Abstract
Introduction
Recent studies suggest that bone marrow adipose tissue (BMAT) might play a role in the pathogenesis of osteoporosis. Previous research using regional magnetic resonance spectroscopy methods to measure BMAT has reported inconsistent findings on the relationship between BMAT and dual-energy absorptiometry (DXA)-measured bone mineral density (BMD).
Methods
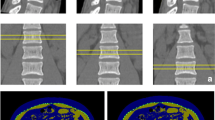
In the present study, total body and pelvic BMAT were evaluated in 56 healthy women (age 18–88 yrs, mean ± SD, 47.4 ± 17.6 yrs; BMI, 24.3 ± 4.2 kg/m2) with T1-weighted whole-body magnetic resonance imaging (MRI). BMD was measured using the whole-body DXA mode (GE Lunar DPX, software version 4.7).
Results
A strong negative correlation was observed between pelvic BMAT and BMD (total-body BMD, R = −0.743, P < 0.001; pelvic BMD, R = −0.646, P < 0.001), and between total-body BMAT and BMD (total-body BMD, R = −0.443, P < 0.001; pelvic BMD, R = −0.308, P < 0.001). The inverse association between pelvic BMAT and BMD remained strong after adjusting for age, weight, total body fat, and menopausal status (partial correlation: total-body BMD, R = −0.553, P < 0.001; pelvic BMD, R = −0.513, P < 0.001). BMAT was also highly correlated with age (pelvic BMAT, R = 0.715, P < 0.001; total-body BMAT, R = 0.519, P < 0.001).
Conclusion
MRI-measured BMAT is thus strongly inversely correlated with DXA-measured BMD independent of other predictor variables. These observations, in the context of DXA technical concerns, support the growing evidence linking BMAT with low bone density.



Similar content being viewed by others
References
Rosen CJ, Bouxsein ML (2006) Mechanisms of disease: is osteoporosis the obesity of bone? Nat Clin Pract Rheumatol 2:35–43
Beresford JN, Bennett JH, Devlin C, Leboy PS, Owen ME (1992) Evidence for an inverse relationship between the differentiation of adipocytic and osteogenic cells in rat marrow stromal cell cultures. J Cell Sci 102:341–351
Shih TT, Chang CJ, Hsu CY, Wei SY, Su KC, Chung HW (2004) Correlation of bone marrow lipid water content with bone mineral density on the lumbar spine. Spine 29:2844–2850
Wehrli FW, Hopkins JA, Hwang SN, Song HK, Snyder PJ, Haddad JG (2000) Cross-sectional study of osteopenia with quantitative MR imaging and bone densitometry. Radiology 217:527–538
Schick F, Duda SH, Lutz O, Claussen CD (1996) Lipids in bone tumors assessed by magnetic resonance: chemical shift imaging and proton spectroscopy in vivo. Anticancer Res 16:1569–1574
Schellinger D, Lin CS, Fertikh D, Lee JS, Lauerman WC, Henderson F, Davis B (2000) Normal lumbar vertebrae: anatomic, age, and sex variance in subjects at proton MR spectroscopy-initial experience. Radiology 215:910–916
Schellinger D, Lin CS, Hatipoglu HG, Fertikh D (2001) Potential value of vertebral proton MR spectroscopy in determining bone weakness. AJNR Am J Neuroradiol 22:1620–1627
Griffith JF, Yeung DK, Antonio GE, Lee FK, Hong AW, Wong SY, Lau EM, Leung PC (2005) Vertebral bone mineral density, marrow perfusion, and fat content in healthy men and men with osteoporosis: dynamic contrast-enhanced MR imaging and MR spectroscopy. Radiology 236:945–951
Vande Berg BC, Malghem J, Lecouvet FE, Maldague B (1998) Magnetic resonance imaging of normal bone marrow. Eur Radiol 8:1327–1334
Russell-Aulet M, Wang J, Thornton J, Pierson RNJ (1991) Comparison of dual-photon absorptiometry systems for total-body bone and soft tissue measurements: dual-energy X-rays versus gadolinium 153. J Bone Miner Res 6:411–415
Gallagher D, Belmonte D, Deurenberg P, Wang Z, Krasnow N, Pi-Sunyer FX, Heymsfield SB (1998) Organ-tissue mass measurement allows modeling of REE and metabolically active tissue mass. Am J Physiol 275:E249–E258
Heymsfield SB, Gallagher D, Kotler DP, Wang Z, Allison DB, Heshka S (2002) Body-size dependence of resting energy expenditure can be attributed to nonenergetic homogeneity of fat free mass. Am J Physiol Endocrinol Metab 282:E132–E138
Justesen J, Stenderup K, Ebbesen EN, Mosekilde L, Steiniche T, Kassem M (2001) Adipocyte tissue volume in bone marrow is increased with aging and in patients with osteoporosis. Biogerontology 2:165–171
Schellinger D, Lin CS, Lim J, Hatipoglu HG, Pezzullo JC, Singer AJ (2004) Bone marrow fat and bone mineral density on proton MR spectroscopy and dual-energy X-ray absorptiometry: their ratio as a new indicator of bone weakening. AJR Am J Roentgenol 183:1761–1765
Rickard DJ, Kassem M, Hefferan TE, Sarkar G, Spelsberg TC, Riggs BL (1996) Isolation and characterization of osteoblast precursor cells from human bone marrow. J Bone Miner Res 11:312–324
Gimble JM, Robinson CE, Wu X, Kelly KA (1996) The function of adipocytes in the bone marrow stroma: an update. Bone 19:421–428
Feller JF (2002) MRI of bone marrow: advanced MRI from head to toe. http://mri.cpson.com/pdf/MRI_of_the_Bone_Marrow.pdf. Cited 13 Nov 2006
Bolotin HH, Sievanen H, Grashuis JL (2003) Patient-specific DXA bone mineral density inaccuracies: quantitative effects of nonuniform extraosseous fat distributions. J Bone Miner Res 18:1020–1027
Hangartner TN, Johnston CC (1990) Influence of fat on bone measurements with dual energy absorptiometry. Bone Miner 9:71–81
Bathija A, Davis S, Trubowitz S (1979) Bone marrow adipose tissue: response to acute starvation. Am J Hematol 6:191–198
Greenberger JS (1979) Corticosteroid-dependent differentiation of human marrow preadipocytes in vitro. In Vitro 15:823–828
Kugel H, Jung C, Schulte O, Heindel W (2001) Age- and sex-specific differences in the 1H14 spectrum of vertebral bone marrow. J Magn Reson Imaging 13:263–268
WHO Study Group (1994) Technical report series, no. 843. Assessment of fracture risk and its application to screening for postmenopausal osteoporosis: report of a WHO study group. World Health Organization, Geneva
Lang T, Augat P, Majumdar S, Ouyang X, Genant HK (1998) Noninvasive assessment of bone density and structure using computed tomography and magnetic resonance. Bone 22:149S–153S
Mulkern RV, Huang J, Vajapeyam S, Packard AB, Oshio K, Grinspoon S (2004) Fat fractions and spectral T2 values in vertebral bone marrow in HIV-and non-HIV-infected men: a 1H spectroscopic imaging study. Magn Reson Med 52:552–558
Brix G, Heiland S, Bellemann ME, Koch T, Lorenz WJ (1993) MR imaging of fat-containing tissues: valuation of two quantitative imaging techniques in comparison with localized proton spectroscopy. Magn Reson Imaging 11:977–991
Gimble JM, Zvonic S, Floyd ZE, Kassem M, Nuttall ME (2006) Playing with bone and fat. J Cell Biochem 98:251–266
Acknowledgements
This study was supported by National Institutes of Health Grants NIDDK R21 DK66360-01, R01 DK42618.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Shen, W., Chen, J., Punyanitya, M. et al. MRI-measured bone marrow adipose tissue is inversely related to DXA-measured bone mineral in Caucasian women. Osteoporos Int 18, 641–647 (2007). https://doi.org/10.1007/s00198-006-0285-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00198-006-0285-9