor-I Stimulates Recovery of Bone Lost after a Period of Skeletal Unloading. J Appl Physiol 103:125–131
Kostenuik PJ, Halloran BP, Morey-Holton ER, Bikle DD (1997) Skeletal unloading inhibits the in vitro proliferation and differentiation of rat osteoprogenitor cells. Am J Physiol 273:E1133–E1139
Sakata T, Halloran BP, Elalieh HZ, Munson SJ, Rudner L, Venton L, Ginzinger D, Rosen CJ, Bikle DD (2003) Skeletal unloading induces resistance to insulin-like growth factor I on bone formation. Bone 32:669–680
Sakata T, Wang Y, Halloran BP, Elalieh HZ, Cao J, Bikle DD (2004) Skeletal unloading induces resistance to insulin-like growth factor-I (IGF-I) by inhibiting activation of the IGF-I signaling pathways. J Bone Miner Res 19:436–446
Kostenuik PJ, Harris J, Halloran BP, Turner RT, Morey-Holton ER, Bikle DD (1999) Skeletal unloading causes resistance of osteoprogenitor cells to parathyroid hormone and to insulin-like growth factor-I. J Bone Miner Res 14:21–31
Giancotti FG, Ruoslahti E (1999) Integrin signaling. Science 285:1028–1032
Schwartz MA (2001) Integrin signaling revisited. Trends Cell Biol 11:466–470
Takada Y, Ye X, Simon S (2007) The integrins. Genome biology 8:215
Sheppard D (2000) In vivo functions of integrins: lessons from null mutations in mice. Matrix Biol 19:203–209
Asthagiri AR, Nelson CM, Horwitz AF, Lauffenburger DA (1999) Quantitative relationship among integrin-ligand binding, adhesion, and signaling via focal adhesion kinase and extracellular signal-regulated kinase 2. J Biol Chem 274:27119–27127
Horne WC, Sanjay A, Bruzzaniti A, Baron R (2005) The role(s) of Src kinase and Cbl proteins in the regulation of osteoclast differentiation and function. Immunol Rev 208:106–125
Wary KK, Mainiero F, Isakoff SJ, Marcantonio EE, Giancotti FG (1996) The adaptor protein Shc couples a class of integrins to the control of cell cycle progression. Cell 87:733–743
Wary KK, Mariotti A, Zurzolo C, Giancotti FG (1998) A requirement for caveolin-1 and associated kinase Fyn in integrin signaling and anchorage-dependent cell growth. Cell 94:625–634
Kaiser E, Sato M, Onyia JE, Chandrasekhar S (2001) Parathyroid hormone (1–34) regulates integrin expression in vivo in rat osteoblasts. J Cell Biochem 83:617–630
Lai CF, Chaudhary L, Fausto A, Halstead LR, Ory DS, Avioli LV, Cheng SL (2001) Erk is essential for growth, differentiation, integrin expression, and cell function in human osteoblastic cells. J Biol Chem 276:14443–14450
Gronthos S, Simmons PJ, Graves SE, Robey PG (2001) Integrin-mediated interactions between human bone marrow stromal precursor cells and the extracellular matrix. Bone 28:174–181
Moursi AM, Globus RK, Damsky CH (1997) Interactions between integrin receptors and fibronectin are required for calvarial osteoblast differentiation in vitro. J Cell Sci 110:2187–2196
Pistone M, Sanguineti C, Federici A, Sanguineti F, Defilippi P, Santolini F, Querze G, Marchisio PC, Manduca P (1996) Integrin synthesis and utilization in cultured human osteoblasts. Cell Biol Int 20:471–479
Lai CF, Cheng SL (2005) Alphavbeta integrins play an essential role in BMP-2 induction of osteoblast differentiation. J Bone Miner Res 20:330–340
Cowles EA, Brailey LL, Gronowicz GA (2000) Integrin-mediated signaling regulates AP-1 transcription factors and proliferation in osteoblasts. J Biomed Mater Res 52:725–737
Gronthos S, Stewart K, Graves SE, Hay S, Simmons PJ (1997) Integrin expression and function on human osteoblast-like cells. J Bone Miner Res 12:1189–1197
Globus RK, Doty SB, Lull JC, Holmuhamedov E, Humphries MJ, Damsky CH (1998) Fibronectin is a survival factor for differentiated osteoblasts. J Cell Sci 111:1385–1393
Mizuno M, Fujisawa R, Kuboki Y (2000) Type I collagen-induced osteoblastic differentiation of bone-marrow cells mediated by collagen-alpha2beta1 integrin interaction. J Cell Physiol 184:207–213
Zimmerman D, Jin F, Leboy P, Hardy S, Damsky C (2000) Impaired bone formation in transgenic mice resulting from altered integrin function in osteoblasts. Dev Biol 220:2–15
Miura Y, Miura M, Gronthos S, Allen MR, Cao C, Uveges TE, Bi Y, Ehirchiou D, Kortesidis A, Shi S, Zhang L (2005) Defective osteogenesis of the stromal stem cells predisposes CD18-null mice to osteoporosis. Proc Natl Acad Sci USA 102:14022–14027
Ross FP, Teitelbaum SL (2005) alphavbeta3 and macrophage colony-stimulating factor: partners in osteoclast biology. Immunol Rev 208:88–105
Lane NE, Yao W, Nakamura MC, Humphrey MB, Kimmel D, Huang X, Sheppard D, Ross FP, Teitelbaum SL (2005) Mice lacking the integrin beta5 subunit have accelerated osteoclast maturation and increased activity in the estrogen-deficient state. J Bone Miner Res 20:58–66
Faccio R, Teitelbaum SL, Fujikawa K, Chappel J, Zallone A, Tybulewicz VL, Ross FP, Swat W (2005) Vav3 regulates osteoclast function and bone mass. Nat Med 11:284–290
Zou W, Kitaura H, Reeve J, Long F, Tybulewicz VL, Shattil SJ, Ginsberg MH, Ross FP, Teitelbaum SL (2007) Syk, c-Src, the alphavbeta3 integrin, and ITAM immunoreceptors, in concert, regulate osteoclastic bone resorption. J Cell Biol 176:877–888
Murphy MG, Cerchio K, Stoch SA, Gottesdiener K, Wu M, Recker R (2005) Effect of L-000845704, an alphaVbeta3 integrin antagonist, on markers of bone turnover and bone mineral density in postmenopausal osteoporotic women. J Clin Endocrinol Metab 90:2022–2028
Dufour C, Holy X, Marie PJ (2007) Skeletal unloading induces osteoblast apoptosis and targets alpha5beta1-PI3K-Bcl-2 signaling in rat bone. Exp Cell Res 313:394–403
Liedert A, Kaspar D, Blakytny R, Claes L, Ignatius A (2006) Signal transduction pathways involved in mechanotransduction in bone cells. Biochem Biophys Res Commun 349:1–5
Pavalko FM, Norvell SM, Burr DB, Turner CH, Duncan RL, Bidwell JP (2003) A model for mechanotransduction in bone cells: the load-bearing mechanosomes. J Cell Biochem 88:104–112
Meyers VE, Zayzafoon M, Douglas JT, McDonald JM (2005) RhoA and cytoskeletal disruption mediate reduced osteoblastogenesis and enhanced adipogenesis of human mesenchymal stem cells in modeled microgravity. J Bone Miner Res 20:1858–1866
Meyers VE, Zayzafoon M, Gonda SR, Gathings WE, McDonald JM (2004) Modeled microgravity disrupts collagen I/integrin signaling during osteoblastic differentiation of human mesenchymal stem cells. J Cell Biochem 93:697–707
Wang Y, McNamara LM, Schaffler MB, Weinbaum S (2007) A model for the role of integrins in flow induced mechanotransduction in osteocytes. Proc Natl Acad Sci USA 104:15941–15946
Carvalho RS, Bumann A, Schaffer JL, Gerstenfeld LC (2002) Predominant integrin ligands expressed by osteoblasts show preferential regulation in response to both cell adhesion and mechanical perturbation. J Cell Biochem 84:497–508
Morinobu M, Ishijima M, Rittling SR, Tsuji K, Yamamoto H, Nifuji A, Denhardt DT, Noda M (2003) Osteopontin expression in osteoblasts and osteocytes during bone formation under mechanical stress in the calvarial suture in vivo. J Bone Miner Res 18:1706–1715
Fujihara S, Yokozeki M, Oba Y, Higashibata Y, Nomura S, Moriyama K (2006) Function and regulation of osteopontin in response to mechanical stress. J Bone Miner Res 21:956–964
Ishijima M, Tsuji K, Rittling SR, Yamashita T, Kurosawa H, Denhardt DT, Nifuji A, Noda M (2002) Resistance to unloading-induced three-dimensional bone loss in osteopontin-deficient mice. J Bone Miner Res 17:661–667
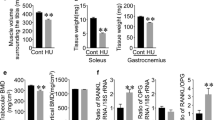
Iwaniec UT, Wronski TJ, Amblard D, Nishimura Y, van der Meulen MC, Wade CE, Bourgeois MA, Damsky CD, Globus RK (2005) Effects of disrupted beta1-integrin function on the skeletal response to short-term hindlimb unloading in mice. J Appl Physiol 98:690–696
Borges E, Jan Y, Ruoslahti E (2000) Platelet-derived growth factor receptor beta and vascular endothelial growth factor receptor 2 bind to the beta 3 integrin through its extracellular domain. J Biol Chem 275:39867–39873
Maile LA, Badley-Clarke J, Clemmons DR (2001) Structural analysis of the role of the beta 3 subunit of the alpha V beta 3 integrin in IGF-I signaling. J Cell Sci 114:1417–1425
Zheng B, Clemmons DR (1998) Blocking ligand occupancy of the alphaVbeta3 integrin inhibits insulin-like growth factor I signaling in vascular smooth muscle cells. Proc Natl Acad Sci USA 95:11217–11222
Clemmons DR, Maile LA (2005) Interaction between insulin-like growth factor-I receptor and alphaVbeta3 integrin linked signaling pathways: cellular responses to changes in multiple signaling inputs. Mol Endocrinol 19:1–11
Long RK, Nishida S, Bikle DD (2005) IGF-1 stimulated b3 integrin recruitment is associated with potentiation of the IGF-1 receptor. J Bone Min Res 20:S250
Maile LA, Busby WH, Sitko K, Capps BE, Sergent T, Badley-Clarke J, Clemmons DR (2006) Insulin-like growth factor-I signaling in smooth muscle cells is regulated by ligand binding to the 177CYDMKTTC184 sequence of the beta3-subunit of alphaVbeta3. Mol Endocrinol 20:405–413
Boutahar N, Guignandon A, Vico L, Lafage-Proust MH (2004) Mechanical strain on osteoblasts activates autophosphorylation of focal adhesion kinase and proline-rich tyrosine kinase 2 tyrosine sites involaved in ERK activation. J Biol Chem 279:30588–30599
Acknowledgements
The author has drawn from the work of his current and former postdoctoral fellows Drs. Paul Kostenuik, Takashi Sakata, Shigeki Nishida, Yongmei Wang, and Roger Long as well as his long standing collaboration with Dr. Bernard Halloran. The work has been and is supported by a Veterans Affairs Research Enhancement Award Program, a grant from the National Aeronautics and Space Administration (NNA04CC67G), a grant from the National Institutes of Health (RO1 DK54793) to the author, and a National Space Biomedical Research Institute Postdoctoral Fellowship to Dr. Long.
Conflict of interest statement
No conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Additional information
The skeletal response to mechanical load is critical for maintenance of skeletal integrity. This review will assess the interacting roles that insulin like growth factor I (IGF-I) signaling and selected integrins play in this response. Skeletal unloading results in decreased integrin expression, resistance to the anabolic actions of IGF-I, and bone loss.
Rights and permissions
About this article
Cite this article
Bikle, D.D. Integrins, insulin like growth factors, and the skeletal response to load. Osteoporos Int 19, 1237–1246 (2008). https://doi.org/10.1007/s00198-008-0597-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-008-0597-z