Abstract
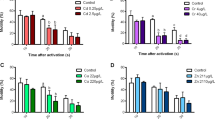
Present investigation aimed to assess the alterations in branchial architecture of Ctenopharyngodon idellus exposed to chlorpyrifos. 96 h LC50 of chlorpyrifos to C. idellus was found to be 7.24 µg/L. Fish were exposed chronically to 2.41 and 1.44 µg/L sublethal concentrations (ecologically relevant concentrations) of chlorpyrifos for 15, 30 and 60 days. The gills of the CPF exposed fish showed partially affected secondary lamellae with shortening and curling, increased inter-lamellar space, sloughed off epithelium, hyperplasia and necrosis of pavement cells. On 60 days exposure, more pronounced degenerative changes, including severe necrosis, hyperplasia and hypertrophy of pavement cells, eroded and uplifted lamellar epithelium were noticed. Ultrastructural alterations include distorted nucleus and mitochondria in pavement cells, marked contraction in cisternae of golgi complex. Particularly in chloride cells, abundant mitochondria with distorted shape and size, dilation in between their cristae and tubular network were observed. The severity of damage was more profound at 60 days exposure. Thus, the observed cytopathological lesions of fish gills can be used as sensitive biomarkers for pesticide contamination.


Similar content being viewed by others
Abbreviations
- LC50:
-
50% Lethal concentration
- CPF:
-
Chlorpyrifos
- OP:
-
Organophosphate
- GC:
-
Gas chromatography
- IAEC:
-
Institutional Animal Ethical Committee
- AIIMS:
-
All India Institute of Medical Sciences
- PBS:
-
Phosphate buffer solution
- PVC:
-
Pavement cells
- MRC:
-
Mitochondrial rich cells
- ER:
-
Endoplasmic reticulum
References
APHA (2012) Standard methods for the examination of water and waste water, 22nd edn. American Public Health Association, Washington
Bernet D, Schmidt-Posthaus H, Meir W, Burkhardt-Holm P, Wahli T (1999) Histopathology in fish: Proposed for a protocol to assess aquatic pollution. J Fish Dis 22:25–34
Boran H, Capkin E, Altinok I, Terzi E (2012) Assessment of acute toxicity and histopathology of the fungicide captan in rainbow trout. Exp Toxicol Pathol 64:175 – 79
Bradbury SP, Mekim JM, Loats RJ (1987) Physiological response of rainbow trout (Salmo gairdueri) to acute fenvalerate intoxication. Pest Biochem Physiol 27:275–288
Brunelli E, Mauceri A, Maisano M, Bernab I, Giannetto A, DeDoenico E, Corapi E, Tripepi S, Fasulo S (2011) Ultrastructural and immunohistochemical investigation on the gills of the teleost, Thalassoma pavo L. exposed to cadmium. Acta Histochem 113:201–213
Conte FP (1969) Salt secretion. In: Hoar WS, Randall DJ (eds) Fish physiology, vol 1. Academic Press, New York, pp 241–283
Deb N, Das S (2013) Chlorpyrifos toxicity in fish: a review. Curr World Environ 8(1):77–84
Evans DE, Piermarini PM, Choe KP (2005) The multifunctional fish gill: dominant site of gas exchange. Physiol Rev 85:97–177
Fernandes MN, Mazon AF (2003) Environmental pollution and fish gill morphology. In: Val AL, Kapoor BG (eds) Fish adaptations. Science Publishers, Enfield, pp 203–231
Finney DJ (1980) Probit analysis, 3rd edn. Cambridge University Press, London
Fuat MG, Talas ZS, Erdogan K, Orun I (2014) The effects of propolis on gill, liver, muscle tissues of rainbow trout (Oncorhynchus mykiss) exposed to various concentrations of cypermethrin. Iran J Fish Sci 13(3):684–701
Ghasemzadeh J, Sinaei M, Bolouki M (2015) Biochemical and histological changes in fish, spotted scat (Scatophagus argus) exposed to diazinon. Bull Environ Contam Toxicol 94(2):164–170
Kaur M, Jindal R (2016) SEM study of ultrastructural changes in branchial architecture of Ctenopharyngodon idella (Cuvier & Valenciennes) exposed to chlorpyrifos. Arch Biol Sci 68(2):393–398
Kaur M, Jindal R (2017) Oxidative stress response in liver, kidney and gills of Ctenopharyngodon Idellus (Cuvier & Valenciennes) exposed to chlorpyrifos. MOJ Biol Med 1(4):00021. https://doi.org/10.15406/mojbm.2017.01.00021
Kaur R, Johal MS, Sandhu GS (2005) Histological changes in the branchial architecture of Heteropneustes fossilis induced by fenvalerate, a synthetic pyrethroid. Indian J Animal Sci 75:1159–1162
Lee JW, Kim JE, Shin YJ, Ryu JS, Eoma IC, Lee JS, Kim Y, Kim PJ, Choi KH, Lee BC (2014) Serum and ultrastructure responses of common carp (Cyprinus carpio L.) during long-term exposure to zinc oxide nanoparticles. Ecotoxicol Environ Saf 104:9–17
Magare SR, Patil HT (2000) Effects of pesticides on oxygen consumption, red blood cell count and metabolites of a fish, Punctius ticto. Environ Ecol 18:891–894
Marutirao GJ (2012) Histopathological changes in the gills of Puntius ticto (Ham.) under dimethoate toxicity. The Bioscan 7(3):423–426
Myers MS, Johnson LL, Hom T, Collier TK, Stein JE, Varanasi U (1998) Toxicopathic hepatic lesions in sub adult English sole (Pleuronectes vetulus) from Puget Sound, Washington, USA: relationship with other biomarkers of contaminant exposure. Mar Environ Res 45:47–67
Nowak B (1992) Histological changes in gills induced by residue of endosulfan. Aqua Toxicol 23(1):65–84
Prusty AK, Meena DK, Mohapatra S, Das P, Gupta SK, Behera BK (2015) Synthetic pyrethroids (Type II) and freshwater fish culture: perils and mitigations. Aquat Res 7:163
Rajbanshi KG (1995) Aquaculture extension services review: Nepal. FAO Fisheries Circular No. 896. FAO, Rome
Reynolds ES (1963) The use of lead citrate at high pH as an electron opaque stain in electron microscopy. J Cell Biol 17:208–212
Sawhney AK. Johal MS (2000) Effects of an organophosphorus insecticide, malathion, on pavement cells of the gill epithelia of Channa punctatus (Bloch). Pol Arch Hydrobiol 47(2):195–203
Schlenk D, Handy R, Steinert S, Depledge MH, Benson W (2008) Biomarkers. In: Di Giulio RT, Hinton DE (eds) The toxicology of fishes. CRC Press, Boca Raton, FL, pp 683–732
Schwaiger J, Ferling H, Mallow U, Wintermagr H, Negele RD (2004) Toxic effects of the non-steroidal anti-inflammatory drug diclofenac. Part I: histopathological alterations and bioaccumulation in rainbow trout. Aquat Toxicol 68:141–150
Theurkar SV, Gaikwad AN, Ghadage MK, Patil SB (2014) Impact of monocrotophos on the histopathological changes in the gills of mosquito fish, Gambusia affinis. Res J Recent Sci 3:29–32
Tilak KS, Koteswara RD, Veeraiah K (2005) Effects of chlorpyrifos on histopathology of fish Catla catla. J Ecotoxicol Environ Monit 15(2):127–140
Velisek J, Stara A, Machova J, Svobodova Z (2012) Effects of long-term exposure to simazine in real concentrations on common carp (Cyprinus carpio L.). Ecotoxicol Environ Saf 76:79–86
Wannee JK, Upatham ES, Kruatrachue M, Sahaphong S, Vichasri-Grams S, Prayad P (2002) Histopathological effects of round up, a glyphosate herbicide, on Nile tilapia Oreochromis niloticus. Sci Asia 28:121–127
Acknowledgements
Authors are thankful to the Chairperson, Department of Zoology, Panjab University, Chandigarh for providing necessary research facility, AIIMS, New Delhi for TEM analyses and to the University Grants Commission, New Delhi, India for providing financial assistance to Mandeep Kaur to carry out this work.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kaur, M., Jindal, R. Toxicopathic Branchial Lesions in Grass Carp (Ctenopharyngodon idellus) Exposed to Chlorpyrifos. Bull Environ Contam Toxicol 100, 665–671 (2018). https://doi.org/10.1007/s00128-018-2320-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00128-018-2320-x