Abstract
Purpose
Chemokines are major promoters of repair and may regulate nerve ingrowth that is essential in tendon healing. The purpose of this study was to assess the temporal occurrence of different chemokines during Achilles tendon healing in relation to sensory nerve regeneration. Chemokine presence in tendon healing has not been studied previously.
Methods
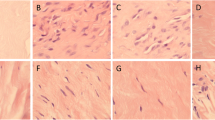
Chemokine expression, nerve regeneration, angiogenesis and inflammatory cell occurrence during healing of Achilles tendon rupture in the rat were studied by immunohistochemistry and histology including semiquantitative assessment. Markers for chemokines (CCL5, CCL2, CCL3, CXCL10), nerves (PGP-9.5) and sensory neuropeptide substance P (SP) were analysed at different time points (1 day–16 weeks) post-rupture.
Results
In intact tendons (controls) immunoreactivity to all chemokines, PGP-9.5 and SP were confined to the tendon surroundings. After rupture, there was rapid increase in the tendon proper of the chemokines studied, all exhibiting their peak expression at week 1. Subsequently, at weeks 2–6, emerging inflammatory cells and maximum sprouting of PGP-/SP-positive nerves were observed close to newly formed blood vessels within the tendon proper, while chemokine expression already decreased. During weeks 6–8, PGP-/SP-positive nerves withdrew from the rupture site and relocated together with the chemokines in the surrounding tendon.
Conclusions
Early chemokine expression in the healing tendon precedes ingrowth of new nerves, angiogenesis and emergence of inflammatory cells. The fine-tuned temporal and spatial appearance of chemokines suggests a chemoattractant role for inflammatory cell migration and possibly also a role in angiogenesis and neurogenesis. Chemokines may thus exhibit vital targets for biological modulation of tendon repair.
Level of evidence
III.





Similar content being viewed by others
References
Ackermann PW (2013) Neuronal regulation of tendon homoeostasis. Int J Exp Pathol 94(4):271–286
Ackermann PW, Ahmed M, Kreicbergs A (2002) Early nerve regeneration after Achilles tendon rupture-a prerequisite for healing? A study in the rat. J Orthop Res 20(4):849–856
Ackermann PW, Domeij-Arverud E, Leclerc P, Amoudroz P, Nader GA (2013) Anti-inflammatory cytokine profile in early human tendon repair. Knee Surg Sports Traumatol Arthrosc 21(8):1801–1806
Ackermann PW, Li J, Lundeberg T, Kreicbergs A (2003) Neuronal plasticity in relation to nociception and healing of rat Achilles tendon. J Orthop Res 21(3):432–441
Ackermann PW, Salo PT, Hart DA (2009) Neuronal pathways in tendon healing. Front Biosci 14:5165–5187
Angiolillo AL, Sgadari C, Taub DD, Liao F, Farber JM, Maheshwari S, Kleinman HK, Reaman GH, Tosato G (1995) Human interferon-inducible protein 10 is a potent inhibitor of angiogenesis in vivo. J Exp Med 182(1):155–162
Baggiolini M, Dewald B, Moser B (1997) Human chemokines: an update. Annu Rev Immunol 15:675–705
Behm B, Babilas P, Landthaler M, Schremi S (2012) Cytokines, chemokines and growth factors in wound healing. J Eur Acad Dermatol Venereol 26(7):812–820
Bring DK, Heidgren ML, Kreicbergs A, Ackermann PW (2005) Increase in sensory neuropeptides surrounding the Achilles tendon in rats with adjuvant arthritis. J Orthop Res 23(2):294–301
Burssens P, Steyaert A, Forsyth R, van Ovost EJ, Depaepe Y, Verdonk R (2005) Exogenously administered substance P and neutral endopeptidase inhibitors stimulate fibroblast proliferation, angiogenesis and collagen organization during Achilles tendon healing. Foot Ankle Int 26(10):832–839
Carlsson O, Schizas N, Li J, Ackermann PW (2011) Substance P injections enhance tissue proliferation and regulate sensory nerve ingrowth in rat tendon repair. Scand J Med Sci Sports 21(4):562–569
Charo IF, Ransohoff RM (2006) The many roles of chemokines and chemokine receptors in inflammation. N Engl J Med 354(6):610–621
DiPietro LA, Burdick M, Low QE, Kunkel SL, Strieter RM (1998) MIP-1alpha as a critical macrophage chemoattractant in murine wound repair. J Clin Invest 101(8):1693–1698
Dvorak HF, Brown LF, Detmar M, Dvorak AM (1995) Vascular permeability factor/vascular endothelial growth factor, microvascular hyperpermeability, and angiogenesis. Am J Pathol 146(5):1029–1039
Frank S, Kämpfer H, Wetzler C, Stallmeyer B, Pfellschifter J (2000) Large induction of the chemotactic cytokine RANTES during cutaneous wound repair: a regulatory role for nitric oxide in keratinocyte-derived RANTES expression. Biochem J 347(Pt 1):265–273
Glaser J, Gonzalez R, Sadr E, Keirstead HS (2006) Neutralization of the chemokine CXCL10 reduces apoptosis and increases axon sprouting after spinal cord injury. J Neurosci Res 84(4):724–734
Hong HS, Lee J, Lee E, Kwon YS, Lee E, Ahn W, Jiang MH, Kim JC, Son Y (2009) A new role of substance P as an injury-inducible messenger for mobilization of CD29(+) stromal-like cells. Nat Med 15(4):425–435
Kalehua AN, Nagel JE, Whelchel LM, Gides JJ, Pyle RS, Smith RJ, Kusiak JW, Taub DD (2004) Monocyte chemoattractant protein-1 and macrophage inflammatory protein-2 are involved in both excitotoxin-induced neurodegeneration and regeneration. Exp Cell Res 297(1):197–211
Low QE, Drugea IA, Duffner LA, Quinn DG, Cook DN, Rollins BJ, Kovacs EJ, DiPietro LA (2001) Wound healing in MIP-1alpha(−/−) and MCP-1(−/−) mice. Am J Pathol 159(2):457–463
Mantovani A, Biswas SK, Galdiero MR, Sica A, Locati M (2013) Macrophage plasticity and polarization in tissue repair and remodelling. J Pathol 229(2):176–185
Nicoletti M, Neri G, Maccauro G, Tripodi D, Varvara G, Saggini A, Patalivo G, Castellani ML, Fulcheri M, Rosati M, Toniato E, Caraffa A, Antinolfi P, Cerulli G, Pandolfi F, Galzio R, Conti P, Theoharides TC (2012) Impact of neuropeptide substance P an inflammatory compound on arachidonic acid compound generation. Int J Immunopathol Pharmacol 25(4):849–857
Olsson N, Nilsson-Helander K, Karlsson J, Eriksson BI, Thomee R, Faxen E, Silbernagl KG (2011) Major functional deficits persist 2 years after acute Achilles tendon rupture. Knee Surg Sports Traumatol Arthrosc 19(8):1385–1393
Sharma P, Maffulli N (2005) Tendon injury and tendinopathy: healing and repair. J Bone Joint Surg Am 87(1):187–202
Sharma P, Maffulli N (2006) Biology of tendon injury: healing, modeling and remodeling. J Musculoskelet Neuronal Interact 6(2):181–190
Shiraha H, Glading A, Gupta K, Wells A (1999) IP-10 inhibits epidermal growth factor-induced motility by decreasing epidermal growth factor receptor-mediated calpain activity. J Cell Biol 146(1):243–254
Suffee N, Hlawaty H, Meddahl-Pelle A, Maillard L, Louedec L, Haddad O, Martin L, Laguillier C, Richard B, Oudar O, Letourneur D, Charnaux N, Sutton A (2012) RANTES/CCL5-induced pro-angiogenic effects depend on CCR1, CCR5 and glycosaminoglycans. Angiogenesis 15(4):727–744
Suffee N, Richard B, Hlawaty H, Oudar O, Charnaux N, Sutton A (2011) Angiogenic properties of the chemokine RANTES/CCL5. Biochem Soc Trans 39(6):1649–1653
Taskinen HS, Röyttä M (2000) Increased expression of chemokines (MCP-1, MIP-1alpha, RANTES) after peripheral nerve transection. J Peripher Nerv Syst 5(2):75–81
Tonnesen MG, Feng X, Clark RA (2000) Angiogenesis in wound healing. J Investig Dermatol Symp Proc 5(1):40–46
Wetzler C, Kämpfer H, Stallmeyer B, Pfeilschifter J, Frank S (2000) Large and sustained induction of chemokine during impaired wound healing in the genetically diabetic mouse: prolonged persistence of neutrophils and macrophages during the late phase of repair. J Invest Dermatol 115(2):245–253
Acknowledgments
This study was supported by the regional agreement on medical training and clinical research (ALF) between Stockholm County Council and Karolinska Institutet (project nr. SLL20100168) and the Swedish National Centre for Sports Research as well as the Swedish Research Council (project nr. 2012-3510).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Stålman, A., Bring, D. & Ackermann, P.W. Chemokine expression of CCL2, CCL3, CCL5 and CXCL10 during early inflammatory tendon healing precedes nerve regeneration: an immunohistochemical study in the rat. Knee Surg Sports Traumatol Arthrosc 23, 2682–2689 (2015). https://doi.org/10.1007/s00167-014-3010-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00167-014-3010-9