Abstract
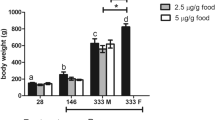
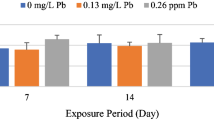
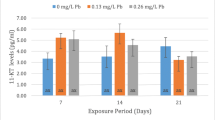
This study describes the effects of 17-alpha-ethynylestradiol (EE2) on the structure of the excretory system of the kidney in tench. Adult male tench were exposed to sub-lethal doses of EE2 (50, 100 and 500 μg/kg b.w.) under semistatic conditions for a period of 30 days. The nephrosomatic index and histology (including a morphometric analysis) of the kidney were examined. Histopathological lesions in the kidney of exposed tench were: dilation of glomerular capillaries and increase in the area of the renal corpuscle, hyaline degeneration in the epithelial cells of the proximal tubules leading to necrotic changes, hemorrhages in the interstitial tissue and deposits of eosinophilic material. These lesions were observed with a greater degree of severity as the exposure doses were increased. These results indicate that long-term exposure to EE2 could produce clear negative effects on the excretory system of the kidney in tench and consequently on their physiological functions.

Similar content being viewed by others
References
Caille N, Rodina M, Kocour M, Gela D, Flajšhans M, Linhart O (2006) Quantity, motility and fertility of tench Tinca tinca (L.) sperm in relation to LHRH analogue and carp pituitary treatments. Aquacult Int 14:75–87
Ferguson HW (1989) Systemic pathology of fish. A text and atlas of comparative tissue responses in diseases of teleosts, 1st edn. Iowa State University Press, Ames
Herman RL, Kincaid HL (1998) Pathological effects of orally administered estradiol to rainbow trout. Aquaculture 72:165–172
Hibiya T (1982) Atlas of fish histology. Normal and pathological features. Editorial Kodhansa Scientific Books, Tokyo
Länge R, Hutchinson TH, Croudace CP, Siegmund F, Schweinfurth H, Hampe P, Panter GH, Sumpter JP (2001) Effects of the synthetic estrogen 17α-ethynylestradiol on the life-cycle of the fathead minnow (Pimephales promelas). Environ Toxicol Chem 20:1216–1227
Larsson DGJ, Adolfsson-Erici M, Parkkonen J, Pettersson M, Berg AH, Olsson PE, Förlin L (1999) Ethinyloestradiol—an undesired fish contraceptive. Aquat Toxicol 45:91–97
López de Alda MJ, Gil A, Paz E, Barceló D (2002) Ocurrence and analysis of estrogens and progestogens in river sediments by liquid chromatography-electrospray-mass spectrometry. Anal 127:1299–1304
Magri MacMahon AF (1946) Fishlore. Pelican Books, Gretna
Palace VP, Wautier KG, Evans RE, Blanchfield P, Mills KH, Chalanchuk SM, Godard D, McMaster ME, Tetreault GR, Peters LE, Vandenbyllaardt L, Kidd KA (2006) Biochemical and histopathological effects in pearl dace (Margariscus margarita) chronically exposed to a synthetic estrogen in a whole lake experiment. Environ Toxicol Chem 25(4):1114–1125
REAL DECRETO 1201/2005, de 10 de octubre, sobre protección de los animales utilizados para experimentación y otros fines científicos. B.O.E. no 252. Ministerio de la Presidencia. Madrid (Spain)
Schwaiger J, Spieser OH, Bauer C, Ferling H, Mallow U, Kalbfus W, Negele RD (2000) Chronic toxicity of nonylphenol and ethynylestradiol: haematological and histopathological effects in juvenile common carp (Cyprinus carpio). Aquat Toxicol 51:69–78
Tong J, Yu X, Liao X, Wang J, Hu S, Guan M, Linhart O, Flajshans M (2002) Preliminary survey of genetic variation of tench (Tinca tinca) from the Czech Republic and China. In: Proceedings XXth genetic days 303–304
Weber LP, Hill RL, Janz DM (2003) Developmental estrogenic exposure in zebrafish (Danio rerio): II. Histological evaluation of gametogenesis and organ toxicity. Aquat Toxicol 63:431–446
Xu J, Wu LS, Chen WP, Jiang PP, Chang ACS (2009) Pharmaceuticals and personal care products (PPCPs), and endocrine disrupting compounds (EDCs) in runoff from a potato field irrigated with treated wastewater in southern California. J Health Sci 55:306–310
Zar JH (1996) Biostatistical analysis. Prentice Hall International Editions, New Jersey
Zaroogian G, Gardner G, Horowitz BD, Gutjahr-Gobell R, Haebler R, Mills L (2001) Effect of 17β-estradiol, o, p-DDT, octylphenol and p, p-DDE on gonadal development and liver and kidney pathology in juvenile male summer flounder (Paralochthys dentatus). Aquat Toxicol 54:101–112
Zha J, Wang Z, Wang N, Ingersoll C (2007) Histological alternation and vitellogenin induction in adult rare minnow (Gobiocypris rarus) after exposure to ethynylestradiol and nonylphenol. Chemosphere 66:488–495
Zhang X, Li QZ, Li GX, Wang ZS, Yan CZ (2009) Levels of estrogenic compounds in Xiamen Bay sediment, China. Mar Pollut Bull 58:1210–1216
Acknowledgments
This study was supported by the University of Extremadura (Plan de iniciación a la investigación, desarrollo tecnológico e innovación. 2008), Spain.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Oropesa, A.L., Jiménez, B., Fallola, C. et al. Histological Alterations on the Structure of the Excretory Renal System in Tench (Tinca tinca) After Exposure to 17-Alpha-Ethynylestradiol. Bull Environ Contam Toxicol 91, 623–629 (2013). https://doi.org/10.1007/s00128-013-1088-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00128-013-1088-2