Abstract
Objective
The aim of this study was to investigate the association of the common apolipoprotein E gene (APOE) variants with cognitive function and cognitive decline in adult mid-life and explore the possibility that APOE genotype mediates the link between socioeconomic status (SES) and cognitive function.
Methods
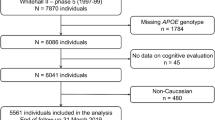
Data on cognitive function, as measured by five cognitive tests, together with APOE genotype were obtained in an occupational cohort (the Whitehall II study) of 6,004 participants aged 44–69 years (1997–1999). Cognitive change was examined in 2,717 participants who had cognitive function measured at baseline (1991–1993).
Results
SES based on civil service employment grade was strongly related to cognitive function. There was no association between APOE genotype and employment grade. In women, participants with APOE-ɛ4 had a lower memory score (p<0.05), but the result was sensitive to data from a small number of individuals. A marginal cross-sectional difference in the semantic fluency score was found (p=0.07), and there was a relative decline at follow-up (p<0.001, net change=−1.19; 95% CI, −1.90 to −0.49) in those with APOE-ɛ4 genotypes.
Conclusions
APOE-ɛ4 has little influence on cognitive decline in mid-life, whereas SES is a strong determinant, although APOE genotype may emerge as an important factor in cognitive function in later life.
Similar content being viewed by others
References
Aartsen MJ, Martin M, Zimprich D (2004) Gender differences in level and change in cognitive functioning. Results from the longitudinal aging study Amsterdam. Gerontology 50:35–38
Anstey K, Christensen H (2000) Education, activity, health, blood pressure and apolipoprotein E as predictors of cognitive change in old age: a review. Gerontology 46:163–177
Bartres-Faz D, Junqu C, Moral P et al (2002) Apolipoprotein E gender effects on cognitive performance in age associated memory impairment. J Neuropsychiatry Clin Neurosci 14:80–83
Bolla MK, Wood N, Humphries SE (1999) Rapid determination of apolipoprotein E genotype using a heteroduplex generator. J Lipid Res 40:2340–2345
Borkowski JG, Benton AL, Spreen O (1967) Word fluency and brain damage. Neuropsychologia 5:135–140
Deary IJ, Whiteman MC, Pattie A et al (2002) Cognitive change and the APOE e4 allele. Nature 418:932
de Geus EJC, Wright MJ, Martin NG et al (2001) Genetics of brain function and cognition. Behav Genet 31:489–495
Dik MGC, Jonker MD, Comijs HC et al (2001) Memory complaints and APOE-4 accelerate cognitive decline in cognitively normal elderly. Neurology 57:2217–2222
Ewbank D (2001) Demography in the age of genomics: a first look at the prospects. In: Finch CE, Vaupel JW, Kinsella K (eds) Cells and surveys: should biological measures be included in social science research? The National Academies Press
Feskens EJ, Havekes LM, Kalmjin S et al (1994) Apolipoprotein e4 allele and cognitive decline in elderly men. BMJ 309:1202–1206
Fuhrer R, Head J, Marmot MG (1999) Social position, age and memory performance in the Whitehall II study. Ann NY Acad Sci 896:359–362
Gallacher JEJ, Elwood PC, Hopkinson C et al (1999) Cognitive function in the Caerphilly study: association with age, social class, education and mood. Eur J Epidemiol 15:161–169
Goldberg DP (1972) The detection of psychiatric illness by questionnaire. Oxford University Press, Oxford
Haan MN, Shemanski L, Jagust WJ et al (1999) The role of APOE e4 in modulating effects of other risk factors for cognitive decline in elderly persons. JAMA 281:40–46
Hallman DM, Boerwinkle E, Saha N et al (1991) The apolipoprotein E polymorphism: a comparison of allele frequencies and effects in nine populations. Am J Hum Genet 49:338–349
Heim AW (1970) AH4 group test of general intelligence ASE. NFER-Nelson Publishing
Helkala EL, Koivisto K, Hannien T et al (1996) Memory function in human subjects with different apolipoprotein E phenotypes during a 3-year population-base study. Neurosci Lett 204:177–180
Hofer SM, Christensen H, Mackinnon AJ et al (2002) Change in cognitive functioning associated with ApoE genotype in a community sample of older adults. Psychol Aging 17:194–208
Hyman BT, Gomez-Isla T, Briggs M et al (1996) Apolipoprotein E and cognitive changes in an elderly population. Ann Neurol 40:55–66
Jacqmin-Gadda H, Fabrigoule C, Commenges D et al (1997) A 5-year longitudinal study of the mini-mental state examination in normal aging. Am J Epidemiol 145:498–506
Jonker C, Schmand B, Lindeboom J et al (1998) Association between apolipoprotein E epsilon 4 and the rate of cognitive decline in community-dwelling elderly individuals with and without dementia. Arch Neurol 55:1065–1069
Juva K, Verkkoniemi A, Viramo P et al (2000) APOE epsilon4 does not predict mortality, cognitive decline, or dementia in the oldest old. Neurology 54:412–415
Kalmijn S, Feskens FJ, Launer LJ et al (1997) Longitudinal study of the effect of apolipoprotein e4 allele on the association between education and cognitive decline in elderly men. BMJ 314:34–35
Korten AE, Henderson AS, Christensen H et al (1997) A prospective study of cognitive function in the elderly. Psychol Med 27:919–930
Marmot MG, Smith GD, Stansfeld S et al (1991) Health inequalities among British civil servants: the Whitehall II study. Lancet 337:1387–1393
Mayeux R, Small SA, Tang MX et al (2001) Memory performance in healthy elderly without Alzheimer’s disease: effects of time and apolipoprotein-E. Neurobiol Aging 22:683–689
Miller SA, Dykes DD, Polesky HF (1998) A simple salting out procedure for extracting DNA from human nucleated cells. Nucleic Acids Res 16:1215
Mortensen EL, Hogh P (2001) A gender difference in the association between APOE genotype and age related cognitive decline. Neurology 57:89–95
O’Hara R, Yesavage JA, Kraemer HC et al (1998) The APOE-e4 allele is associated with decline on delayed recall performance in community-dwelling adults. J Am Geriatr Soc 46:1493–1498
Pendleton N, Payton A, van den Boogerd EH et al (2002) Apolipoprotein E genotype does not predict decline in intelligence in healthy older adults. Neurosci Lett 324:74–76
Rabbitt P, Diggle P, Smith D et al (2001) Identifying and separating the effects of practice and of cognitive ageing during a large longitudinal study of elderly community residents. Neuropsychologia 39:532–543
Raven JC (1965) Guide to using the Mill Hill vocabulary scale with progressive matrices. HK Lewis
Richards M, Sacker A (2003) Lifetime antecedents of cognitive reserve. J Clin Exp Neuropsychol 25:614–624
Rosenberg RN (2000) The molecular and genetic basis of AD: the end of the beginning. The 2000 Wartenberg lecture. Neurology 54:2045–2054
Saunders AM, Strittmatter WJ, Schmechel D et al (1993) Association of apolipoprotein E allele e4 with late-onset familial and sporadic Alzheimer’s disease. Neurology 43:1467–1472
Singh-Manoux A, Richards M, Marmot M (2003) Leisure activities and cognitive function in middle age: evidence from the Whitehall II study. J Epidemiol Community Health 57:907–913
Small BJ, Graves AB, McEvoy CL et al (2000) Is APOE-e4 a risk factor for cognitive impairment in normal aging? Neurology 54:2082–2088
Turrell G, Lynch JW, Kaplan GA et al (2002) Socioeconomic position across the lifecourse and cognitive function in late middle age. J Gerontol Ser B Psychol Sci Soc Sci 57:S43–S51
Wilson JA, Grove WR (1999) The age-period-cohort conundrum and verbal ability: empirical relationships and their interpretation: reply to Glenn and Alwin and McCammon. Am Sociol Rev 64:287–302
Winnock M, Letenneur L, Jacqmin-Gadda H et al (2002) Longitudinal analysis of the effect of apolipoprotein E epsilon4 and education on cognitive performance in elderly subjects: the PAQUID study. J Neurol Neurosurg Psychiatry 72:794–797
Acknowledgements
The Whitehall II study has been supported by grants from the Medical Research Council; British Heart Foundation; Health and Safety Executive; Department of Health; National Heart Lung and Blood Institute (HL36310), US, NIH: National Institute on Aging (AG13196), US, NIH; Agency for Health Care Policy Research (HS06516); and the John D and Catherine T MacArthur Foundation Research Networks on Successful Midlife Development and Socioeconomic Status and Health. M.M. is supported by an MRC Research Professorship. S.E.H, P.S.T. and E.H. are supported by the British Heart Foundation (RG2000/015). We also thank all participating civil service departments and their welfare, personnel and establishment officers; the Occupational Health and Safety Agency; the Council of Civil Service Unions; and all participating civil servants in the Whitehall II study: Paul Clarke, Martin Shipley, Susan Yazdgerdi and other members of the Whitehall II study team.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhao, J.H., Brunner, E.J., Kumari, M. et al. APOE polymorphism, socioeconomic status and cognitive function in mid-life. Soc Psychiat Epidemiol 40, 557–563 (2005). https://doi.org/10.1007/s00127-005-0925-y
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00127-005-0925-y