Abstract
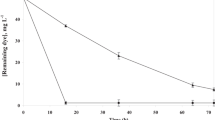
The study was conducted by a novel salt tolerant bacterium Gracilibacillus sp. GTY. The strain was identified on the basis of morphological and physio-biochemical characteristics, and 16S rDNA sequence analysis. Decolorization was performed by growing and resting cells, as well as by extracted azo reductase. Strain grown in the media containing 15% (w/v) of NaCl showed the best performance in decolorization. Decolorization was observed by the UV–visible absorbance spectra. The maximum absorption peak in the visible area decreased to a minimum level after 96 h of incubation. On the other hand, strain grown in very low, or high concentrations, of salt did not show good performance in decolorization; suggesting that salt concentrations in the surroundings control the production of azo reductase.






Similar content being viewed by others
References
Chung KT, Stevens Jr SE (1993) Degradation of azo dyes by environmental microorganisms and helminthes. Environ Toxicol Chem 12:2121–2132
Hamid-Reza KH, Akihiko Sakurai MS (2007) Decolorization of synthetic dyes by a new manganese peroxidase-producing white rot fungus. Dyes Pigments 72:157–162
Haugh W, Schmidt A, Norman B, Hempel DC, Stolz A, Knackmuss HJ (1991) Mineralization of the sulfonated azo dye mordant yellow 3 by a 6-aminonaphthalene-2-sulfonate-degrading bacterial consortium. Appl Environ Microbiol 57:3144–3149
Medvedev ZA, Crowne HM, Medvedeva MN (1988) Age related variations of hepatocarcinogenic effect of azo dye (3′-MDAB) as liked to the level of hepatocyte polyploidization. Mech Ageing Dev 46:159–174
Neilan BA (1995) Identification and phylogenetic analysis of toxigenic cyanobacteria using a multiplex RAPD-PCR. Appl Environ Microbiol 61:2286–2291
O’Neil C, Lopez A, Esteves S, Hawkes FR, Hawkes DL, Wilcox S (2000) Azo-dye degradation in an anaerobic-aerobic treatment system operating on simulated textile effluent. Appl Microbiol Biotechnol 53:249–254
Panswad T, Anan C (1999) Specific oxygen, ammonia, and nitrate uptake rates of a biological nutrient removal process treating elevated salinity wastewater. J Bioresour Technol 70:237–243
Percy AJ, Moore N, Chipman JK (1989) Formation of nuclear anomalies in rat intestine by benzidine and its biliary metabolites. Toxicology 57:217–223
Rafii F, Cerniglia CE (1993) Localization of the azo reductase of Clostridium perfringens by immuno-electron microscopy. Curr Microbiol 27:143–145
Weber EJ (1991) Studies of benzidine-based dyes in sediment-water systems. Environ Toxicol Chem 9:609–614
Zhilong W, Fengsheng Z, Daijie C, Daotang L (2006) Biotransformation of phytosterol to produce androsta-diene-dione by resting cells of Mycobacterium in cloud point system. Process Biochemistry 41:557–561
Acknowledgments
We are grateful to the Chinese Ministry of Education for the financial support of this study. We also thank Professor Zhou Jiti for his scientific and technical advice and providing us the laboratory facility.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Uddin, M.S., Zhou, J., Qu, Y. et al. Biodecolorization of Azo Dye Acid Red B under High Salinity Condition. Bull Environ Contam Toxicol 79, 440–444 (2007). https://doi.org/10.1007/s00128-007-9260-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00128-007-9260-1