Abstract
Introduction and hypothesis
Validated questionnaires are commonly used in research, but successful completion rates in clinical settings are largely unknown. The primary goal of this study was to assess the frequency of appropriate completion of a validated research survey. We secondarily examined relationships between demographics and successful questionnaire completion.
Methods
New patients completed a paper form of the Pelvic Floor Bother Questionnaire (PFBQ) to assess pelvic floor symptoms and level of bother. Various aspects of successful survey completion were assessed, including unanswered questions, affirmative responses without selection of a level of bother, or choosing a level of bother despite reporting not having a symptom. Relationships between self-reported demographic characteristics and completion of the survey were also evaluated.
Results
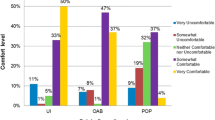
Five hundred and fourteen questionnaires were completed by a cohort of women with a mean age of 57.5 ± 14.4 years (range 19–97). Overall, 45.3% of women (n = 233) completed the entire PFBQ properly as originally described. Women skipped at least one entire question 16.5% of the time. On logistic regression, older age was significantly associated with improper questionnaire completion (55.0 years ±14.9 successful vs 59.6 years ±13.6 unsuccessful completion, p < 0.001 for overall completion). Age was significantly associated with proper completion of every individual PFBQ question, except question 8 concerning fecal incontinence (p = 0.06). Education level was not significantly associated with successful questionnaire completion.
Conclusions
Overall successful completion of the survey as designed and validated was low, even in a highly educated population. Ways to simplify the instrument to enhance completion, such as electronic smart questionnaires, should be further investigated.

Similar content being viewed by others
References
Peterson TV, Karp DR, Aguilar VC, Davila GW. Validation of a global pelvic floor symptom bother questionnaire. Int Urogynecol J. 2010;21(9):1129–35.
Ebert JF, Huibers L, Christensen B, Christensen MB. Paper- or web-based questionnaire invitations as a method for data collection: cross-sectional comparative study of differences in response rate, completeness of data, and financial cost. J Med Internet Res. 2018;20(1):e24.
Brubaker L, Litman HJ, Kim HY, Zimmern P, Dyer K, Kusek JW, et al. Missing data frequency and correlates in two randomized surgical trials for urinary incontinence in women. Int Urogynecol J. 2015;26(8):1155–9.
Kelley K, Clark B, Brown V, Sitzia J. Good practice in the conduct and reporting of survey research. Int J Quality Health Care. 2003;15(3):261–6.
Little RJ, D’Agostino R, Cohen ML, Dickersin K, Emerson SS, Farrar JT, et al. The prevention and treatment of missing data in clinical trials. NEJM. 2012;367(14):1355–60.
Funding
This was an unfunded study that received exempt determination status from The University of Michigan Health Sciences and Behavioral Sciences Institutional Review Board (HUM00131050).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
Nones.
Rights and permissions
About this article
Cite this article
Berger, M.B., Schimpf, M.O. Completion of a validated pelvic floor symptom and bother instrument in real-life practice. Int Urogynecol J 29, 1841–1844 (2018). https://doi.org/10.1007/s00192-018-3692-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00192-018-3692-5