Abstract
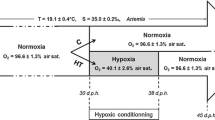
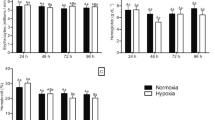
Hypoxia often occurs in aquatic environments as dissolved oxygen (DO) concentration decrease to a level where it is detrimental to aquatic organisms. To investigate the effects of hypoxia on the physiology of zebrafishes (Danio rerio), the organisms were kept at normoxic conditions (DO: 8 ± 0.3 mg L−1: control group) or were subjected to mild (DO: 3 ± 0.3 mg L−1) or severe hypoxia (DO: 1 ± 0.2 mg L−1) for 48 h and a subsequent restoration of oxygen concentrations (DO: 8 ± 0.3 mg L−1) for another 96 h at 25°C. We found that the enzyme activities show different initial responses, acclimation and recovery to severe hypoxia relative to normoxic conditions, but no significant difference was observed between normoxic conditions and mild hypoxia. The results suggest that zebrafishes can acclimate to the mild hypoxia (3 mg L−1) quickly but oxidative damage would occur when DO decreased below 1 mg L−1. Our findings could be useful for water resource managers to set protection limits of DO for aquatic organisms.



Similar content being viewed by others
References
Barrionuevo WR, Burggren WW (1999) O2 consumption and heart rate in developing zebrafish (Danio rerio): influence of temperature and ambient O2. Am J Physiol 276(2 Pt 2):R505–R513
Barrionuevo W, Fernandes M, Rocha O (2010) Aerobic and anaerobic metabolism for the zebrafish, Danio rerio, reared under normoxic and hypoxic conditions and exposed to acute hypoxia during development. Braz J Biol 70:425–434
Blokhina O, Virolainen E, Fagerstedt KV (2003) Antioxidants, oxidative damage and oxygen deprivation stress: a review. Ann Bot 91:179–194
Bradford MM (1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72:248–254
Choi SB, Normi YM, Wahab HA (2011) Revealing the functionality of hypothetical protein KPN00728 from Klebsiella pneumonia MGH78578: molecular dynamics simulation approaches. BMC Bioinform 12(Suppl 13):S11
Claiborne A (1985) Catalase activity. In: Greenwald RA (ed) CRC handbook of methods in oxygen radical research. CRC Press, Boca Raton
Cochran RE, Burnett LE (1996) Respiratory responses of the salt marsh animals, Fundulus heteroclitus, Leiostomus xanthurus, and Palaemonetes pugio to environmental hypoxia and hypercapnia and to the organophosphate pesticide, azinphosmethyl. J Exp Mar Biol Ecol 195:125–144
Cooper RU, Clough LM, Farwell MA (2002) Hypoxia-induced metabolic and antioxidant enzymatic activities in the estuarine fish Leiostomus xanthurus. J Exp Mar Biol Ecol 279:1–20
Dalla VJ, Van den Thillart G, Cattani O, Cortesi P (1998) Behavioural responses and biochemical correlates in Solea solea to gradual hypoxic exposure. Can J Zool 76:108–2113
Dong M, Zhu LS, Zhu SY, Wang JH, Wang J, Xie H, Du ZK (2013) Toxic effects of 1-decyl-3-methylimidazolium bromide ionic liquid on the antioxidant enzyme system and DNA in zebrafish (Danio rerio) livers. Chemosphere 91:1107–1112
Flohe L, Otting F (1984) Superoxide dismutase assays. Methods Enzymol 105:93–104
Giordano G, Kavanagh TJ, Costa LG (2008) Neurotoxicity of a polybrominated diphenyl ether mixture (DE-71) in mouse neurons and astrocytes is modulated by intracellular glutathione levels. Toxicol Appl Pharmacol 232:161–168
Hou H, Li B, Zhao X, Zhuang Y, Ren G, Yan M, Cai Y, Zhang X, Chen L (2009) The effect of pacific cod (Gadus macrocephalus) skin gelatin polypeptides on UV radiation-induced skin photoaging in ICR mice. Food Chem 115:945–950
Hu XR, Zhou XY, He B, Xu CW, Wu L, Cui B, Wen HZ, Lu ZB, Jiang H (2010) Minocycline protects against myocardial ischemia and reperfusion injury by inhibiting high mobility group box 1 protein in rats. Eur J Pharmacol 638:84–89
Jensen FB, Nikinmaa M, Weber RE (1993) Environmental perturbations of oxygen transport in teleost fishes: causes, consequences and compensations. Fish Ecophysiol 9:161–179
Lushchak VI (2011) Environmentally induced oxidative stress in aquatic animals. Aquat Toxicol 101:13–30
Lushchak VI, Bagnyukova TV (2007) Hypoxia induces oxidative stress in tissues of a goby, the rotan Perccottus glenii. Comp Biochem Physiol B Biochem Mol Biol 148:390–397
Lushchak VI, Lushchak LP, Mota AA, Hermes-Lima M (2001) Oxidative stress and antioxidant defenses in goldfish Carassius auratus during anoxia and reoxygenation. Am J Physiol 280:R100–R107
Mandic M, Todgham AE, Richards JG (2009) Mechanisms and evolution of hypoxia tolerance in fish. Proc R Soc B 276:735–744
Marcelo HL, Tania ZS (2002) Animal response to drastic changes in oxygen availability and physiological oxidative stress. Comp Biochem Physiol C Toxicol Pharmacol 133:537–556
McKenzie DJ, Steffensen JF, Korsmeyer K, Whiteley NM, Bronzi P, Taylor EW (2007) Swimming alters responses to hypoxia in the Adriatic sturgeon Acipenser naccarii. J Fish Biol 70:651–658
Muggeo VMR (2008) Segmented: an R package to fit regression models with broken-line relationships. R News 8:20–25
Nilsson GE, Renshaw GMC (2004) Hypoxic survival strategies in two fishes: extreme anoxia tolerance in the North European crucian carp and natural hypoxic preconditioning in a coral-reef shark. J Exp Biol 207:3131–3139
Olsvik PA, Kristensen T, Waagb R, Rosseland BO, Tollefsen KE, Baeverfjord G, Berntssen MH (2005) mRNA expression of antioxidant enzymes (SOD, CAT and GSH-Px) and lipid peroxidative stress in liver of Atlantic salmon (Salmo salar) exposed to hyperoxic water during smoltification. Comp Biochem Physiol C 141:314–323
Parrilla TDP, Zenteno ST (2011) Antioxidant enzyme activities in Pacific white shrimp (Litopenaeus vannamei) in response to environmental hypoxia and reoxygenation. Aquaculture 318:379–383
Raymond EE, Larissa BP, Andrew AR, David MP (2007) Zebrafish in the wild: a review of natural history and new notes from the field. Zebrafish 4:21–40
Rees BB, Sudradjat FA, Love JW (2001) Acclimation to hypoxia increases survival time of zebrafish, Danio rerio, during lethal hypoxia. J Exp Zool 289:266–272
Richards JG (2009) Metabolic and molecular responses of fish to hypoxia. In: Richards JG, Farrell AP, Brauner CJ (eds) Hypoxia, vol 27. Elsevier, San Diego, pp 443–485
Salas-Leiton E, Cánovas-Conesa B, Zerolo R, López-Barea J, Cañavate JP, Alhama J (2009) Proteomics of juvenile Senegal sole (Solea senegalensis) affected by gas bubble disease in hyperoxygenated ponds. Mar Biotechnol 11:473–487
Thomas PM, Pankhurst NW, Bremner HA (1999) The effect of stress and exercise on post-mortem biochemistry of Atlantic salmon and rainbow trout. J Fish Biol 54:1177–1196
US Environmental Protection Agency (1986) Ambient water quality criteria for dissolved oxygen. EPA 440/5-86-003. USEPA, Criteria and Standards Division, Washington, DC
Zhang W, Cao ZD, Peng JL, Chen BJ, Fu SJ (2010) The effects of dissolved oxygen level on the metabolic interaction between digestion and locomotion in juvenile southern catfish (Silurus meridionalis Chen). Comp Biochem Physiol A Mol Integr Physiol 157:212–219
Acknowledgments
We thank the two anonymous reviewers for highly professional critical analysis of the manuscript that helped substantially improve the work. This research was funded by the Major Science and Technology Program for Water Pollution Control and Treatment (2012ZX07501-003) and Tianjin Research Program of Application Foundation and Advanced Technology (15JCYBJC22800).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Feng, J., Guo, Y., Gao, Y. et al. Effects of Hypoxia on the Physiology of Zebrafish (Danio rerio): Initial Responses, Acclimation and Recovery. Bull Environ Contam Toxicol 96, 43–48 (2016). https://doi.org/10.1007/s00128-015-1668-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00128-015-1668-4