Abstract
Summary
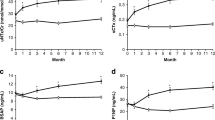
In the PaTH trial, among the 119 women randomized to parathyroid hormone PTH(1–84) and 60 to alendronate, we found much greater variation in BMD and markers in response to PTH(1–84) compared to alendronate. No baseline participant characteristic consistently predicted increased bone density response to PTH(1–84), although women with larger changes in 1,25 dihydroxyvitamin D during therapy had larger increases in BMD.
Introduction
We examined variability in BMD and markers of bone turnover in response to treatment with PTH(1–84) or alendronate in the PaTH trial.
Methods
Differences in SD were examined using Levine’s test for homogeneity of variance. Change in BMD across quartiles of participant characteristics was examined using ANOVA.
Results
We found much greater variation in response to PTH(1–84) compared to alendronate. The SD for change in cancellous spine BMD (by QCT) was 32% on PTH(1–84) compared to 13% on alendronate (p < 0.0001). The higher variability in the PTH(1–84) group was due to substantial numbers of women with large increases in BMD on PTH(1–84). Similarly, the SD of changes in markers of formation and resorption were significantly higher on PTH(1–84) than on ALN. No baseline participant characteristics predicted increased bone density response to PTH(1–84) therapy. However, change in 1,25-OH2D explained 16% of the variance in BMD response to PTH(1–84).
Conclusion
There is significant variability in the skeletal response to PTH(1–84), which exceeds that observed with alendronate. Changes in 1,25-OH2D were related to larger gains in BMD. This finding may have implications for elucidating either the pathway by which PTH affects the skeleton or traits that result in particular responsiveness to PTH therapy.



Similar content being viewed by others
References
Black DM, Cummings SR, Karpf DB, Cauley JA, Thompson DE, Nevitt MC, Bauer DC, Genant HK, Haskell WL, Marcus R, Ott SM, Torner JC, Quandt SA, Reiss TF, Ensrud KE (1996) Randomised trial of effect of alendronate on risk of fracture in women with existing vertebral fractures. Fracture Intervention Trial Research Group. Lancet 348(9041):1535–1541
Delmas PD, Ensrud KE, Adachi JD, Harper KD, Sarkar S, Gennari C, Reginster JY, Pols HA, Recker RR, Harris ST, Wu W, Genant HK, Black DM, Eastell R (2002) Efficacy of raloxifene on vertebral fracture risk reduction in postmenopausal women with osteoporosis: four-year results from a randomized clinical trial. J Clin Endocrinol Metab 87(8):3609–3617
McClung MR, Geusens P, Miller PD, Zippel H, Bensen WG, Roux C, Adami S, Fogelman I, Diamond T, Eastell R, Meunier PJ, Reginster JY (2001) Effect of risedronate on the risk of hip fracture in elderly women. Hip Intervention Program Study Group. N Engl J Med 344(5):333–340
Chesnut CH III, Silverman S, Andriano K, Genant H, Gimona A, Harris S, Kiel D, LeBoff M, Maricic M, Miller P, Moniz C, Peacock M, Richardson P, Watts N, Baylink D (2000) A randomized trial of nasal spray salmon calcitonin in postmenopausal women with established osteoporosis: the prevent recurrence of osteoporotic fractures study. PROOF Study Group. Am J Med 109(4):267–276
Kurland ES, Cosman F, McMahon DJ, Rosen CJ, Lindsay R, Bilezikian JP (2000) Parathyroid hormone as a therapy for idiopathic osteoporosis in men: effects on bone mineral density and bone markers. J Clin Endocrinol Metab 85(9):3069–3076
Kurland ES, Heller SL, Diamond B, McMahon DJ, Cosman F, Bilezikian JP (2004) The importance of bisphosphonate therapy in maintaining bone mass in men after therapy with teriparatide [human parathyroid hormone(1–34)]. Osteoporos Int 15(12):992–997
Neer RM, Arnaud CD, Zanchetta JR, Prince R, Gaich GA, Reginster JY, Hodsman AB, Eriksen EF, Ish-Shalom S, Genant HK, Wang O, Mitlak BH (2001) Effect of parathyroid hormone (1–34) on fractures and bone mineral density in postmenopausal women with osteoporosis. N Engl J Med 344(19):1434–1441
Lindsay R, Nieves J, Formica C, Henneman E, Woelfert L, Shen V, Dempster D, Cosman F (1997) Randomised controlled study of effect of parathyroid hormone on vertebral-bone mass and fracture incidence among postmenopausal women on oestrogen with osteoporosis. Lancet 350(9077):550–555
Lane NE, Sanchez S, Modin GW, Genant HK, Pierini E, Arnaud CD (1998) Parathyroid hormone treatment can reverse corticosteroid-induced osteoporosis. Results of a randomized controlled clinical trial. J Clin Invest 102(8):1627–1633
Black DM, Greenspan SL, Ensrud KE, Palermo L, McGowan JA, Lang TF, Garnero P, Bouxsein ML, Bilezikian JP, Rosen CJ (2003) The effects of parathyroid hormone and alendronate alone or in combination in postmenopausal osteoporosis. N Engl J Med 349(13):1207–1215
Chen TC, Turner AK, Holick MF (1990) A method for the determination of the circulating concentration of 1,25-dihydroxyvitamin D. J Nutr Biochem 1(6):320–327
Bauer DC, Garnero P, Bilezikian JP, Greenspan SL, Ensrud KE, Rosen CJ, Palermo L, Black DM (2006) Short-term changes in bone turnover markers and bone mineral density response to parathyroid hormone in postmenopausal women with osteoporosis. J Clin Endocrinol Metab 91(4):1370–1375
Body JJ, Gaich GA, Scheele WH, Kulkarni PM, Miller PD, Peretz A, Dore RK, Correa-Rotter R, Papaioannou A, Cumming DC, Hodsman AB (2002) A randomized double-blind trial to compare the efficacy of teriparatide [recombinant human parathyroid hormone (1–34)] with alendronate in postmenopausal women with osteoporosis. J Clin Endocrinol Metab 87(10):4528–4535
Finkelstein JS, Hayes A, Hunzelman JL, Wyland JJ, Lee H, Neer RM (2003) The effects of parathyroid hormone, alendronate, or both in men with osteoporosis. N Engl J Med 349(13):1216–1226
Orwoll ES, Scheele WH, Paul S, Adami S, Syversen U, ez-Perez A, Kaufman JM, Clancy AD, Gaich GA (2003) The effect of teriparatide [human parathyroid hormone (1–34)] therapy on bone density in men with osteoporosis. J Bone Miner Res 18(1):9–17
Hodsman AB, Hanley DA, Ettinger MP, Bolognese MA, Fox J, Metcalfe AJ, Lindsay R (2003) Efficacy and safety of human parathyroid hormone-(1–84) in increasing bone mineral density in postmenopausal osteoporosis. J Clin Endocrinol Metab 88(11):5212–5220
Bauer DC, Black DM, Garnero P, Hochberg M, Ott S, Orloff J, Thompson DE, Ewing SK, Delmas PD (2004) Change in bone turnover and hip, non-spine, and vertebral fracture in alendronate-treated women: the fracture intervention trial. J Bone Miner Res 19(8):1250–1258
Acknowledgements
This study was funded by NIAMS (NIAMS-045, N01-AR-9-2245). Study medications were provided by NPS Pharmaceuticals (parathyroid hormone and matching placebo) and Merck & Co., Inc. (alendronate and matching placebo).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Sellmeyer, D.E., Black, D.M., Palermo, L. et al. Hetereogeneity in skeletal response to full-length parathyroid hormone in the treatment of osteoporosis. Osteoporos Int 18, 973–979 (2007). https://doi.org/10.1007/s00198-007-0336-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-007-0336-x