Abstract
Introduction and hypothesis
Botulinum toxin-A (BoNT-A) is a potent neurotoxin that is an effective treatment for patients with pharmacologically refractory detrusor overactivity (DO). Data assessing the effectiveness of trigonal BoNT-A are limited. This study evaluates adverse events (AEs) and short-term efficacy associated with trigonal and extratrigonal BoNT-A.
Methods
Electronic databases (PubMed, EMBASE, and the Cochrane database) were searched for studies comparing trigonal and extratrigonal BoNT-A for DO. Meta-analyses were performed using the random effects model. Outcome measures included incidence of AEs and short-term efficacy.
Results
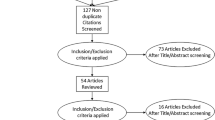
Six studies describing 258 patients met the inclusion criteria. The meta-analysis did not show significant differences between trigonal and extratrigonal BoNT-A for acute urinary retention (AUR; 4.2 vs 3.7 %; odds ratio [OR]: 1.068, 95 % confidence interval [CI]: 0.239–4.773; P = 0.931) or high post-void residual (PVR; 25.8 vs 22.2 %; OR: 0.979; 95 % CI: 0.459–2.088; P = 0.956). The incidence of urinary tract infection (UTI; 7.5 vs 21.0 %; OR: 0.670; 95 % CI: 0.312–1.439; P = 0.305), haematuria (15.8 vs 25.9 %; OR: 0.547; 95 % CI: 0.264–1.134; P = 0.105) and post-operative muscle weakness (9.2 vs 11.3 %; OR: 0.587; 95 % CI: 0.205–1.680, P = 0.320) was similar in both groups. Finally, differences in short-term cure rates between two study arms were not statistically significant (52.9 vs 56.9 %; OR: 1.438; 95 % CI: 0.448–4.610; P = 0.542).
Conclusions
Although data are limited, no significant differences between trigonal and extratrigonal BoNT-A in terms of AEs and short-term efficacy were observed. Additional randomised controlled trials are required to define optimal injection techniques and sites for administering intra-vesical BoNT-A.



Similar content being viewed by others
Abbreviations
- AEs:
-
Adverse events
- AUR:
-
Acute urinary retention
- BoNT-A:
-
Botulinum toxin-A
- DO:
-
Detrusor overactivity
- DSD:
-
Detrusor sphincter dysynergia
- IDO:
-
Idiopathic detrusor overactivity
- NDO:
-
Neurogenic detrusor overactivity
- OAB:
-
Overactive bladder
- QoL:
-
Quality of life
- SIC:
-
Self intermittent catheterisation
- UTI:
-
Urinary tract infection
- VUR:
-
Vesicoureteric reflux
References
Mangera A, Andersson KE, Apostolidis A, Chapple C, Dasgupta P et al (2011) Contemporary management of lower urinary tract disease with botulinum toxin A: a systematic review of botox (onabotulinumtoxinA) and dysport (abobotulinumtoxinA). Eur Urol 60:784–795
Kuo HC (2011) Bladder base/trigone injection is safe and as effective as bladder body injection of onabotulinumtoxinA for idiopathic detrusor overactivity refractory to antimuscarinics. Neurourol Urodyn 30:1242–1248
Karsenty G, Baverstock R, Carlson K, Diaz DC, Cruz F et al (2014) Technical aspects of botulinum toxin type A injection in the bladder to treat urinary incontinence: reviewing the procedure. Int J Clin Pract 68:731–742
Ju R, Garrett J, Wu JM (2014) Anticholinergic medication use for female overactive bladder in the ambulatory setting in the United States. Int Urogynecol J 25:479–484
Tubaro A (2004) Defining overactive bladder: epidemiology and burden of disease. Urology 64:2–6
Karsenty G, Elzayat E, Delapparent T, St-Denis B, Lemieux MC et al (2007) Botulinum toxin type a injections into the trigone to treat idiopathic overactive bladder do not induce vesicoureteral reflux. J Urol 177:1011–1014
Schurch B, Schmid DM, Stohrer M (2000) Treatment of neurogenic incontinence with botulinum toxin A. N Engl J Med 342:665
DerSimonian R, Laird N (1986) Meta-analysis in clinical trials. Control Clin Trials 7:177–188
Dunne C, Burke JP, Morrow M, Kell MR (2009) Effect of margin status on local recurrence after breast conservation and radiation therapy for ductal carcinoma in situ. J Clin Oncol 27:1615–1620
Lau J, Ioannidis JP, Schmid CH (1997) Quantitative synthesis in systematic reviews. Ann Intern Med 127:820–826
Manecksha RP, Cullen IM, Ahmad S, McNeill G, Flynn R et al (2012) Prospective randomised controlled trial comparing trigone-sparing versus trigone-including intradetrusor injection of abobotulinumtoxinA for refractory idiopathic detrusor overactivity. Eur Urol 61:928–935
Mascarenhas F, Cocuzza M, Gomes CM, Leao N (2008) Trigonal injection of botulinum toxin-A does not cause vesicoureteral reflux in neurogenic patients. Neurourol Urodyn 27:311–314
Lucioni A, Rapp DE, Gong EM, Fedunok P, Bales GT (2006) Intravesical botulinum type A toxin injection in patients with overactive bladder: trigone versus trigone-sparing injection. Can J Urol 13:3291–3295
Abdel-Meguid TA (2010) Botulinum toxin-A injections into neurogenic overactive bladder–to include or exclude the trigone? a prospective, randomized, controlled trial. J Urol 184:2423–2428
Moher D, Liberati A, Tetzlaff J, Altman DG (2009) Preferred reporting items for systematic reviews and meta-analyses: the PRISMA statement. PLoS Med 6:e1000097
Dykstra DD, Sidi AA, Scott AB, Pagel JM, Goldish GD (1988) Effects of botulinum A toxin on detrusor-sphincter dyssynergia in spinal cord injury patients. J Urol 139:919–922
Herschorn S, Gajewski J, Ethans K, Corcos J, Carlson K et al (2011) Efficacy of botulinum toxin A injection for neurogenic detrusor overactivity and urinary incontinence: a randomized, double-blind trial. J Urol 185:2229–2235
Flynn MK, Amundsen CL, Perevich M, Liu F, Webster GD (2009) Outcome of a randomized, double-blind, placebo controlled trial of botulinum A toxin for refractory overactive bladder. J Urol 181:2608–2615
Acknowledgements
None
Funding
None.
Financial disclaimer/conflict of interest
None.
Author participation
Niall Davis: collection of data, manuscript writing; John Burke: statistical analysis; Elaine Redmond: collection of data; Saif Elamin: collection of data; Ciaran Brady: manuscript editing; Hugh Flood: project design, manuscript editing.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Davis, N.F., Burke, J.P., Redmond, E.J. et al. Trigonal versus extratrigonal botulinum toxin-A: a systematic review and meta-analysis of efficacy and adverse events. Int Urogynecol J 26, 313–319 (2015). https://doi.org/10.1007/s00192-014-2499-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00192-014-2499-2