Abstract
Treatment strategies for anterior cruciate ligament (ACL) injuries continue to evolve. Evidence supporting best practice guidelines for the management of ACL injury is to a large extent based on studies with low-level evidence. An international consensus group of experts was convened to collaboratively advance toward consensus opinions regarding the best available evidence on operative vs. non-operative treatment for ACL injury. The purpose of this study is to report the consensus statements on operative vs. non-operative treatment of ACL injuries developed at the ACL Consensus Meeting Panther Symposium 2019. Sixty-six international experts on the management of ACL injuries, representing 18 countries, were convened and participated in a process based on the Delphi method of achieving consensus. Proposed consensus statements were drafted by the Scientific Organizing Committee and Session Chairs for the three working groups. Panel participants reviewed preliminary statements prior to the meeting and provided the initial agreement and comments on the statement via an online survey. During the meeting, discussion and debate occurred for each statement, after which a final vote was then held. Eighty percent agreement was defined a-priori as consensus. A total of 11 of 13 statements on operative v. non-operative treatment of ACL injury reached the consensus during the Symposium. Nine statements achieved unanimous support, two reached strong consensus, one did not achieve consensus, and one was removed due to redundancy in the information provided. In highly active patients engaged in jumping, cutting, and pivoting sports, early anatomic ACL reconstruction is recommended due to the high risk of secondary meniscus and cartilage injuries with delayed surgery, although a period of progressive rehabilitation to resolve impairments and improve neuromuscular function is recommended. For patients who seek to return to straight plane activities, non-operative treatment with structured, progressive rehabilitation is an acceptable treatment option. However, with persistent functional instability, or when episodes of giving way occur, anatomic ACL reconstruction is indicated. The consensus statements derived from international leaders in the field will assist clinicians in deciding between operative and non-operative treatments with patients after an ACL injury.
Level of evidence V.
Similar content being viewed by others
Introduction
Anterior cruciate ligament (ACL) injuries are one of the most common injuries of the knee, with an incidence of approximately 85 per 100 000 in patients aged between 16 and 39 years [26, 36, 49]. The ACL is the primary stabilizer of the knee limiting anterior tibial translation and internal rotation, with deficiency resulting in anterior and rotatory instability [54, 91]. The commonest mode of injury is a non-contact mechanism during pivoting, cutting, and jumping with the knee slightly flexed and in a valgus position [1, 5].
Both operative and non-operative treatments of an ACL injury continue to evolve [21, 22, 32, 78]. Improved understanding of the structure and function of the native ACL has supported the development and adoption of anatomic ACL reconstruction techniques [3]. In parallel, increased recognition of the resilience of the neuromuscular system in achieving dynamic, functional knee stability despite ACL deficiency has concurrently supported non-operative treatment as a viable strategy in some patients [13, 23].
Successful outcomes following both operative and non-operative treatment necessitate progressive rehabilitation, which entails staged and phase-adjusted physical therapy with the aim to address impairments, achieve functional stability, and to safely return to sport [64]. The acute phase after the injury or surgery focuses on the elimination of residual symptoms (effusion, pain) and impairments (range of motion, quadriceps activation, and strength). Subsequently, neuromuscular and perturbation training are implemented to improve knee stabilization [9, 19]. The last phase aims to further optimize muscular strength, return to pre-injury sports level through sport-specific exercises, and assess psychological readiness for the return to sport [3]. Any discussion of non-operative treatment within this consensus document implies the completion of a progressive, staged rehabilitation protocol.
Similarly, any discussion of operative treatment implies anatomic ACL reconstruction (Table 1), which intends to restore the ACL to its native dimensions, collagen orientation, and insertion sites [83]. Anatomic ACL reconstruction includes both single- and double-bundle techniques, followed by a progressive rehabilitation program that considers the natural healing cascade and ligamentization of the graft [65]. Following fixation during ACL reconstruction, biological graft transitions from a tendon to a structure with ultrastructural, biochemical, and mechanical properties more similar to the native ACL [74]. These properties of the graft depend on the phase of ligamentization, with the minimum graft strength occurring between 4 and 12 weeks postoperatively [65, 74]. Comprehensive rehabilitation after operative ACL reconstruction is also paramount for clinical outcome and return to sports.
Whereas operative treatment aims to reduce laxity, non-operative treatments aim to reduce functional instability and both thereby prevent further damage to the menisci and cartilage, which may contribute to post-traumatic osteoarthritis [58, 84]. Functional bracing, intended to reduce the risk of ACL injury by decreasing peak ligament strain, has not yet been conclusively shown to achieve this goal, as the evidence is still limited [29, 75].
There is still uncertainty as to which patients should undergo immediate surgery and which patients may be successfully treated non-operatively. Three different patient responses after ACL injury have been described: (1) a coper can return to the pre-injury level without surgery and subjective instability; (2) an adapter reduces his/her level of activity to avoid subjective instability; (3) a non-coper cannot return to pre-injury activity level due to subjective instability and episodes of giving way [61]. A screening tool to differentiate potential copers from non-copers was developed and included a combination of hop tests, questionnaires on general knee function, and the frequency of giving-way episodes [18, 60]. Patients categorized as potential copers thereafter participated in structured progressive rehabilitation with additional perturbation training [9, 19]. Regardless of this three-response concept, there is a strong historical view that the treatment approach should be determined through a shared decision-making process between the patient and the provider [8]. In particular, the physician should share information on the evidence-based treatment options while also considering the patient’s expectations and goals. While the patient and provider are the primary stakeholders in the shared decision-making process, the potential influence of secondary stakeholders, such as family and coaches, should be anticipated so as to minimize interests potentially conflicting with the health of the patient.
Taken as a whole, the current body of evidence regarding the treatment of ACL injury is to a large extent based on low level of evidence. Therefore, an international, multidisciplinary group of experts was assembled to develop expert- and evidence-based consensus statements to assist clinicians in managing this difficult pathology. The purpose of this article is to report the results of the consensus group addressing the best available evidence on operative vs. non-operative treatments of ACL injury that were developed at the 2019 Panther Symposium ACL consensus meeting.
Materials and methods
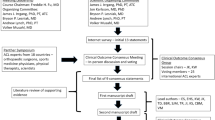
An international and multidisciplinary group of experts of ACL injury, including orthopedic surgeons, sports medicine physicians, physical therapists, and scientists, were convened in a 1-year consensus-building effort, which culminated in the consensus meeting, at the University of Pittsburgh, PA, USA (Table 2). The symposium included experts from 18 countries, spanning six continents. Experts were assigned to one or more, of the three consensus groups defined by a specific subtopic within ACL injury. The operative vs. non-operative treatment consensus groups consisted of 34 participants. A modified Delphi method was used to develop the consensus statements.
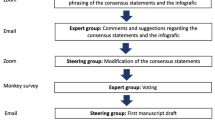
The scientific organizing committee and session chairs proposed a series of statements on the basis of a literature review. These were drafted with the aim of addressing areas of current controversy within the treatment of ACL injury, intended to assist clinicians in the management of this injury. Prior to the meeting, the proposed statements were presented to the panelists via a web-based survey. Each panelist indicated the extent of agreement or disagreement with each statement, and was asked to provide comments on each statement. On the third day of the 2019 Panther Symposium, after 2 days of presentations by symposium delegates on the current knowledge, a consensus discussion was held.
A total of 13 statements on the operative vs. non-operative treatment of ACL injury were discussed. The session was moderated by two experts (LE and ADL). Initial results and comments from the web-based survey were presented for each statement followed by discussion, debate, and revision by the working group. The consensus was determined by a show of hands. Satisfactory consensus was defined as 80% agreement. Opposing views were documented and discussed. Statements with less than 80% agreement were included in the consensus paper, noting the percentage of agreement. Statements felt to be irrelevant or redundant were excluded from this final paper.
This consensus group was assigned two liaisons (TD and BBR) who were responsible for amending each statement as requested over the course of the discussion. Liaisons transcribed the discussion and subsequently completed a literature review of MEDLINE for each finalized statement. To reduce the potential for bias in the data analysis and/or literature review, liaisons did not submit answers to the online questionnaire nor did they partake in the voting process.
Results
Of the 13 statements discussed by this working group, nine achieved unanimous consensus, two achieved non-unanimous consensus, one did not achieve consensus, and one was excluded due to redundancy in the information provided (Table 3). The 12 finalized statements, with supporting literature, are as follows.
Operative and non-operative treatments are both acceptable treatment options for ACL injury.
Agree 23/23, 100%
After ACL injury, some patients are able to regain good functional knee stability following non-operative treatment entailing progressive rehabilitation and are able to return to pre-injury sports activity level without an ACL reconstruction (copers) [27, 28], but the identification of these patients has been challenging [80]. In a prospective study, the combination of hop tests, muscle strength, subjective instability (episodes of giving way), and knee function was found to be a moderate predictive tool for the identification of potential copers [18, 28, 32, 60]. A randomized-controlled trial comparing operative and non-operative treatments in 121 young active, non-elite patients with isolated ACL tears demonstrated no superiority of either treatment with regard to patient-reported outcomes at 2- and 5-year follow-up [21, 22]. However, almost 40% of the patients who were initially assigned to the non-operative treatment group required delayed ACL reconstruction and 32% of the patients (29 menisci in 19 patients) had subsequent surgery for meniscal pathology during the 2 year follow-up period. In contrast, 34 patients (56%) who underwent the early ACL reconstruction also had meniscus treatment (24 partial resection and 10 fixation) simultaneous with the ACL reconstruction, but only 10% (6 meniscal injuries in 5 patients) in the operatively treated group had meniscal injuries that required surgical treatment during follow-up [21]. With regard to knee laxity, as measured by KT-1000 and pivot shift test, non-operative treatment resulted in a larger anterior tibial translation (9.0 mm vs. 6.6 mm) and higher rate of rotatory laxity (positive pivot shift test: 78% vs. 25%). A matched-paired study based on the Swedish National ACL registry comparing operative and non-operative treatments after ACL injury reported superior results for quality of life, knee function, and symptoms at 1, 2, and 5 year follow-up for ACL reconstruction compared with non-operative treatment [40]. Another prospective trial with highly active patients included 832 patients at baseline with sub-acute ACL tear, whereas 345 patients were initially screened for the possibility of non-operative treatment. Based on the results of various hop tests, subjective instability, and general knee function, 146 patients were classified as potential copers, and at the final follow-up after 10 years, only 25 patients had not undergone ACL reconstruction [32].
Conclusion: Operative and non-operative are both acceptable treatment options after ACL injury, and a decision based on concomitant injuries, risk factors, level of activity, and patient’s expectations and goals is recommended as demonstrated in the following statements.
Operative versus non-operative treatment should be reached via a shared decision-making process that considers the patient’s presentation, goals, and expectations as well as a balanced presentation of the available evidence-based literature.
Agree 19/23, 82.6%
Before a particular treatment approach is pursued, the provider (physician and/or physical therapist) should present the evidence for operative and non-operative treatment options for an ACL injury to the patient. Based on the patient’s activity level, goals, and expectations, a decision should be made with the patient (and parents/guardians for minors) and provider as the primary stakeholders [8]. Physicians and physical therapists must be aware that personal and situational factors, such as level of competition, time in season, playing status, and role in the team, could affect the injured athlete's treatment decision. Parents and coaches are often the first individuals from whom athletes seek support or advice [59]. However, the coach may be conflicted by the interests of the team and the athlete’s immediate and future health [20, 33]. For some athletes, reactions and comments of parents related to the athlete’s injury were reported to negatively affect the athlete's treatment decision, with pressure to return to sport [59]. Due to the possible conflict of interest, secondary stakeholders such as family, coaches, and agents, among others, should not be directly involved in the decision-making process, although their indirect involvement may be considered.
Conclusion: Shared decision-making of the treatment option should be based on the evidence for operative and non-operative treatments, patient’s expectations and goals with the provider, and patient as the primary stakeholders.
The (injury) status of other stabilizing and supporting structures (e.g. meniscus, other ligaments, cartilage) affects the decision to pursue operative or non-operative treatment.
Agree 23/23, 100%
ACL injuries often occur together with concomitant injury to other knee structures, with meniscal injuries reported in 23–42%, cartilage lesions in 27%, and combined meniscal and chondral lesions in 15% of cases (Fig. 1) [6, 11, 41].
However, most studies investigating non-operative ACL treatment or studies comparing non-operative and operative treatment are limited to isolated ACL tears [21, 22, 32]. Based on clinical and biomechanical studies, an ACL reconstruction with concomitant meniscus repair may restore knee kinematics and results in improved patient-reported outcomes at short- and long-term follow-up [47, 69, 73, 90]. In contrast, simultaneously performed meniscectomy with ACL reconstruction is associated with poorer clinical outcomes, inferior knee kinematics, and a high rate (48–100%) of osteoarthritis in the long-term follow-up [12, 30, 50, 53, 89]. In case of delayed ACL reconstruction, a meniscectomy is more often performed than a meniscus repair [39]. The presence of concomitant knee injuries should, therefore, always be considered in the decision-making process, given the worse outcomes for meniscus injuries with delayed ACL reconstruction and higher rate of osteoarthritis in the long-term follow-up. In case of concomitant meniscus injury, anatomic ACL reconstruction with additional treatment of the meniscus injury is recommended.
In case of multiple ligament injuries involving the ACL and at least one other ligament, the literature has consistently demonstrated that operative management is superior to non-operative management [45, 66, 71]. Based on a recent systematic review, early (within 3 weeks after injury) reconstruction in a multiple ligament-injured knee was superior to delayed reconstruction with regard to clinical outcome measurements (Lysholm score, 90 vs. 82 out of 100 points) and resulted in higher rate of excellent/good IKDC scores (47% vs. 31%) [45]. Although failure after ligament reconstruction is not consistently defined in the literature (i.e., the need for revision vs. objective laxity vs. re-rupture on imaging vs. KOOS score < 44), the failure rate in a multiple ligament-injured knee is lower for reconstruction (6–9%) compared with repair techniques (37–40%) [44, 78].
Conclusion: The presence of a repairable meniscal lesion or a multiple ligament injury is an indication for an early anatomic ACL reconstruction with concomitant treatment of the other injured structures (meniscus repair and ligament repair/augmentation).
Individual anatomical differences (e.g., tibial slope, femoral morphology, alignment, etc.) may affect the stability of the knee after ACL injury and should be considered in the decision-making process for operative versus non-operative treatment.
Agree 22/23, 95.7%
Bony morphology and soft tissue injury patterns have been demonstrated to influence knee joint laxity. An increased posterior tibial slope is associated with increased anterior tibial translation, as well as with increased rotatory instability (Fig. 2) [70, 86]. In addition, an increased lateral femoral condyle ratio resulted in increased rotatory instability [67, 68]. Severe varus limb alignment (> 5°) was demonstrated to increase the risk for more rapid degeneration of the medial compartment in the ACL-deficient knee, and is also a risk factor for secondary failure after an ACL reconstruction [34, 62]. Whereas lateral meniscus tears and a complete lateral meniscectomy result in increased rotatory instability [31, 55], a complete medial meniscectomy more strongly affects anterior tibial translation. However, general joint laxity (Beighton hypermobility score > 4) is not associated with increased rotatory laxity in the ACL-deficient knee [79].
Conclusion: Bony morphology features (increased posterior tibial slope, severe varus limb alignment, etc.) and concomitant injuries associated with increased or persistent knee instability should be considered in the decision-making process and are a relative indication for operative treatment.
After an ACL injury, patients may be offered a period of progressive rehabilitation to improve impairments and improve overall function.
Agree 23/23, 100%
Knee joint effusion, limited range of motion, and decreased quadriceps strength in the injured leg are common impairments initially after an ACL injury [10, 48]. Effusion can limit quadriceps function and in turn affect knee joint mechanics [63]. Progressive rehabilitation is useful in treating these initial impairments [32]. In patients with the possibility of non-operative treatment (absence of concomitant meniscus injuries or multi-ligament injuries requiring surgical treatment) before the evaluation of knee instability, a phase of rehabilitation is recommended to treat the initial impairments. Afterward, evaluation by hop tests, assessment of strength, overall knee function, and subjective instability are recommended to quantify the patient's potential for non-operative treatment. If progressive rehabilitation does not provide a satisfactory outcome, then operative intervention needs to be pursued and the progressive rehabilitation will have enhanced the post-surgical outcome [14]. In a cohort study with 2,187 patients after the resolution of impairments, one group was treated with neuromuscular training (i.e., strengthening and neuromuscular training) before ACL reconstruction and was compared to immediate ACL reconstruction. At 2-year follow-up, preoperative progressive rehabilitation before ACL reconstruction resulted in better patient-reported outcome (KOOS and IKDC), compared with ACL reconstruction without preoperative rehabilitation [14]. Whereas 63% of the patients without preoperative rehabilitation returned to sport at 2-year follow-up, which is similar to the reported rate (65%) in a meta-analysis from 2016, the rate increased to 72% in the group that completed preoperative rehabilitation [14].
Conclusion: Preoperative resolution of impairments and a period of rehabilitation is recommended for operative and non-operative treatments.
An individual presenting with instability in their desired activity despite optimal rehabilitation should be referred for operative treatment.
Agree 23/23, 100%
Persistent instability is a risk factor for further damage to the meniscus and cartilage [35]. Although the definitions of recurrent instability and episodes of instability vary in the current literature, a correlation between persistent and recurrent instability after ACL injury and meniscus and cartilage lesions has been demonstrated in several studies [2, 38, 77]. In a cohort study of 62 patients with acute ACL reconstructions, 37 with sub-acute ACL reconstructions, and 36 with chronic ACL reconstructions, one episode of giving way was associated with threefold higher odds for lateral meniscus tears. Timing of surgery and episodes of instability influenced the incidence of lateral meniscus tears with 1.45 higher odds in sub-acute (6–12 weeks) ACL reconstruction and 2.82 higher odds in chronic (> 12 weeks) ACL reconstruction [2]. Moreover, frequent episodes of instability are correlated with medial meniscus tears and chondral injuries [38]. Chondral defects and meniscectomy have been demonstrated as predictive factors for the development of osteoarthritis after ACL reconstruction [15, 37].
A partial ACL injury progressed to a complete ACL tear in 39% of young active patients treated non-operative, with half of the complete tears presenting with a concomitant meniscal lesion at the time of reconstruction. Age ≤ 20 years and participation in pivoting contact sports were identified as significant risk factors for progression to a complete tear [16].
Conclusion: If patient-reported instability or severe episodes of giving way occur during the progressive rehabilitation, patients should be referred for anatomic ACL reconstruction.
Development of osteoarthritis after an ACL injury is multifactorial and evidence is inconclusive following operative or non-operative treatment.
Agree 23/23, 100%
Osteoarthritis is the most common joint disease, affecting not only the cartilage, but all other tissues of the joint as well [24]. The pathomechanism of post-traumatic osteoarthritis (PTOA) has not been fully elucidated, but based on current research, the process of development of osteoarthritis is multifactorial [24]. Injuries, like ACL ruptures, can affect the joint biomechanics and cause chondral and meniscal lesions, and thereby reduce the sustainability of the joint. Matrix metalloproteases are responsible for cartilage destruction and synovial inflammation, and have been shown to be elevated following ACL injury and reconstruction [81, 85]. A meta-analysis of 24 observational studies found a fourfold increased risk for PTOA after knee injuries, although the definition of an injury was largely heterogeneous among the analyzed studies [56]. After ACL injury, the prevalence of PTOA is increased after both operative and non-operative treatments as compared to those without injury [17, 51, 58, 72]. Based on a recent systematic review with 41 included studies, the rate of OA after ACL reconstruction varied between 1 and 80%, with meniscectomy as the consistent risk factor for the development of OA [46]. Although long-term outcome studies after ACL reconstruction are available, the technique has evolved in the recent years, with a shift from non-anatomic ACL reconstruction to anatomic ACL reconstruction, limiting conclusions on the possible protective effect of anatomic ACL reconstruction.
Conclusion: Osteoarthritis after ACL injury is seen after both operative and non-operative treatments. Therefore, there is still a need for prospective, randomized-controlled trials to evaluate the hypothesized preventative effect of anatomic ACL reconstruction on the development of post-traumatic osteoarthritis.
In active patients wishing to return to jumping, cutting, and pivoting sports (e.g., soccer, football, handball, basketball):
Operative treatment is the preferred option to maintain athletic participation in the medium-to-long term (1–5+ years after injury).
Agree 23/23, 100%
In active patients wishing to return to pivoting and cutting sports, ACL reconstruction is the preferred treatment option to maintain participation in the medium-to-long term. However, overall, only 65% of patients return to their pre-injury sports level after ACL reconstruction and only 55% return to competitive level sport [4]. Although the exact reasons are still unknown, younger age, male gender, professional sports level, and positive psychological response were demonstrated to be associated with a successful return to pre-injury sports level after ACL reconstruction. In general, elite athletes return to their pre-injury level of sports after ACL reconstruction more often than recreational athletes [42, 88]. For instance, over 90% of elite soccer players were reported to return to the pre-injury level after ACL reconstruction [88]. Similarly, in a recent systematic review, the return to sport rate in elite football and basketball players was 78% and 82%, respectively [42]. In contrast, only 12.8% of high-level athletes returned to the pre-injury sports level with non-operative treatment, with a high rate of the secondary meniscus and cartilage damage; after 20 years, 95% of the patients underwent meniscectomy, during which 68% of patients were found to have chondral lesions [18, 57]. Overall, athletes returned to their pre-injury sports level between 6 and 13 months after ACL reconstruction [42].
Conclusion: In active patients, anatomic ACL reconstruction is the preferred treatment due to the higher rate of return to pre-injury sports level.
In active patients wishing to return to jumping, cutting and pivoting sports (e.g., soccer, football, handball, basketball):
Return to cutting and pivoting sports without surgery places the knee at risk of secondary injury (meniscus, cartilage, etc.).
Agree 23/23, 100%
In a prospective randomized-controlled trial, patients with high activity levels (median Tegner activity score of 9) with isolated ACL tears received the early operative treatment or non-operative treatment with the option of delayed ACL reconstruction. Although no differences were evident for patient-reported outcomes, at 2-year follow-up, patients in the “optional” operative treatment group had more self-reported and clinical laxity of the involved knee and more meniscal surgery over a 5-year follow-up period [21]. In a separate cohort, the risk for sustaining at least one additional intra-articular injury increased by 0.6% with each month of delay in operative treatment [7]. The odds of secondary cartilage lesions increased by nearly 1% for each month of delay [25]. A delay in ACL reconstruction of at least 12 months almost doubled the risk for meniscal tears [7, 43]. Increased risk of secondary injury is especially noted in young (< 12 years) and skeletally immature patients [2].
Conclusion: Non-operative treatment increases the risk for secondary injuries if the patient wants to return to jumping, cutting and pivoting sports, due to the increased risk of further episodes of instability.
In active patients wishing to return to cutting and pivoting sports (e.g. soccer, football, handball, basketball):
Delayed operative treatment may be an option for temporary return to athletic participation following non-operative treatment accepting the risk of additional injury.
Agree 10/23, 43.4%
No consensus was reached for this statement. Some professional athletes and active patients want to delay ACL reconstruction to temporarily return to athletic participation (competition). Based on the current evidence, the risk of secondary damage to the knee (e.g., meniscus and cartilage) is high, especially in high-demand sports with jumping, cutting and pivoting. In a recent cross-sectional study, 860 patients were included with 47.2% being professional athletes. With regard to the prevalence of meniscus tears, medial, lateral, and combined lesions were found more often with increasing time from injury (TFI) to surgery (medial meniscus tear prevalence at 0–36-week TFI was 48.2% and when > 61 weeks was 59.3%). Not only did the prevalence of injury increase with time, the rate of meniscectomy also increased (medial meniscectomy at 0–36-week TFI was 7.5%, and when TFI was > 61 weeks, it was 12.8%) [76]
Conclusion: Delayed ACL reconstruction in active patients may be a treatment option, but the provider, as well as the patient, must be aware of the risk of secondary injuries with worse long-term outcomes.
In active patients wishing to return to straight plane activities (e.g., running, cycling, swimming, weight-lifting, etc.): Non-operative treatment is an option.
Agree 23/23, 100%
Straight plane activities are less demanding on the ligamentous stabilizers of the knee and, therefore, are amenable to non-operative treatment. The anteroposterior stability during straight plane activities might be maintained by muscular control, but coronal and rotational stability could not be compensated [87]. With specific neuromuscular training (perturbation training) additional to standard rehabilitation, unphysiological muscular co-contractions during walking can be minimized and normalized the knee kinematics in the ACL-deficient knee [9]. In a matched-paired study, non-operative treatment resulted in an earlier return (non-operative 3–4 months vs. operative 6–12 months) and a higher return to level II sports (non-operative 88.9% vs. operative 77.8%) as compared to operative treatment [28]. Another study demonstrated a significantly higher number of non-operative-treated patients returned to level II and level III sports compared to operative treatment [27].
Conclusion: For return to straight plane activities, non-operative treatment is an option.
In active patients wishing to return to straight plane activities (e.g., running, cycling, swimming, weight-lifting etc.):
In the case of persistent instability in daily life, operative treatment is appropriate for a return to non-rotational activities.
Agree 23/23, 100%
Straight plane activities are less demanding to the ligamentous stabilizers of the knee and are, therefore, amenable to non-operative treatment. If during the non-operative treatment, subjective instability persists or episodes of giving way occur, referral for consideration of anatomic ACL reconstruction is recommended [21, 52]. Moreover, the current evidence for the efficacy of non-operative treatment is limited to isolated ACL tears.
Conclusion: Based on the current evidence, persistent instability in activities of daily living is an indication for anatomic ACL reconstruction to restore knee laxity and prevent secondary injuries.
Conclusion
The expert panel at the ACL Consensus Meeting Panther Symposium 2019 reached consensus, defined as > 80% agreement, on 11 of 12 statements in terms of operative vs. non-operative treatments for ACL injuries. Consensus was reached that both treatment options may be acceptable, depending on patient characteristics, including the type of sporting demands and the presence of concomitant injuries. In highly active patients engaged in jumping, cutting, and pivoting sports, the early anatomic ACL reconstruction is recommended due to the high risk of secondary meniscus and cartilage injuries with delayed surgery, although a period of progressive rehabilitation to resolve impairments and improve neuromuscular function may be recommended. For patients who want to return to straight plane activities, non-operative treatment with structured, progressive rehabilitation is an acceptable treatment option. However, with persistent functional instability, or episodes of giving way occur, anatomic ACL reconstruction is indicated.
Despite strong consensus by experts, there is a need for larger randomized trials with longer term follow-up in which the early surgery (followed by rehabilitation) is compared with a strategy of early rehabilitation and delayed surgery. There are insufficient data to guide treatment in instances when there are concomitant meniscal and collateral ligament injuries. Data on long-term clinical outcomes are needed to better understand the effect of ACL treatment of injuries, subsequent injuries to meniscus and cartilage, and the development of osteoarthritis.
Change history
25 September 2020
The author decided to opt for Open Choice and to make the article an Open Access publication.
References
Alentorn-Geli E, Myer GD, Silvers HJ, Samitier G, Romero D, Lazaro-Haro C et al (2009) Prevention of non-contact anterior cruciate ligament injuries in soccer players. Part 1: mechanisms of injury and underlying risk factors. Knee Surg Sports Traumatol Arthrosc 17:705–729
Anderson AF, Anderson CN (2015) Correlation of meniscal and articular cartilage injuries in children and adolescents with timing of anterior cruciate ligament reconstruction. Am J Sports Med 43:275–281
Ardern CL, Österberg A, Tagesson S, Gauffin H, Webster KE, Kvist J (2014) The impact of psychological readiness to return to sport and recreational activities after anterior cruciate ligament reconstruction. Br J Sports Med 48:1613–1619
Ardern CL, Taylor NF, Feller JA, Webster KE (2014) Fifty-five per cent return to competitive sport following anterior cruciate ligament reconstruction surgery: an updated systematic review and meta-analysis including aspects of physical functioning and contextual factors. Br J Sports Med 48:1543
Boden BP, Dean GS, Feagin JA Jr, Garrett WE Jr (2000) Mechanisms of anterior cruciate ligament injury. Orthopedics 23:573–578
Borchers JR, Kaeding CC, Pedroza AD, Huston LJ, Spindler KP, Wright RW et al (2011) Intra-articular findings in primary and revision anterior cruciate ligament reconstruction surgery: a comparison of the MOON and MARS study groups. Am J Sports Med 39:1889–1893
Brambilla L, Pulici L, Carimati G, Quaglia A, Prospero E, Bait C et al (2015) Prevalence of associated lesions in anterior cruciate ligament reconstruction: correlation with surgical timing and with patient age, sex, and body mass index. Am J Sports Med 43:2966–2973
Charles C, Gafni A, Whelan T (1997) Shared decision-making in the medical encounter: what does it mean? (or it takes at least two to tango). Soc Sci Med 44:681–692
Chmielewski TL, Hurd WJ, Rudolph KS, Axe MJ, Snyder-Mackler L (2005) Perturbation training improves knee kinematics and reduces muscle co-contraction after complete unilateral anterior cruciate ligament rupture. Phys Ther 85:740–749
Chmielewski TL, Stackhouse S, Axe MJ, Snyder-Mackler L (2004) A prospective analysis of incidence and severity of quadriceps inhibition in a consecutive sample of 100 patients with complete acute anterior cruciate ligament rupture. J Orthop Res 22:925–930
Cimino PM (1994) The incidence of meniscal tears associated with acute anterior cruciate ligament disruption secondary to snow skiing accidents. Arthroscopy 10(2):198–200
Cohen M, Amaro JT, Ejnisman B, Carvalho RT, Nakano KK, Peccin MS et al (2007) Anterior cruciate ligament reconstruction after 10–15 years: association between meniscectomy and osteoarthrosis. Arthroscopy 23:629–634
Eitzen I, Moksnes H, Snyder-Mackler L, Risberg MA (2010) A progressive 5-week exercise therapy program leads to significant improvement in knee function early after anterior cruciate ligament injury. J Orthop Sports Phys Ther 40:705–721
Failla MJ, Logerstedt DS, Grindem H, Axe MJ, Risberg MA, Engebretsen L et al (2016) Does extended preoperative rehabilitation influence outcomes 2 years after ACL reconstruction? A comparative effectiveness study between the MOON and Delaware-Oslo ACL cohorts. Am J Sports Med e 44:2608–2614
Fairbank TJ (1948) Knee joint changes after meniscectomy. J Bone Joint Surg Br 30b:664–670
Fayard JM, Sonnery-Cottet B, Vrgoc G, O'Loughlin P, de Mont Marin GD, Freychet B et al (2019) Incidence and risk factors for a partial anterior cruciate ligament tear progressing to a complete tear after nonoperative treatment in patients younger than 30 years. Orthop J Sports Med 7:2325967119856624
Fithian DC, Paxton EW, Stone ML, Luetzow WF, Csintalan RP, Phelan D et al (2005) Prospective trial of a treatment algorithm for the management of the anterior cruciate ligament-injured knee. Am J Sports Med 33:335–346
Fitzgerald GK, Axe MJ, Snyder-Mackler L (2000) A decision-making scheme for returning patients to high-level activity with nonoperative treatment after anterior cruciate ligament rupture. Knee Surg Sports Traumatol Arthrosc 8:76–82
Fitzgerald GK, Axe MJ, Snyder-Mackler L (2000) The efficacy of perturbation training in nonoperative anterior cruciate ligament rehabilitation programs for physically active individuals. Phys Ther 80:128
Flint FA, Weiss MR (1992) Returning injured athletes to competition: a role and ethical dilemma. Can J Sport Sci 17:34–40
Frobell R, Roos E, Roos H, Ranstam J, Lohmander L (2010) A randomized trial of treatment for acute anterior cruciate ligament tears. N Engl J Med 363(4):331–342
Frobell RB, Roos HP, Roos EM, Roemer FW, Ranstam J, Lohmander LS (2013) Treatment for acute anterior cruciate ligament tear: 5 year outcome of randomised trial. BMJ 346:f232
Fu FH, van Eck CF, Tashman S, Irrgang JJ, Moreland MS (2015) Anatomic anterior cruciate ligament reconstruction: a changing paradigm. Knee Surg Sports Traumatol Arthrosc 23:640–648
Glyn-Jones S, Palmer AJ, Agricola R, Price AJ, Vincent TL, Weinans H, Carr AJ (2015) Osteoarthritis. Lancet 386(9991):376–387
Granan L-P, Bahr R, Lie SA, Engebretsen L (2009) Timing of anterior cruciate ligament reconstructive surgery and risk of cartilage lesions and meniscal tears: a cohort study based on the Norwegian National Knee Ligament Registry. Am J Sports Med 37:955–961
Granan L-P, Bahr R, Steindal K, Furnes O, Engebretsen L (2007) Development of a National Cruciate Ligament Surgery Registry: The Norwegian National Knee Ligament Registry. Am J Sports Med 36:308–315
Grindem H, Eitzen I, Engebretsen L, Snyder-Mackler L, Risberg MA (2014) Nonsurgical or surgical treatment of ACL injuries: knee function, sports participation, and knee reinjury: The Delaware-Oslo ACL Cohort Study. J Bone Joint Surg Am 96:1233–1241
Grindem H, Eitzen I, Moksnes H, Snyder-Mackler L, Risberg MA (2012) A pair-matched comparison of return to pivoting sports at 1 year in anterior cruciate ligament-injured patients after a nonoperative versus an operative treatment course. Am J Sports Med 40:2509–2516
Hangalur G, Brenneman E, Nicholls M, Bakker R, Laing A, Chandrashekar N (2016) Can a knee brace reduce the strain in the anterior cruciate ligament? A study using combined in vivo/in vitro method. Prosthet Orthot Int 40:394–399
Hertel P, Behrend H, Cierpinski T, Musahl V, Widjaja G (2005) ACL reconstruction using bone-patellar tendon-bone press-fit fixation: 10-year clinical results. Knee Surg Sports Traumatol Arthrosc 13:248–255
Hoshino Y, Miyaji N, Nishida K, Nishizawa Y, Araki D, Kanzaki N et al (2018) The concomitant lateral meniscus injury increased the pivot shift in the anterior cruciate ligament-injured knee. Knee Surg Sports Traumatol Arthrosc. https://doi.org/10.1007/s00167-018-5209-7
Hurd WJ, Axe MJ, Snyder-Mackler L (2008) A 10-year prospective trial of a patient management algorithm and screening examination for highly active individuals with anterior cruciate ligament injury: part 2, determinants of dynamic knee stability. Am J Sports Med 36:48–56
Ineke V, John MH (1999) Coaches’ decision policies about the participation of injured athletes in competition. Sport Psychologist 13:42–56
Jan van de Pol G, Arnold MP, Verdonschot N, van Kampen A (2008) Varus alignment leads to increased forces in the anterior cruciate ligament. Am J Sports Med 37:481–487
Jonsson H, Riklund-Ahlstrom K, Lind J (2004) Positive pivot shift after ACL reconstruction predicts later osteoarthrosis: 63 patients followed 5–9 years after surgery. Acta Orthop Scand 75:594–599
Kaeding CC, Leger-St-Jean B, Magnussen RA (2017) Epidemiology and diagnosis of anterior cruciate ligament injuries. Clin Sports Med 36:1–8
Keays SL, Newcombe PA, Bullock-Saxton JE, Bullock MI, Keays AC (2010) Factors involved in the development of osteoarthritis after anterior cruciate ligament surgery. Am J Sports Med 38:455–463
Kluczynski MA, Marzo JM, Bisson LJ (2013) Factors associated with meniscal tears and chondral lesions in patients undergoing anterior cruciate ligament reconstruction: a prospective study. Am J Sports Med 41(12):2759–2765
Krutsch W, Zellner J, Baumann F, Pfeifer C, Nerlich M, Angele P (2017) Timing of anterior cruciate ligament reconstruction within the first year after trauma and its influence on treatment of cartilage and meniscus pathology. Knee Surg Sports Traumatol Arthrosc 25:418–425
Kvist J, Kartus J, Karlsson J, Forssblad M (2014) Results from the Swedish National Anterior Cruciate Ligament Register. Arthroscopy 30:803–810
Kvist J, Kartus J, Karlsson J, Forssblad M (2014) Results from the Swedish national anterior cruciate ligament register. Arthroscopy 30(7):803–810
Lai CCH, Ardern CL, Feller JA, Webster KE (2018) Eighty-three per cent of elite athletes return to preinjury sport after anterior cruciate ligament reconstruction: a systematic review with meta-analysis of return to sport rates, graft rupture rates and performance outcomes. Br J Sports Med 52:128
Lawrence JT, Argawal N, Ganley TJ (2011) Degeneration of the knee joint in skeletally immature patients with a diagnosis of an anterior cruciate ligament tear: is there harm in delay of treatment? Am J Sports Med 39:2582–2587
Levy BA, Dajani KA, Morgan JA, Shah JP, Dahm DL, Stuart MJ (2010) Repair versus reconstruction of the fibular collateral ligament and posterolateral corner in the multiligament-injured knee. Am J Sports Med 38(4):804–809
Levy BA, Dajani KA, Whelan DB, Stannard JP, Fanelli GC, Stuart MJ et al (2009) Decision making in the multiligament-injured knee: an evidence-based systematic review. Arthroscopy 25:430–438
Lie MM, Risberg MA, Storheim K, Engebretsen L, Oiestad BE (2019) What's the rate of knee osteoarthritis 10 years after anterior cruciate ligament injury? An updated systematic review. Br J Sports Med 53(18):1162–1167
Lorbach O, Kieb M, Domnick C, Herbort M, Weyers I, Raschke M et al (2015) Biomechanical evaluation of knee kinematics after anatomic single- and anatomic double-bundle ACL reconstructions with medial meniscal repair. Knee Surg Sports Traumatol Arthrosc 23:2734–2741
Lynch AD, Logerstedt DS, Axe MJ, Snyder-Mackler L (2012) Quadriceps activation failure after anterior cruciate ligament rupture is not mediated by knee joint effusion. J Orthop Sports Phys Ther 42:502–510
Lynch TS, Parker RD, Patel RM, Andrish JT, Spindler KP, Group M (2015) The impact of the Multicenter Orthopaedic Outcomes Network (MOON) research on anterior cruciate ligament reconstruction and orthopaedic practice. J Am Acad Orthop Surg 23:154
Magnussen RA, Mansour AA, Carey JL, Spindler KP (2009) Meniscus status at anterior cruciate ligament reconstruction associated with radiographic signs of osteoarthritis at 5-to 10-year follow-up–a systematic review. J Knee Surg 22:347–357
Meuffels DE, Favejee MM, Vissers MM, Heijboer MP, Reijman M, Verhaar JA (2009) Ten year follow-up study comparing conservative versus operative treatment of anterior cruciate ligament ruptures. A matched-pair analysis of high level athletes. Br J Sports Med 43:347–351
Meunier A, Odensten M, Good L (2007) Long-term results after primary repair or non-surgical treatment of anterior cruciate ligament rupture: a randomized study with a 15-year follow-up. Scand J Med Sci Sports 17(3):230–237
Musahl V, Citak M, O’Loughlin PF, Choi D, Bedi A, Pearle AD (2010) The effect of medial versus lateral meniscectomy on the stability of the anterior cruciate ligament-deficient knee. Am J Sports Med 38:1591–1597
Musahl V, Kopf S, Rabuck S, Becker R, van der Merwe W, Zaffagnini S et al (2012) Rotatory knee laxity tests and the pivot shift as tools for ACL treatment algorithm. Knee Surg Sports Traumatol Arthrosc 20:793–800
Musahl V, Rahnemai-Azar AA, Costello J, Arner JW, Fu FH, Hoshino Y et al (2016) The influence of meniscal and anterolateral capsular injury on knee laxity in patients with anterior cruciate ligament injuries. Am J Sports Med 44:3126–3131
Muthuri SG, McWilliams DF, Doherty M, Zhang W (2011) History of knee injuries and knee osteoarthritis: a meta-analysis of observational studies. Osteoarthr Cartil 19:1286–1293
Nebelung W, Wuschech H (2005) Thirty-five years of follow-up of anterior cruciate ligament—deficient knees in high-level athletes. Arthroscopy 21:696–702
Neuman P, Englund M, Kostogiannis I, Friden T, Roos H, Dahlberg LE (2008) Prevalence of tibiofemoral osteoarthritis 15 years after nonoperative treatment of anterior cruciate ligament injury: a prospective cohort study. Am J Sports Med 36:1717–1725
Nippert AH, Smith AM (2008) Psychologic stress related to injury and impact on sport performance. Phys Med Rehabil Clino N Am 19:399–418
Noyes FR, Barber SD, Mangine RE (1991) Abnormal lower limb symmetry determined by function hop tests after anterior cruciate ligament rupture. Am J Sports Med 19:513–518
Noyes FR, Matthews DS, Mooar PA, Grood ES (1983) The symptomatic anterior cruciate-deficient knee. Part II: the results of rehabilitation, activity modification, and counseling on functional disability. J Bone Joint Surg Am 65:163–174
Noyes FR, Schipplein OD, Andriacchi TP, Saddemi SR, Weise M (1992) The anterior cruciate ligament-deficient knee with varus alignment. An analysis of gait adaptations and dynamic joint loadings. Am J Sports Med 20:707–716
Palmieri-Smith RM, Kreinbrink J, Ashton-Miller JA, Wojtys EM (2007) Quadriceps inhibition induced by an experimental knee joint effusion affects knee joint mechanics during a single-legged drop landing. Am J Sports Med 35:1269–1275
Paterno MV (2017) Non-operative care of the patient with an ACL-deficient knee. Curr Rev Musculoskelet Med 10:322–327
Pauzenberger L, Syre S, Schurz M (2013) "Ligamentization" in hamstring tendon grafts after anterior cruciate ligament reconstruction: a systematic review of the literature and a glimpse into the future. Arthroscopy 29:1712–1721
Peskun CJ, Whelan DB (2011) Outcomes of operative and nonoperative treatment of multiligament knee injuries: an evidence-based review. Sports Med Arthrosc Rev 19:167–173
Pfeiffer TR, Burnham JM, Hughes JD, Kanakamedala AC, Herbst E, Popchak A et al (2018) An increased lateral femoral condyle ratio is a risk factor for anterior cruciate ligament injury. J Bone Joint Surg Am 100:857–864
Pfeiffer TR, Burnham JM, Kanakamedala AC, Hughes JD, Zlotnicki J, Popchak A, Debski RE, Musahl V (2019) Distal femur morphology affects rotatory knee instability in patients with anterior cruciate ligament ruptures. Knee Surg Sports Traumatol Arthrosc 27(5):1514–1519
Phillips M, Rönnblad E, Lopez-Rengstig L, Svantesson E, Stålman A, Eriksson K et al (2018) Meniscus repair with simultaneous ACL reconstruction demonstrated similar clinical outcomes as isolated ACL repair: a result not seen with meniscus resection. Knee Surg Sports Traumatol Arthrosc 26:2270–2277
Rahnemai-Azar AA, Abebe ES, Johnson P, Labrum J, Fu FH, Irrgang JJ et al (2017) Increased lateral tibial slope predicts high-grade rotatory knee laxity pre-operatively in ACL reconstruction. Knee Surg Sports Traumatol Arthrosc 25:1170–1176
Richter M, Bosch U, Wippermann B, Hofmann A, Krettek C (2002) Comparison of surgical repair or reconstruction of the cruciate ligaments versus nonsurgical treatment in patients with traumatic knee dislocations. Am J Sports Med 30:718–727
Sanders TL, Kremers HM, Bryan AJ, Fruth KM, Larson DR, Pareek A et al (2016) Is anterior cruciate ligament reconstruction effective in preventing secondary meniscal tears and osteoarthritis? Am J Sports Med 44:1699–1707
Sarraj M, Coughlin RP, Solow M, Ekhtiari S, Simunovic N, Krych AJ, MacDonald P, Ayeni OR (2019) Anterior cruciate ligament reconstruction with concomitant meniscal surgery: a systematic review and meta-analysis of outcomes. Knee Surg Sports Traumatol Arthrosc 27(11):3441–3452
Scheffler SU, Unterhauser FN, Weiler A (2008) Graft remodeling and ligamentization after cruciate ligament reconstruction. Knee Surg Sports Traumatol Arthrosc 16:834–842
Sitler M, Ryan CJ, Hopkinson LW, Wheeler LJ, Santomier J, Kolb LR et al (1990) The efficacy of a prophylactic knee brace to reduce knee injuries in football: a prospective, randomized study at West Point. Am J Sports Med 18:310–315
Sommerfeldt M, Goodine T, Raheem A, Whittaker J, Otto D (2018) Relationship between time to ACL reconstruction and presence of adverse changes in the knee at the time of reconstruction. Orthop J Sports Med 6(12):2325967118813917
Sommerfeldt M, Raheem A, Whittaker J, Hui C, Otto D (2018) Recurrent instability episodes and meniscal or cartilage damage after anterior cruciate ligament injury: a systematic review. Orthop J Sports Med 6(7):2325967118786507
Stannard JP, Brown SL, Farris RC, McGwin G Jr, Volgas DA (2005) The posterolateral corner of the knee: repair versus reconstruction. Am J Sports Med 33(6):881–888
Sundemo D, Blom A, Hoshino Y, Kuroda R, Lopomo NF, Zaffagnini S et al (2018) Correlation between quantitative pivot shift and generalized joint laxity: a prospective multicenter study of ACL ruptures. Knee Surg Sports Traumatol Arthrosc 26:2362–2370
Thoma LM, Grindem H, Logerstedt D, Axe M, Engebretsen L, Risberg MA, Snyder-Mackler L (2019) "Coper classification early after anterior cruciate ligament rupture changes with progressive neuromuscular and strength training and is associated with 2-year success: the Delaware-Oslo ACL Cohort Study. Am J Sports Med 47(4):807–814
Troeberg L, Nagase H (2012) Proteases involved in cartilage matrix degradation in osteoarthritis. Biochim Biophys Acta 1824:133–145
van Eck CF, Gravare-Silbernagel K, Samuelsson K, Musahl V, van Dijk CN, Karlsson J et al (2013) Evidence to support the interpretation and use of the Anatomic Anterior Cruciate Ligament Reconstruction Checklist. J Bone Joint Surg Am 95:e153
van Eck CF, Lesniak BP, Schreiber VM, Fu FH (2010) Anatomic single- and double-bundle anterior cruciate ligament reconstruction flowchart. Arthroscopy 26:258–268
van Yperen DT, Reijman M, van Es EM, Bierma-Zeinstra SMA, Meuffels DE (2018) Twenty-year follow-up study comparing operative versus nonoperative treatment of anterior cruciate ligament ruptures in high-level athletes. Am J Sports Med 46(5):1129–1136
Vincent TL (2013) Targeting mechanotransduction pathways in osteoarthritis: a focus on the pericellular matrix. Curr Opin Pharmacol 13(3):449–454
Voos JE, Suero EM, Citak M, Petrigliano FP, Bosscher MR, Citak M et al (2012) Effect of tibial slope on the stability of the anterior cruciate ligament–deficient knee. Knee Surg Sports Traumatol Arthrosc 20:1626–1631
Waite JC, Beard DJ, Dodd CAF, Murray DW, Gill HS (2005) In vivo kinematics of the ACL-deficient limb during running and cutting. Knee Surg Sports Traumatol Arthrosc 13:377–384
Walden M, Hagglund M, Magnusson H, Ekstrand J (2011) Anterior cruciate ligament injury in elite football: a prospective three-cohort study. Knee Surg Sports Traumatol Arthrosc 19:11–19
Wu WH, Hackett T, Richmond JC (2002) Effects of meniscal and articular surface status on knee stability, function, and symptoms after anterior cruciate ligament reconstruction: a long-term prospective study. Am J Sports Med 30:845–850
Zaffagnini S, Signorelli C, Grassi A, Hoshino Y, Kuroda R, de Sa D, Sundemo D, Samuelsson K, Musahl V, Karlsson J, Sheean A, Burnham JM, Lian J, Smith C, Popchak A, Herbst E, Pfeiffer T, Araujo P, Oostdyk A, Guenther D, Ohashi B, Irrgang JJ, Fu FH, Nagamune K, Kurosaka M, Marcheggiani Muccioli GM, Lopomo N, Raggi F, Svantesson E, Hamrin Senorski E, Bjoernsson H, Ahlden M, Desai N (2018) Anatomic anterior cruciate ligament reconstruction using hamstring tendons restores quantitative pivot shift. Orthop J Sports Med 6(12):2325967118812364
Zantop T, Herbort M, Raschke MJ, Fu FH, Petersen W (2007) The role of the anteromedial and posterolateral bundles of the anterior cruciate ligament in anterior tibial translation and internal rotation. Am J Sports Med 35:223–227
Acknowledgements
Panther Consensus Group: Olufemi R. Ayeni, Charles H. Brown Jr., Terese L. Chmielewski, Mark Clatworthy, Stefano Della Villa, Theresa Diermeier, Lars Engebretsen, Lucio Ernlund, Christian Fink, Freddie H. Fu, Alan Getgood, Timothy E. Hewett, Yasuyuki Ishibashi, Darren L. Johnson, Jon Karlsson, Andrew D. Lynch, Jeffrey A. Macalena, Robert G. Marx, Jacques Menetrey, Sean J. Meredith, Volker Musahl, Kentaro Onishi, Mark V. Paterno, Thomas Rauer, Benjamin B. Rothrauff, Laura C. Schmitt, Romain Seil, Eric Hamrin Senorski, Rainer Siebold, Lynn Snyder-Mackler, Tim Spalding, Eleonor Svantesson, Kevin E. Wilk, and John W. Xerogeanes.
Funding
There was no funding for this study.
Author information
Authors and Affiliations
Consortia
Corresponding author
Ethics declarations
Conflict of interest
Dr. Ayeni reports Speakers Bureau from Conmed, honoraria from DJO, outside the submitted work. Dr. Engebretsen reports grants from Smith and Nephew, outside the submitted work, and Editorship of JBJS and BJSM. Dr. Xerogeanes reports personal fees and non-financial support from Arthrex, personal fees from Trice Medical, outside the submitted work. Dr. Fu reports educational support and hospitality payments from Smith & Nephew, outside the submitted work. Dr. Musahl reports educational grants, consulting fees, and speaking fees from Smith & Nephew and educational grants from Arthrex. Dr. Diermeier has nothing to disclose. Dr. Rothrauff has nothing to disclose. Dr. Lynch has nothing to disclose. Dr. Paterno has nothing to disclose. Dr. Karlsson reports being Editor-in-Chief of KSSTA. Dr. Svantesson has nothing to disclose. Dr. Hamrin-Senorski has nothing to disclose. Dr. Rauer has nothing to disclose. Dr. Meredith has nothing to disclose.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
Informed consent were not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original version of this article was revised due to a retrospective Open Access order.
Authors details for “The Panther Symposium ACL Treatment Consensus Group” are given in acknowledgement section.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Diermeier, T., Rothrauff, B.B., Engebretsen, L. et al. Treatment after anterior cruciate ligament injury: Panther Symposium ACL Treatment Consensus Group. Knee Surg Sports Traumatol Arthrosc 28, 2390–2402 (2020). https://doi.org/10.1007/s00167-020-06012-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00167-020-06012-6