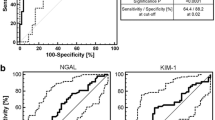
Abstract
Purpose
Acute kidney injury (AKI) is a frequent and severe condition in intensive care units (ICUs). In 2020, the Acute Dialysis Quality Initiative (ADQI) group proposed a new stage of AKI, referred to as stage 1S, which represents subclinical disease (sAKI) defined as a positive biomarker but no increase in serum creatinine (sCr). This study aimed to determine and compare the urinary peptide signature of sAKI as defined by biomarkers.
Methods
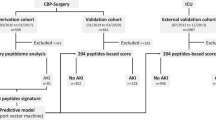
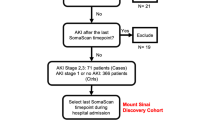
This is an ancillary analysis of the prospective, observational, multinational FROG-ICU cohort study. AKI was defined according to the Kidney Disease Improving Global Outcome definition (AKIKDIGO). sAKI was defined based on the levels of the following biomarkers, which exceeded the median value: neutrophil gelatinase-associated lipocalin (pNGAL, uNGAL), cystatin C (pCysC, uCysC), proenkephalin A 119–159 (pPENKID) and liver fatty acid binding protein (uLFABP). Urinary peptidomics analysis was performed using capillary electrophoresis-mass spectrometry. Samples were collected at the time of study inclusion.
Results
One thousand eight hundred eighty-five patients had all biomarkers measured at inclusion, which included 1154 patients without AKI (non-AKIKDIGO subgroup). The non-AKIKDIGO subgroup consisted of individuals at a median age of 60 years [48, 71], among whom 321 (27.8%) died. The urinary peptide signatures of sAKI, regardless of the biomarkers used for its definition, were similar to the urinary peptide signatures of AKIKDIGO (inflammation, haemolysis, and endothelial dysfunction). These signatures were also associated with 1-year mortality.
Conclusion
Biomarker-defined sAKI is a common and severe condition observed in patients within intensive care units with a urinary peptide signature that is similar to that of AKI, along with a comparable prognosis.
Graphical abstract





Similar content being viewed by others
Data availability
The data that support the findings of this study are available from the corresponding author (FD) upon reasonable request.
References
Hoste EA, Bagshaw SM, Bellomo R et al (2015) Epidemiology of acute kidney injury in critically ill patients: the multinational AKI-EPI study. Intensive Care Med 41(8):1411–1423. https://doi.org/10.1007/s00134-015-3934-7
Section 2: AKI Definition. Kidney Int Suppl (2011). 2012 Mar;2(1):19–36. https://doi.org/10.1038/kisup.2011.32
Córdova-Sánchez BM, Herrera-Gómez Á, Ñamendys-Silva SA (2016) Acute Kidney Injury Classified by Serum Creatinine and Urine Output in Critically Ill Cancer Patients. Biomed Res Int 2016:6805169. https://doi.org/10.1155/2016/6805169
Ronco C, Rosner MH (2012) Acute kidney injury and residual renal function. Crit Care 16(4):144. https://doi.org/10.1186/cc11426
Kannapiran M, Nisha D, Madhusudhana RA (2010) Underestimation of impaired kidney function with serum creatinine. Indian J Clin Biochem 25(4):380–384. https://doi.org/10.1007/s12291-010-0080-4
Chen YW, Wu MY, Mao CH et al (2022) Severe acute kidney disease is associated with worse kidney outcome among acute kidney injury patients. Sci Rep 12(1):6492. https://doi.org/10.1038/s41598-022-09599-7
Noble RA, Lucas BJ, Selby NM (2020) Long-Term Outcomes in Patients with Acute Kidney Injury. Clin J Am Soc Nephrol 15(3):423–429. https://doi.org/10.2215/CJN.10410919
Ostermann M, Zarbock A, Goldstein S et al (2020) Recommendations on acute kidney injury biomarkers from the acute disease quality initiative consensus conference: a consensus statement. JAMA Netw Open 3(10):e2019209. https://doi.org/10.1001/jamanetworkopen.2020.19209
Nehomar PG (2020) Biomarkers in Acute Kidney Injury. J Clin Nephrol 4:027–035. https://doi.org/10.29328/journal.jcn.1001056
Mavrogeorgis E, Mischak H, Latosinska A et al (2021) Collagen-Derived Peptides in CKD: a link to fibrosis. Toxins (Basel) 14(1):10. https://doi.org/10.3390/toxins14010010
He T, Mischak M, Clark AL et al (2021) Urinary peptides in heart failure: a link to molecular pathophysiology. Eur J Heart Fail 23(11):1875–1887. https://doi.org/10.1002/ejhf.2195
Piedrafita A, Siwy J, Klein J et al (2022) A universal predictive and mechanistic urinary peptide signature in acute kidney injury. Crit Care 26(1):344. https://doi.org/10.1186/s13054-022-04193-9
Mebazaa A, Casadio MC, Azoulay E et al (2015) Post-ICU discharge and outcome: rationale and methods of the The French and euRopean Outcome egistry in Intensive Care Units (FROG-ICU) observational study. BMC Anesthesiol 15:143. https://doi.org/10.1186/s12871-015-0129-2
Vincent JL, Moreno R, Takala J et al (1996) The SOFA (Sepsis-related Organ Failure Assessment) score to describe organ dysfunction/failure On behalf of the Working Group on Sepsis-Related Problems of the European Society of Intensive Care Medicine. Intensive Care Med 22(7):707–710. https://doi.org/10.1007/BF01709751
Le Gall JR, Lemeshow S, Saulnier F (1993) A new Simplified Acute Physiology Score (SAPS II) based on a European/North American multicenter study. JAMA 270(24):2957–2963. https://doi.org/10.1001/jama.270.24.2957
Dépret F, Hollinger A, Cariou A et al (2020) Incidence and Outcome of Subclinical Acute Kidney Injury Using penKid in Critically Ill Patients. Am J Respir Crit Care Med 202(6):822–829. https://doi.org/10.1164/rccm.201910-1950OC
Mischak H, Vlahou A, Ioannidis JP (2013) Technical aspects and inter-laboratory variability in native peptide profiling: the CE-MS experience. Clin Biochem 46(6):432–443. https://doi.org/10.1016/j.clinbiochem.2012.09.025
Cuschieri S (2019) The STROBE guidelines. Saudi J Anaesth 13(1):S31–S34. https://doi.org/10.4103/sja.SJA_543_18
UniProt Consortium (2023) UniProt: the Universal Protein Knowledgebase in 2023. Nucleic Acids Res 51(D1):D523–D531. https://doi.org/10.1093/nar/gkac1052
Harris MA, Clark J, Ireland A et al (2004) The Gene Ontology (GO) database and informatics resource. Nucleic Acids Res 32:D258–D261. https://doi.org/10.1093/nar/gkh036
Haase M, Devarajan P, Haase-Fielitz A et al (2011) The outcome of neutrophil gelatinase-associated lipocalin-positive subclinical acute kidney injury: a multicenter pooled analysis of prospective studies. J Am Coll Cardiol 57(17):1752–1761. https://doi.org/10.1016/j.jacc.2010.11.051
Legrand M, Darmon M, Joannidis M (2013) NGAL and AKI: the end of a myth? Intensive Care Med 39(10):1861–1863. https://doi.org/10.1007/s00134-013-3061-2
Legrand M, Mari A, Mebazaa A (2013) The elusive task of biomarkers of renal injury. Crit Care 17(2):132. https://doi.org/10.1186/cc12578
Work Group Membership (2011) Kidney Int Suppl 2(4):281. https://doi.org/10.1038/kisup.2012.39
De Vlieger G, Forni L, Schneider A (2022) New diagnostics for AKI in critically ill patients: what to expect in the future. Intensive Care Med 48:1632–1634. https://doi.org/10.1007/s00134-022-06843-6
von Groote TC, Ostermann M, Forni LG, Meersch-Dini M, Zarbock A, PrevAKI Investigators (2022) The AKI care bundle: all bundle components are created equal-are they? Intensive Care Med 48(2):242–245. https://doi.org/10.1007/s00134-021-06601-0
Zarbock A, Küllmar M, Ostermann M et al (2021) Prevention of Cardiac Surgery-Associated Acute Kidney Injury by Implementing the KDIGO Guidelines in High-Risk Patients Identified by Biomarkers: The PrevAKI-Multicenter Randomized Controlled Trial. Anesth Analg 133(2):292–302. https://doi.org/10.1213/ANE.0000000000005458
Schrezenmeier EV, Barasch J, Budde K, Westhoff T, Schmidt-Ott KM (2017) Biomarkers in acute kidney injury - pathophysiological basis and clinical performance. Acta Physiol (Oxf) 219(3):554–572. https://doi.org/10.1111/apha.12764
Törnblom S, Nisula S, Petäjä L et al (2020) Urine NGAL as a biomarker for septic AKI: a critical appraisal of clinical utility-data from the observational FINNAKI study. Ann Intensive Care 10(1):51. https://doi.org/10.1186/s13613-020-00667-7
Khawaja S, Jafri L, Siddiqui I, Hashmi M, Ghani F (2019) The utility of neutrophil gelatinase-associated Lipocalin (NGAL) as a marker of acute kidney injury (AKI) in critically ill patients. Biomark Res 7:4. https://doi.org/10.1186/s40364-019-0155-1
Shimazui T, Nakada TA, Tateishi Y, Oshima T, Aizimu T, Oda S (2019) Association between serum levels of interleukin-6 on ICU admission and subsequent outcomes in critically ill patients with acute kidney injury. BMC Nephrol 20(1):74. https://doi.org/10.1186/s12882-019-1265-6
Zhang Z, Lu B, Sheng X, Jin N (2011) Cystatin C in prediction of acute kidney injury: a systemic review and meta-analysis. Am J Kidney Dis 58(3):356–365. https://doi.org/10.1053/j.ajkd.2011.02.389
Lagos-Arevalo P, Palijan A, Vertullo L et al (2015) Cystatin C in acute kidney injury diagnosis: early biomarker or alternative to serum creatinine? Pediatr Nephrol 30(4):665–676. https://doi.org/10.1007/s00467-014-2987-0
Boutin L, Legrand M, Sadoune M et al (2022) Elevated plasma Galectin-3 is associated with major adverse kidney events and death after ICU admission. Crit Care 26(1):13. https://doi.org/10.1186/s13054-021-03878-x
Kellum JA, Romagnani P, Ashuntantang G, Ronco C, Zarbock A, Anders HJ (2021) Acute kidney injury. Nat Rev Dis Primers 7(1):52. https://doi.org/10.1038/s41572-021-00284-z
Pickkers P, Darmon M, Hoste E et al (2021) Acute kidney injury in the critically ill: an updated review on pathophysiology and management. Intensive Care Med 47(8):835–850. https://doi.org/10.1007/s00134-021-06454-7
Kellum JA, Ronco C, Bellomo R (2021) Conceptual advances and evolving terminology in acute kidney disease. Nat Rev Nephrol 17(7):493–502. https://doi.org/10.1038/s41581-021-00410-w
Forni LG, Joannidis M, Artigas A et al (2022) Characterising acutekidney injury: The complementary roles of biomarkers of renal stress and renal function. J Crit Care 71:154066. https://doi.org/10.1016/j.jcrc.2022.154066
Schmitt J, Aries P, Deserts DD, M. et al (2022) Before AKI renal microcirculation stress may be detected by urine biochemistry. Intensive Care Med 48:1672–1673. https://doi.org/10.1007/s00134-022-06873-0
Acknowledgments
The authors are particularly grateful to CRAs and healthcare providers of all investigating centers. We also thank the “Centre de Recherche Clinique” (CRC) of Lariboisière University Hospital for his support. N. Deye, C. Fauvaux, C. Damoisel, D. Payen, E.Gayat, E. Azoulay, A. S. Moreau, L. Jacob, O. Marie (Hôpital Saint Louis, Paris), M. Wolf, R. Sonneville, R. Bronchard (Hôpital Bichat, Paris), I. Rennuit, C. Paugam (Hôpital Beaujon, Clichy), J. P. Mira, A. Cariou, A. Tesnieres (Hôpital Cochin, Paris), N. Dufour, N. Anguel, L. Guerin, J. Duranteau, C. Ract (Hôpital Bicêtre, Le Kremlin‐Bicêtre), M. Leone, B. Pastene (CHU De Marseille, Marseille), T. Sharshar, A. Fayssoyl (Hôpital Raymond Poincare, Garches), J.‐L. Baudel, B. Guidet (Hôpital Saint‐Antoine), Q. Lu, W. Jie Gu, N. Brechot, A. Combes (Hôpital La Pitie – Salpetriere, Paris), S. Jaber, A. 6 Pradel, Y. Coisel, M. Conseil (CHU St Eloi, Montpellier), A. Veillard‐Baron, L. Bodson (Hôpital Ambroise Pare, Boulogne), Jy. Lefrant, L. Elotmani, A. Ayral, S. Lloret (CHU Caremeau, Nimes), S. Pily‐Flouri, Jb. Pretalli (Hopital Jean Minjoz, Besançon), Pf. Laterre, V. Montiel, Mf. Dujardin, C. Berghe (Clinique Saint‐Luc, Belgium). We thank Nicholas Fong for his revision of the manuscript.
Funding
FROG‐ICU (ClinicalTrials.gov Identifier NCT01367093) was funded by the “Programme Hospitalier de la Recherche Clinique” (AON 10‐216) 5 and by a research grant from the “Société Française d’Anesthésie – Réanimation”. Abbott, Sphingotec, Roche Diagnostics, and Critical Diagnostics provided unrestricted free kits to “Assistance Publique – Hôpitaux de Paris” to conduct biomarker analyses. The original FROG-ICU study was supported by grants from “Assistance Publique‐Hôpitaux de Paris” (AOR01004) and from “Société Française d’Anesthésie–Réanimation”. The study funders had no role in the design and conduct of the study; collection, management, analysis, and interpretation of the data; preparation, review, or approval of the manuscript; or decision to submit the manuscript for publication.
Author information
Authors and Affiliations
Contributions
FD and LB had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. Concept and design: LB, AL, HM, FD. Acquisition, analysis, or interpretation of data: LB, AL, HM, ML, AM, FD. Drafting of the manuscript: LB, AL, HM, BD, ML, AM, FD. Critical revision of the manuscript for important intellectual content: LB, AL, HM, BD, AA, ML, EG, AM, CEC, FD. Statistical analysis: LB. Obtained funding: ML, EG, AM, FD. Supervision: ML, AM, FD.
Corresponding author
Ethics declarations
Conflict of interest
HM is the cofounder and co-owner of Mosaiques Diagnostics (Hannover, Germany) and AL is employee of Mosaiques Diagnostics. EG received a research grant from Sphingotec and consultancy fees from Magnisense and Roche Diagnostics. AM received speaker’s honoraria from Abbott, Novartis, Orion, Roche, and Servier and a fee as a member of the advisory board and/or steering committee from Cardiorentis, Adrenomed, MyCartis, Neurotronik, and Sphingotec. The other authors have no conflicts of interest to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Boutin, L., Latosinska, A., Mischak, H. et al. Subclinical and clinical acute kidney injury share similar urinary peptide signatures and prognosis. Intensive Care Med 49, 1191–1202 (2023). https://doi.org/10.1007/s00134-023-07198-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-023-07198-2