Abstract
Purpose
On a regular basis, the intensivist encounters the patient who is difficult to wean from mechanical ventilatory support. The causes for failure to wean from mechanical ventilatory support are often multifactorial and involve a complex interplay between cardiac and pulmonary dysfunction. A potential application of point of care ultrasonography relates to its utility in the process of weaning the patient from mechanical ventilatory support.
Methods
This article reviews some applications of ultrasonography that may be relevant to the process of weaning from mechanical ventilatory support.
Results
The authors have divided these applications of ultrasonography into four separate categories: the assessment of cardiac, diaphragmatic, and lung function; and the identification of pleural effusion; which can all be evaluated with ultrasonography during a dynamic process in which the intensivist is uniquely positioned to use ultrasonography at the point of care.
Conclusions
Ultrasonography may have useful application during the weaning process from mechanical ventilatory support.
Similar content being viewed by others
Introduction
Ultrasonography is well established as a useful bedside tool when performed by the intensivist at point of care for evaluation of cardiopulmonary failure. As ultrasonography is an evolving part of critical care medicine, an interesting feature of the discipline is that it lends itself to innovative applications. One of these relates to its potential utility in the process of weaning the patient from mechanical ventilatory support. As this utility is not yet well defined, this article reviews some applications of ultrasonography that may be relevant to the weaning process. We have divided these into four separate categories: the assessment of cardiac, diaphragmatic, pleural effusion and lung function. The purpose of this review is to inform the reader as to the potential utility of ultrasonography to aid in the process of weaning from mechanical ventilatory support. In reviewing the literature on the subject, it is important to define what constitutes successful weaning. Most investigators define success as when the patient remains extubated for 48 h following removal of the endotracheal tube. Some studies report on the outcome of a spontaneous breathing trial (SBT), which is generally defined as a period of observation during which the patient is breathing through a T-piece with an appropriate amount of supplemental oxygen. Adequate SBT performance does not necessarily result in successful extubation.
Assessment of cardiac function
Weaning may be considered an exercise requiring an elevation in cardiac index, oxygen demand and consumption [1]. Jubran et al. reported that patients who failed an SBT had significant reduction in mixed venous oxygen saturation (SvO2) compared to patients who pass the SBT where SvO2 remained constant [2]. In the failure group, an uncoupling between the left ventricle and the systemic circulation and between the right ventricle and the pulmonary circulation occurred [2]. While cardiac function is crucial to the weaning process, the incidence of cardiac-related weaning failure is not known. It is difficult to evaluate its incidence, as increases in respiratory load and cardiac load are strongly interrelated [3]; so it is difficult to identify to what extent a cardiac problem is the cause for the failure. Cabello et al. reported that cardiac-related weaning failure occurred in 42 % of cases among 76 patients who failed SBT [4]. In this study, difficult to wean patients performed an SBT with a T-piece versus pressure support ventilation (PSV) with and without PEEP. The failure rate on T-piece was higher than that on PSV, indicating that loading conditions vary according the ventilator settings used during the SBT. This is a potential confounder in analysing the results of studies related to SBT.
Besides the increase in respiratory and cardiac load that occurs during weaning, spontaneous ventilation may stress the cardiovascular system compared to positive pressure ventilation. Negative intrathoracic pressure has been reported to depress left ventricular (LV) performance [5], and to be coupled with a significant increase in systemic venous return [6] and LV ischemia [7]. These factors may alter LV diastolic function with relaxation impairment, decreased compliance and increased filling pressure [8] precipitating cardiogenic pulmonary oedema with resultant respiratory failure.
It follows that the use of critical care echocardiography (CCE) might be useful to evaluate the left ventricle during the weaning process, owing to its ability to measure LV diastolic and systolic performance [9]. LV diastolic and systolic dysfunction are involved in cardiac-related weaning failure, with diastolic dysfunction being the predominant factor [10]. Evaluation of diastolic function requires competence in advanced CCE as the intensivist must be able to use and correctly interpret pulsed wave Doppler (PWD) and tissue Doppler imaging (TDI) [11, 12]. The profile of the mitral inflow recorded by the PWD, with the E wave in early diastole and the A wave in late diastole, depends on LV diastolic function and filling pressure, while TDI of the mitral annulus allows separate evaluation of LV relaxation (e′ wave at early diastole) and the LV filling pressure (E/e′ ratio). The E wave velocity and E/e′ ratio should be used with caution in patients with mitral regurgitation, mitral stenosis and mitral prosthetic valve, as these indices have not been validated for measurement of diastolic function.
The ability of CCE to predict weaning failure remains uncertain, as the cause may be multifactorial; cardiac dysfunction may contribute but other complex factors may be involved. Although CCE may accurately identify the high-risk patient before performing an SBT, there is no definitive data to support its use during SBT as a help to decide whether to extubate the patient. Two studies done in unselected critically ill patients reported that CCE performed before and during an SBT identified related changes in LV diastolic function and pressure with an increase in E/A, a decrease in E wave deceleration time and an increase in E/e′ [13, 14]. As a result of the variability of these measurements and the relatively small value of the changes, this approach cannot be recommended for routine identification of patients in whom cardiac dysfunction occurs during the SBT. However, an echocardiographic evaluation before the SBT seems valuable to identify patients at high risk of weaning failure. In a population of 117 patients, Caille et al. reported that the 23 patients who failed to be weaned had a lower LV ejection fraction (EF) and a trend towards a higher E/e′ before SBT [13]. Moschietto et al. reported in 68 patients no difference in LVEF but an increased E/e′ in the failure group [15]. Gerbaud et al. did not confirm these results, but the population of their study was highly selected with patients hospitalised for heart failure with a mean LVEF of 40 % [16]. Papanikolaou et al. reported the impact of LV diastolic function on weaning failure in 50 patients with preserved LV systolic function who fulfilled standard criteria for SBT [17]. LV diastolic dysfunction was graded in three stages according to its severity based on PWD and TDI evaluation, as previously proposed [18]. Weaning failure occurred in 35 % of cases in patients with normal diastolic function, in 57 % in patients with grade 1 (mild) diastolic dysfunction and in 80 % in patients with grade 2 (moderate) or 3 (severe) [17]. Dres et al. recently proposed to test the cardiovascular system before the SBT, using the passive leg raising (PLR) manoeuvre [19]. They hypothesised that preload independency, i.e. no cardiac output increase during PLR, would be associated with cardiac-related weaning failure. Cardiac output was measured by the pulse contour analysis method, but it could easily be measured by echocardiography. Among the 57 SBT included in the study, 11 SBT were successful; 10 of these patients were preload responsive. Among the 46 failed SBT, 45 patients were preload unresponsive, and all had cardiac dysfunction as evidenced by an elevation of pulmonary artery occlusion pressure [19]. During the weaning process, advanced CCE may be used to predict weaning failure but also to diagnose whether a cardiogenic cause is responsible for the failure. When the pretest probability of weaning failure is high, as in patients with known heart failure, CCE has limited utility. When the pretest probability is low or intermediate, as in an unselected population of critically ill patients, CCE might be useful in predicting weaning failure.
Advanced CCE may have special application for the intensivist treating the patient with multiple medical problems who fails SBT criteria outright or who needs reintubation in the 48 h following extubation. Except for patients who were initially ventilated for a cardiac cause, LVEF is frequently preserved and cardiac-related weaning failure is generally due to an LV diastolic dysfunction in patients with several risk factors such as age, arrhythmia, hypertension (frequently due to delirium or discomfort upon cessation of sedation) and kidney disease [20]. On the basis of standard guidelines for evaluation diastolic function [18], cardiac-related weaning failure may be suspected when the LV filling pressure is elevated with an E/A ratio >2 if the ejection fraction is reduced or an E/e′ > 12 if the ejection fraction is normal. For measurement of diastolic function in the context of weaning failure from ventilatory support, the advanced CCE examination should be performed in close temporal relationship to the clinical event i.e. during the SBT or immediately following the failure of the SBT and before any treatment has been initiated to treat cardiac dysfunction. Lamia et al., by performing left ventricular filling pressure estimation by CCE at the end of SBT, detected weaning-induced pulmonary artery occlusion pressure (PAOP) elevation in 39 patients [21] with concomitant changes in E/A and E/e′. Rare cases also reported weaning failure due to hypertrophic obstructive cardiomyopathy [22]. This is easily detectable by CCE and has a strong clinical impact since diuretics, inotropes or vasodilators are then contraindicated. The need to perform the advanced CCE examination when the cardiovascular system is under load in close conjunction with the SBT emphasises that standard cardiology consultative echocardiography is not useful for evaluation of the dynamic changes in diastolic function that may occur with the SBT. The intensivist with advanced CCE capability is uniquely positioned to do this type of examination at point of care.
To summarise, cardiac-related weaning failure may be due to either a systolic or an isolated diastolic LV dysfunction. Advanced CCE can be useful to diagnose, and sometimes predict, cardiac-related weaning failure which likely occurs as a result of the precipitation of cardiogenic pulmonary oedema. However, at this time there is no randomised clinical trial that demonstrates benefit of performing CCE to guide weaning in terms of shortening of weaning duration or survival improvement. In absence of this level of evidence, the results of the echocardiography examination may be used to develop a treatment plan that is based upon sound clinical reasoning. How CCE may be combined with cardiac biomarkers for improving accuracy remains to be elucidated. Table 1 summarizes some measurements of cardiac function that may have application to weaning from mechanical ventilatory support.
Assessment of diaphragmatic function
Ultrasonography allows easy visualisation of the two hemidiaphragms and their excursion during respiratory cycles. Two different ultrasonographic parameters have been described to evaluate diaphragmatic performance. The first parameter refers to the measurement of diaphragmatic excursion (E) during inspiration [23]. E can be easily measured with a 3- to 5-MHz probe either in B- or M-mode, with so-called anatomical M-mode allowing placement of the M-mode line parallel to the diaphragmatic excursion [24, 25]. The second parameter describes diaphragmatic muscle thickening during inspiration in the zone of apposition of the diaphragm to the rib cage with at least a 10-MHz probe using the B- or M-mode [26]. Thickening fraction (TF = [thickness at end-inspiration − thickness at end-expiration]/thickness at end-expiration) is reported in most studies. For both parameters, technical tips to improve reproducibility and accuracy of measurements have been published in detail allowing excellent operating characteristics [24, 25].
In patients breathing spontaneously, diaphragmatic excursion is the result of a given diaphragmatic contraction for a given mechanical burden (i.e. compliance of the respiratory system including abdominal compliance). In patients receiving mechanical support, diaphragmatic excursion also depends on the amount of support and of PEEP level. Indeed PEEP increases end-expiratory lung volume; the corresponding increase in lung volume lowers the diaphragmatic dome, which can result in decreased diaphragmatic excursion [27]. Measurements of diaphragmatic thickening can be used as a direct index of diaphragmatic efficiency as a pressure generator, although thickness may also be influenced by lung volume in a non-linear relationship [26, 28, 29]. Both E and TF have been shown to correlate with functional measurements of diaphragmatic function (typically transdiaphragmatic pressure measurements, Pdi) in spontaneously breathing patients [23]. As a result of the effect of PEEP and level of ventilatory support on the diaphragmatic contour on E, only muscle thickening measurements perform well in analysing diaphragmatic function in patients receiving mechanical ventilation [29, 30]. No study has compared the performance of E and TF in spontaneously breathing patients to assess diaphragmatic performance.
Diaphragmatic dysfunction in patients receiving mechanical ventilation most frequently occurs relatively symmetrically secondary to neuromuscular disorders or non-myopathic diseases such as chronic obstructive pulmonary disease, to ICU-acquired neuromyopathy and/or to ventilator-induced diaphragmatic dysfunction [31–33]. Alternatively, phrenic nerve injury, especially during cardiac surgery, may lead to asymmetric diaphragmatic dysfunction [34]. Accordingly, most studies, performed in non-cardiac surgery patients, recorded E or TF on only one hemidiaphragm (usually on the right side) taken as a proxy for the whole diaphragmatic performance [29, 30, 34, 35]. Only a few studies considered the worst or the mean value between right and left [36–38]. In cardiac surgery patients, Lerolle et al. performed bilateral assessment of diaphragmatic excursion and identified the best E (E max) as the functional parameter of diaphragmatic performance [23]. In cardiac surgery patients, injury of a phrenic nerve may lead to complete paralysis of a hemidiaphragm while the other remains unaffected. This generally does not lead to global functional impairment. In this study, patients with unilateral diaphragmatic paralysis could be weaned off the ventilator without delay when the contralateral diaphragm performed well enough i.e. E max > 25 mm. An E max < 25 mm, indicating bilateral diaphragmatic paresis or paralysis, had a positive likelihood ratio of 6.7 and a negative likelihood ratio of 0 for having a severe diaphragmatic dysfunction as diagnosed with Pdi measurements. This study shows that cardiac surgery patients, exposed to a specific form of phrenic nerve injury, require bilateral examination of the diaphragm. Although in other settings unilateral measurements may perform well in assessing global diaphragmatic function, it may be appropriate to screen first for asymmetric diaphragmatic dysfunction.
Four studies described the use of ultrasonographic evaluation of the diaphragm in the process of weaning in non-cardiac surgery patients. Jiang et al. considered the mean value of inspiratory excursions of the liver and the spleen measured during an SBT [37]. Among the 55 patients who underwent extubation, those who required reintubation within 72 h had significantly lower mean values of liver and spleen displacements than those who did not require reintubation. At a cutoff value of 1.1 cm, the sensitivity and specificity to predict successful extubation were 84 and 83 %. In a cohort of 82 patients, Kim et al. defined diaphragmatic dysfunction by an E value < 10 mm for either hemidiaphragm [36]. The patients with diaphragmatic dysfunction according to this criterion had longer weaning time and higher frequency of reintubation. The applicability of these two studies is questionable considering the unusually high rates of weaning failure in unselected patients (41 % and 65 % respectively). In 2014, two studies were published evaluating TF in the weaning process. Di Nino et al. studied 63 patients who were extubated after diaphragmatic assessment during an SBT or a pressure support trial (PST) [34]. Twenty-two per cent of the patients required reintubation within 48 h. A threshold of TF ≥ 30 % was associated with a positive predictive value of 91 % and a negative predictive value of 63 % for extubation success. Performance of TF was similar if the measurements were performed during an SBT or a PST. Similarly, Ferrari et al. found in 46 patients with repeated weaning failure that a cutoff value of a TF > 36 % measured during an SBT on tracheostomy tube was associated with a positive predictive value of 92 % and a negative predictive value of 75 % for success or failure from discontinuation of mechanical support at 48 h [35]. These studies confirm the potential interest of these indices, either E or TF, in the weaning process. However, much remains to be performed to define their place vs. clinical parameters. In the aforementioned studies, the performance of ultrasonographic indexes in predicting extubation failure or success was compared with such clinical parameters as the Rapid Shallow Breathing Index, and proved equivalent or slightly better, although the number of patients studied was low. It is likely that ultrasonography may be clinically relevant in specific situations such as neuromuscular disease or in extensive ICU-acquired neuromyopathy. Ultrasonography may be particularly useful to monitor recovery over time. Mariani et al. studied diaphragmatic excursion in 34 patients with prolonged (>7 days) mechanical ventilation; repeated measurements were performed in seven patients with bilateral dysfunction (E < 11 mm) [39]. Progressive improvement of E was observed over time in 71 % of cases. Grosu et al. observed diaphragmatic thinning over time in patients receiving volume-controlled mechanical ventilation [40]. Table 2 summarizes some measurements of diaphragmatic function that may have application to weaning from mechanical ventilatory support.
Assessment of pleural effusion
In considering whether removal of pleural fluid might improve the likelihood of successful weaning, it is useful to review the physiological consequences of pleural fluid on respiratory function. Pleural fluid accumulation results in relaxation atelectasis of the lung that is adjacent to the pleural effusion. This is in association with the hydrostatic fluid pressure generated by the effusion. The degree of atelectasis is readily observed with lung ultrasonography. Large pleural effusions may cause lobar or even total lung atelectasis. The atelectatic lung is airless. As such, it must result in loss of alveolar volume available for gas exchange. Even large effusions may have surprisingly little effect on oxygenation function, presumably as a result of a reduction in blood flow to the non-aerated atelectatic lung.
As the elastic recoil of the intercostal chest wall is no longer opposed by that of the lung, the configuration of the chest wall changes in the area of the effusion. The resulting outward movement of the chest wall causes a reduction in length tension efficiency of the intercostal muscles. The diaphragm is uncoupled from the visceral pleural surface, such that diaphragmatic movement has an attenuated effect on lung inflation. With large effusions, pleural pressure may be so elevated as to result in reverse curvature of the ipsilateral diaphragm. This results in profound alteration of the preload function of the diaphragm, which is optimal only when the diaphragm has normal concavity.
Given the predictable negative physiological effects of pleural fluid, it is reasonable to consider whether removing a pleural effusion might increase the chances of successful weaning. This is an intuitively attractive concept, given the empiric clinical observation that removal of pleural fluid in the spontaneously breathing patient will often result in immediate improvement in dyspnoea and associated respiratory distress. The effect of pleural fluid removal has been studied in patients on mechanical ventilatory support. Ahmed et al. reported no change in PaO2 following fluid removal, but reduction in pulmonary artery occlusion, central venous pressure and pulmonary shunt fraction in association with an increase in VO2 and DO2 [41]. Doelken et al. reported no improvement in PaO2, physiological dead space, static or dynamic respiratory system compliance and end-inspiratory airway resistance; passive inflation work was reduced from 3.42 to 2.99 J/L [42]. This reduction in ventilator performed work might predict a reduction in the work performed by the spontaneously breathing patient. Razazi et al. reported improvement in respiratory system compliance, end-inspiratory transpulmonary pressure, PaO2/FIO2 ratio and end-expiratory lung volume [43]. A lesser improvement in oxygenation occurred in patients with ARDS. These improvements were sustained when measured at 3 and 24 h after pleural fluid drainage. In a recent meta-analysis of five studies comprising 118 patients, there was an overall 18 % improvement in PaO2/FIO2 following pleural fluid removal [44]. With focus on patient outcome, Kupfer et al. reported that continuous drainage of transudative pleural effusions with an indwelling catheter was associated with a reduction in days of ventilatory support (3.8 ± 0.5 vs. 6.5 ± 1.1 days). This was a retrospective non-randomised observational study. Average fluid removal was 1.2 L [45]. Talmor et al. reported in a retrospective study improvement in oxygenation in patients who required substantial PEEP with ARDS [46].
The lack of consistent major measurable improvement in gas exchange or respiratory mechanics should not discourage the intensivist from removing the pleural effusion. Sound physiological reasoning, the visual observation of a large area of non-functioning atelectatic lung on ultrasonography and the empiric observation that pleural fluid removal in the spontaneously breathing patient promptly reduces respiratory distress all indicate that thoracentesis may improve the likelihood of successful extubation, particularly in the patient with marginal cardiopulmonary function who is approaching extubation attempt or who has failed SBT.
In considering the option of pleural fluid drainage, the intensivist needs to consider several factors. Thoracentesis with ultrasonography guidance has a low risk of complication in the patient on mechanical ventilatory support [47, 48]. While the volume of the effusion may be accurately estimated with ultrasonography [49, 50], it is not clear what volume indicates for removal of the effusion, given that the aforementioned studies were performed with a wide range of pleural fluid volume removal. It is unlikely that removal of a small effusion will have positive influence on the weaning process; and the larger the effusion is, the more likely its removal will improve the likelihood of successful extubation. Table 3 summarizes an approach to pleural effusion that may have application to weaning from mechanical ventilatory support. On the basis of a single retrospective study [51], the clinician may consider the option of placing an indwelling catheter into the pleural space for continuous pleural effusion drainage as the patient approaches extubation attempt, rather than performing removal of the pleural fluid as a single event.
Assessment of lung aeration
The utility of lung ultrasonography (LUS) for guiding the weaning process in the ICU derives from the observation that the initiation of spontaneous breathing after a period of mechanical ventilation is associated with some loss of lung aeration in the critically ill [52]. LUS assessment before and at the end of the SBT is useful for detecting aeration loss, which is relevant for prediction of post-extubation distress, irrespective of its primary cause i.e. cardiac, respiratory or diaphragmatic.
Soummer et al. demonstrated that LUS predicts weaning failure with some accuracy by identifying global and regional lung de-recruitment [52]. A LUS score has been validated with the aim of providing quantifiable comparable measures of progressive changes in aeration [53–56]. This score originates from the conversion of lung ultrasound patterns into numeric values, which are allocated according to the worst ultrasound pattern observed for a given chest region. The final score, ranging from 0 to 36, is the sum of the values, from 0 to 3, assigned to the LUS patterns visualized in each of the 12 regions examined. The 12 anterior, lateral and posterior regions are delimited by anatomic landmarks as stated in the consensus conference recommendations for point-of-care LUS [57]. The four ultrasound patterns identified for each region correspond to progressive degrees of lung aeration (Table 4). While this LUS score calculated on 12 chest areas allows the measurement of the degree of lung aeration at a precise moment, the following step is to score the change in aeration in successive stages, for instance before and after treatment. For this purpose, a LUS dynamic re-aeration score can be calculated for each region by adding 1, 3 or 5 points respectively in case of a slight, moderate or substantial increase in lung aeration, or subtracting the same points in case of aeration loss [53].
In the setting of weaning, the LUS score was accurate in predicting the occurrence of post-extubation distress by measuring lung aeration defects during the SBT [52]. The score was increased in patients who failed the trial and went on to develop post-extubation distress. This correlation was irrespective of the underlying cause and lung condition. By contrast, the LUS score remained significantly under a cutoff value in patients who went on to be successfully weaned. The identified cutoff to predict 85 % risk of post-extubation failure was >17, whereas the safest value below which the risk of developing post-extubation failure was negligible was <13 (negative likelihood ratio of 0.20). A score between these two values, found in 25 % of patients, was inconclusive in predicting recurrence of respiratory distress or successful weaning. Table 5 summarizes LUS that may have application to weaning from mechanical ventilatory support. In this study, the LUS score was more accurate than B-type natriuretic peptide and transthoracic echocardiography in discriminating failure from success.
Beyond its proven utility in predicting the outcome of the SBT, the LUS score has potential in management of respiratory failure. The LUS allows assessment of regional lung aeration. A regional quantification allows identification of regions that are predominantly involved in lung de-recruitment and therefore contribute more than other areas to the patient’s inability to maintain spontaneous breathing. Both global and regional assessment of lung de-recruitment provides useful insights into the aetiology of the weaning failure [45]. This information might then be used to optimise the patient’s condition before SBT by detecting and treating alteration of the aeration pattern of the lung, such as de-recruited areas due to compressive phenomenon (effusion) and obstructive atelectasis [45, 58]. Thus, LUS may be helpful in deciding the correct timing of an SBT. A high score during mechanical ventilation could indicate that the patient is not yet ready to begin the weaning process, whereas a lower score could be useful in shortening the weaning period and minimising time on mechanical ventilation. The effect of any therapeutic strategy in the post-SBT phase may also be monitored at the bedside by the assessment of changes in time of the LUS score [4]. This is currently under study, and the preliminary results of a trial were recently presented at the ESICM Congress [59].
The LUS evaluation of pulmonary aeration in the context of weaning from ventilatory support has some limitations. The ultrasound is a surface imaging technique and not a panoramic tool. Lung pathology that is surrounded by aerated lung will not be visible, as aerated lung blocks transmission of ultrasound. This limitation is seldom relevant in the critically ill patient because diseases characteristic of critical care medicine usually extend to the visceral pleural surface and are therefore visible with ultrasonography. Finally, the LUS score as presently defined has utility as a research tool, but may be overly complicated for the frontline intensivist to use in a busy ICU.
Despite these limitations, LUS is a useful bedside tool that allows real-time assessment of the lung during an SBT. On the basis of the available literature, the LUS examination, including a baseline examination and repeated assessment during and after SBT, is useful for the routine management of the weaning process. More prospective studies are needed to further validate the technique and to increase evidence of its utility [60]. Future research may combine LUS with the Rapid Shallow Breathing Index [61] and ultrasound examination of diaphragm function to further improve the predictive value of the LUS score.
Summary
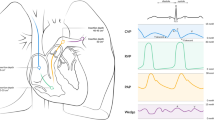
Usual practice, physiology and well-known causes of weaning failure all support the use of ultrasonography to identify patients who are at high risk for weaning failure. When ultrasonography is used to diagnose the cause(s) of the failure to wean, the clinician can use this information to optimise physiological function in order to improve the likelihood of successful discontinuation of ventilatory support. Advanced CCE allows the intensivist to detect systolic or diastolic LV dysfunction, while thoracic ultrasonography may help by detecting loss of lung aeration, pleural effusion or diaphragmatic dysfunction. However, the cause of weaning failure is often multifactorial. Ultrasonography will not generally identify a single cause for weaning failure, as several different mechanisms may be present. For instance, in a patient with LV diastolic dysfunction and diaphragmatic dysfunction, loss of lung aeration during the weaning may induce an increase in respiratory work, then an increase in cardiac demand and finally LV diastolic pressure elevation-related weaning failure with resultant cardiogenic pulmonary oedema. Ultrasonography will identify these processes and lead to directed intervention. If this patient also has co-existing severe emphysema and loss of airway clearance, neither of which is detectable with ultrasonography, the patient may still fail extubation despite useful results from the ultrasonography of the heart and thorax. Figure 1 presents a potential approach for use of ultrasonography to evaluate different factors involved in the weaning. In using this approach, several caveats are in order. To be useful, echocardiographic evaluation of the patient who is failing weaning has to be done as close as possible to the respiratory distress, i.e. during the SBT and before replacing the patient under positive pressure ventilation, or before reintubation of the patient in case of failed extubation, when possible. Likewise, lung ultrasonography should be performed in close conjunction to the SBT or failed extubation attempt. Rapid changes in respiratory and cardiac load occur during SBT and may manifest with dynamic changes in ultrasonography that are only visible with real-time scanning during the SBT. Ultrasonographic findings are always considered within the broader clinical context. The intensivist is therefore uniquely qualified to use ultrasonography for the bedside evaluation of weaning failure, as they have full knowledge of the clinical condition of their patient. As a corollary, ultrasonography should never be used as a final determinant of the success or failure of a weaning attempt. It may be helpful in identifying the high-risk patient, in defining a mechanism(s) for failure and to guide intervention that may increase the success of the next attempt. In the case of the patient at low risk of failure based on clinical assessment, ultrasonography may increase the accuracy of prediction; whereas in the other situations, it may help to determine the cause of failure.
Potential utility of ultrasonography for evaluation of spontaneous breathing trial. A pulsed wave Doppler late mitral valve inflow velocity, E pulsed wave Doppler early mitral valve inflow velocity, e′ mitral annular tissue Doppler velocity, LUS lung ultrasonography score, LV left ventricle, PLR passive leg raise, SBT spontaneous breathing trial. Results of the SBT may differ according to whether a T-piece or pressure support ventilation with PEEP (PSV 7 cmH2O/PEEP 0 or 5 cm) is used. Use of T-piece during the SBT may be associated with precipitation of cardiac failure when compared to the SBT performed with PSV, so the operator should consider this confounder when interpreting ultrasonography results [4]
Conclusion
This narrative review summarises the potential utility of ultrasonography to aid in the process of weaning from mechanical ventilatory support. Although there are no studies that have demonstrated improvement in outcome when using ultrasonography for this purpose, this review suggests the intensivist may productively use ultrasonography to identify impediments to successful extubation. This knowledge may then lead to logical therapeutic interventions. In using ultrasonography for this application, it is best to combine both cardiac and thoracic (diaphragm, lung and pleura) assessments, rather than using a limited examination approach. This allows the clinician to develop an integrative analysis that combines the results of a multi-system scanning approach. Future research is required to see if ultrasonography has utility to improve the process of weaning from mechanical ventilatory support, and how it can be productively combined with other clinical indicators.
References
De Backer D, El Haddad P, Preiser JC, Vincent JL (2000) Hemodynamic response to a successful weaning from mechanical ventilation after cardiovascular surgery. Intensive Care Med 26:1201–1206
Jubran A, Mathru M, Dries D, Tobin M (1998) Continuous recordings of mixed venous oxygen saturation during weaning from mechanical ventilation and the ramifications thereof. Am J Respir Crit Care Med 158:1763–1769
Boles JM, Bion J, Connors A, Herridge M, Marsh B, Melot C, Pearl R, Silverman H, Stanchina M, Vieillard-Baron A, Welte T (2007) Weaning from mechanical ventilation. Eur Respir J 29:1033–1056
Cabello B, Thille AW, Roche-Campo F, Brochard L, Gomez FJ, Mancebo J (2010) Physiological comparison of the three spontaneous breathing trials in difficult-to-wean patients. Intensive Care Med 36:1171–1179
Buda AJ, Pinsky MR, Ingels Jr NB, Daughters 2nd GT, Stinson EB, Alderman EL (1979) Effect of intrathoracic pressure on left ventricular performance. N Engl J Med 30:453–459
Richard C, Teboul JL, Archambaud F, Hebert JL, Michaut P, Auzepy P (1994) Left ventricular function during weaning of patients with chronic obstructive pulmonary disease. Intensive Care Med 20:181–186
Hurford WE, Favorito F (1995) Association of myocardial ischemia with failure to wean from mechanical ventilation. Crit Care Med 23:1475–1480
Lemaire F, Teboul JL, Cinotti L, Giotto G, Abrouk F, Steg G, Macquin-Mavier I, Zapol WM (1988) Acute left ventricular dysfunction during unsuccessful weaning from mechanical ventilation. Anesthesiology 69:171–179
Mayo PH, Beaulieu Y, Doelken P, Feller-Kopman D, Harrod C, Kaplan A, Oropello J, Vieillard-Baron A, Axler O, Lichtenstein D, Maury E, Slama M, Vignon P (2009) American College of Chest Physicians/La Société de Réanimation de Langue Française statement on competence in critical care ultrasonography. Chest 135:1050–1060
Saleh M, Vieillard-Baron A (2012) On the role of left ventricular diastolic function in the critically ill patient. Intensive Care Med 38:189–191
Expert Round Table on Ultrasound in ICU (2011) International expert statement on training standards for critical care ultrasonography. Intensive Care Med 37:1077–1083
Expert Round Table on Echocardiography in ICU (2014) International consensus statement on training standards for advanced critical care echocardiography. Intensive Care Med 40:654–666
Caille V, Amiel JB, Charron C, Belliard G, Vieillard-Baron A, Vignon P (2010) Echocardiography: a help in the weaning process. Crit Care 14:R120
Ait-Oufella H, Tharaux PL, Baudel JL, Vandermeersch S, Meyer P, Tonnelier M, Dussaule JC, Guidet B, Offenstadt G, Maury E (2007) Variation in natriuretic peptides and mitral flow indexes during successful ventilatory weaning: a preliminary study. Intensive Care Med 33:1183–1186
Moschietto S, Doyen D, Grech L, Dellamonica J, Hyvernat H, Bernardin G (2012) Transthoracic echocardiography with Doppler tissue imaging predicts weaning failure from mechanical ventilation: evolution of the left ventricle relaxation rate during a spontaneous breathing trial is a key factor in weaning outcome. Crit Care 16:R81
Gerbaud E, Erickson M, Grenouillet-Delacre M, Beauvieux MC, Coste P, Durrieu-Jaïs C, Hilbert G, Castaing Y, Vargas F (2012) Echocardiographic evaluation and N-terminal pro-brain natriuretic peptide measurement of patients hospitalized for heart failure during weaning from mechanical ventilation. Minerva Anestesiol 78:415–425
Papanikolaou J, Makris D, Saranteas T, Karakitsos D, Zintzaras E, Karabinis A, Kostopanagiotou G, Zakynthinos E (2011) New insights into weaning from mechanical ventilation: left ventricular diastolic dysfunction is a key player. Intensive Care Med 37:1976–1985
Nagueh SF, Appleton CP, Gillebert TC, Marino PN, Oh JK, Smiseth OA, Waggoner AD, Flachskampf FA, Pellikka PA, Evangelista A (2009) Recommendations for the evaluation of left ventricular diastolic function by echocardiography. Eur J Echocardiogr 10:165–193
Dres M, Teboul JL, Anguel N, Guerin L, Richard C, Monnet X (2015) Passive leg raising performed before a spontaneous breathing trial predicts weaning-induced cardiac dysfunction. Intensive Care Med 41:487–494
Owan TE, Hodge DO, Herges RM, Jacobsen SJ, Roger VL, Redfield MM (2006) Trends in prevalence and outcome of heart failure with preserved ejection fraction. N Engl J Med 355:251–259
Lamia B, Maizel J, Ochagavia A, Chemla D, Osman D, Richard C, Teboul JL (2009) Echocardiographic diagnosis of pulmonary artery occlusion pressure elevation during weaning from mechanical ventilation. Critical Care Med 37:1696–1701
Adamopoulos C, Tsagourias M, Arvaniti K, Veroniki F, Matamis D (2005) Weaning failure from mechanical ventilation due to hypertrophic obstructive cardiomyopathy. Intensive Care Med 31:734–737
Lerolle N, Guérot E, Zegdi R, Faisy C, Fagon J-Y, Diehl J-L (2009) Ultrasonographic diagnostic criterion for severe diaphragmatic dysfunction after cardiac surgery. Chest 135(2):401–407
Pasero D, Koeltz A, Placido R, Fontes Lima M, Haun O, Rienzo M et al (2015) Improving ultrasonic measurement of diaphragmatic excursion after cardiac surgery using the anatomical M-mode: a randomized crossover study. Intensive Care Med 41(4):650–656
Matamis D, Soilemezi E, Tsagourias M, Akoumianaki E, Dimassi S, Boroli F et al (2013) Sonographic evaluation of the diaphragm in critically ill patients. Technique and clinical applications. Intensive Care Med 39(5):801–810
Summerhill EM, El-Sameed YA, Glidden TJ, McCool FD (2008) Monitoring recovery from diaphragm paralysis with ultrasound. Chest J 133(3):737
Beaumont M, Lejeune D, Marotte H, Harf A, Lofaso F (1997) Effects of chest wall counterpressures on lung mechanics under high levels of CPAP in humans. J Appl Physiol 83(2):591–598
Cohn D, Benditt JO, Eveloff S, McCool FD (1997) Diaphragm thickening during inspiration. J Appl Physiol 83(1):291–296
Vivier E, Mekontso Dessap A, Dimassi S, Vargas F, Lyazidi A, Thille AW et al (2012) Diaphragm ultrasonography to estimate the work of breathing during non-invasive ventilation. Intensive Care Med 38(5):796–803
Umbrello M, Formenti P, Longhi D, Galimberti A, Piva I, Pezzi A et al (2015) Diaphragm ultrasound as indicator of respiratory effort in critically ill patients undergoing assisted mechanical ventilation: a pilot clinical study. Crit Care 19:161
Powers SK, Kavazis AN, Levine S (2009) Prolonged mechanical ventilation alters diaphragmatic structure and function. Crit Care Med 37:S347–S353
Doorduin J, van Hees HWH, van der Hoeven JG, Heunks LMA (2013) Monitoring of the respiratory muscles in the critically ill. Am J Respir Crit Care Med 187(1):20–27
Demoule A, Jung B, Prodanovic H, Molinari N, Chanques G, Coirault C et al (2013) Diaphragm dysfunction on admission to the intensive care unit. Prevalence, risk factors, and prognostic impact-a prospective study. Am J Respir Crit Care Med 188(2):213–219
DiNino E, Gartman EJ, Sethi JM, McCool FD (2014) Diaphragm ultrasound as a predictor of successful extubation from mechanical ventilation. Thorax 69(5):431–435
Ferrari G, De Filippi G, Elia F, Panero F, Volpicelli G, Aprà F (2014) Diaphragm ultrasound as a new index of discontinuation from mechanical ventilation. Crit Ultrasound J 6(1):8
Kim WY, Suh HJ, Hong S-B, Koh Y, Lim C-M (2011) Diaphragm dysfunction assessed by ultrasonography: influence on weaning from mechanical ventilation. Crit Care Med 39(12):2627–2630
Jiang J-R, Tsai T-H, Jerng J-S, Yu C-J, Wu H-D, Yang P-C (2004) Ultrasonographic evaluation of liver/spleen movements and extubation outcome. Chest 126(1):179–185
Valette X, Seguin A, Daubin C, Brunet J, Sauneuf B, Terzi N et al (2015) Diaphragmatic dysfunction at admission in intensive care unit: the value of diaphragmatic ultrasonography. Intensive Care Med 41(3):557–559
Mariani LF, Bedel J, Gros A, Lerolle N, Milojevic K, Laurent V et al (2015) Ultrasonography for screening and follow-up of diaphragmatic dysfunction in the ICU: a pilot study. J Intensive Care Med. doi:10.1177/088506661558363910.1177/0885066615583639
Grosu HB, Lee YI, Lee J, Eden E, Eikermann M, Rose KM (2012) Diaphragm muscle thinning in patients who are mechanically ventilated. Chest J 142(6):1455
Ahmed SH, Ouzounian SP, Dirusso S, Sullivan T, Savino J, Del Guercio L (2004) Hemodynamic and pulmonary changes after drainage of significant pleural effusions in critically ill, mechanically ventilated surgical patients. J Trauma 57:1184–1188
Doelken P, Abreu R, Sahn S, Mayo PH (2006) Effects of thoracentesis on respiratory mechanics and gas exchange in the mechanically ventilated patient. Chest 130:1354–1361
Razazi K, Thille AW, Carteaux G, Beji O, Brun-Buisson C, Brochard L, Dessap AM (2014) Effects of pleural effusion drainage on oxygenation, respiratory mechanics, and hemodynamics in mechanically ventilated patients. Ann Am Thorac Soc 11:1018–1024
Graf J (2009) Pleural effusion in the mechanically ventilated patient. Curr Opin Crit Care 15:10–17
Mongodi S, Via G, Bouhemad B, Storti E, Mojoli F, Braschi A (2013) Usefulness of combined bedside lung ultrasound and echocardiography to assess weaning failure from mechanical ventilation: a suggestive case. Crit Care Med 41(8):e182–e185
Talmor M, Hydo L, Gershenwald JG, Barie PS (1998) Beneficial effects of chest tube drainage of pleural effusion in acute respiratory failure refractory to positive end-expiratory pressure ventilation. Surgery 123:137–143
Lichtenstein D, Hulot JS, Rabiller A, Tostivint I, Mezière G (1999) Feasibility and safety of ultrasound-aided thoracentesis in mechanically ventilated patients. Intensive Care Med 25:955–958
Mayo PH, Goltz HR, Tafreshi M, Doelken P (2004) Safety of ultrasound-guided thoracentesis in patients receiving mechanical ventilation. Chest 125:1059–1062
Vignon P, Chastagner C, Berkane V, Chardac E, François B, Normand S, Bonnivard M, Clavel M, Pichon N, Preux PM, Maubon A, Gastinne H (2005) Quantitative assessment of pleural effusion in critically ill patients by means of ultrasonography. Crit Care Med 33:1757–1763
Begot E, Grumann A, Duvoid T, Dalmay F, Pichon N, François B, Clavel M, Vignon P (2014) Ultrasonographic identification and semiquantitative assessment of unloculated pleural effusions in critically ill patients by residents after a focused training. Intensive Care Med 40:1475–1480
Kupfer Y, Seneviratne C, Chawla K, Ramachandran K, Tessler S (2011) Chest tube drainage of transudative pleural effusions hastens liberation from mechanical ventilation. Chest 139:519–523
Soummer A, Perbet S, Brisson H, Arbelot C, Constantin JM, Lu Q, Rouby JJ, The Lung Ultrasound Study Group (2012) Ultrasound assessment of lung aeration loss during a successful weaning trial predicts postextubation distress. Crit Care Med 40:2064–2072
Bouhemad B, Liu ZH, Arbelot C, Zhang M, Ferarri F, Le-Guen M, Girard M, Lu Q, Rouby JJ (2010) Ultrasound assessment of antibiotic-induced pulmonary reaeration in ventilator-associated pneumonia. Crit Care Med 38:84–92
Bouhemad B, Brisson H, Le-Guen M, Arbelot C, Lu Q, Rouby JJ (2011) Bedside ultrasound assessment of positive end-expiratory pressure-induced lung recruitment. Am J Respir Crit Care Med 183(3):341–347
Arbelot C, Ferrari F, Bouhemad B, Rouby JJ (2008) Lung ultrasound in acute respiratory distress syndrome and acute lung injury. Curr Opin Crit Care 14:70–74
Bouhemad B, Zhang M, Lu Q, Rouby JJ (2007) Clinical review: bedside lung ultrasound in critical care practice. Crit Care 11:205
Volpicelli G, Elbarbary M, Blaivas M, Lichtenstein DA, Mathis G, Kirkpatrick AW, Melniker L, Gargani L, Noble VE, Via G, Dean A, Tsung JW, Soldati G, Copetti R, Bouhemad B, Reissig A, Agricola E, Rouby JJ, Arbelot C, Liteplo A, Sargsyan A, Silva F, Hoppmann R, Breitkreutz R, Seibel A, Neri L, Storti E, Petrovic T, International Liaison Committee on Lung Ultrasound (ILC-LUS) for the International Consensus Conference on Lung Ultrasound (ICC-LUS) (2012) International evidence-based recommendations for point-of-care lung ultrasound. Intensive Care Med 38:577–591
Via G, Storti E, Gulati G, Neri L, Mojoli F, Braschi A (2012) Lung ultrasound in the ICU: from diagnostic instrument to respiratory monitoring tool. Minerva Anestesiol 78:1282–1296
Perbet S, Gerst A, Chabanne R, Soummer A, Faure JS, Pascal J, Pereira B, Rouby JJ, Constantin JM (2014) High-flow nasal oxygen cannula versus conventional oxygen therapy to prevent postextubation lung aeration loss: a multicentric randomized control lung ultrasound study. Oral session 0446. Abstracts ESICM LIVES 2014 27th Annual Congress Barcelona, Spain. Intensive Care Med 40(Suppl 1)
Oropello J, Rahamanian M (2013) Can chest sonography predict and facilitate successful ventilator weaning? Crit Care Med 41(8):2065–2067
Yang KL, Tobin MJ (1991) A prospective study of indexes predicting the outcome of trials of weaning from mechanical ventilation. N Engl J Med 324:1445–1450
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
The authors do not declare any conflict of interest.
Rights and permissions
About this article
Cite this article
Mayo, P., Volpicelli, G., Lerolle, N. et al. Ultrasonography evaluation during the weaning process: the heart, the diaphragm, the pleura and the lung. Intensive Care Med 42, 1107–1117 (2016). https://doi.org/10.1007/s00134-016-4245-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00134-016-4245-3