Abstract
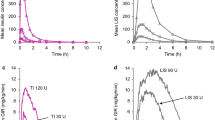
Aims/hypothesis. The AERx insulin Diabetes Management System (AERx iDMS) is a liquid aerosol device that enables insulin to be administered to the peripheral parts of the lung. This study aimed to compare the pharmacokinetic and pharmacodynamic properties of insulin which is inhaled using AERx iDMS with insulin which is subcutaneously administered. Methods. In total, 18 C-peptide negative patients with Type I (insulin-dependent) diabetes mellitus participated in this randomised, open-label, 5-period cross-over trial. Human regular insulin was administered subcutaneously (0.12 U/kg body weight) or inhaled by means of the AERx iDMS (dosages 0.3, 0.6, 1.2, and 1.8 U/kg body weight). Thereafter plasma glucose was kept constant at 7.2 mmol/l for a 10-h period (glucose clamp technique). Results. Inhaled insulin provided a dose-response relation that was close to linear for both pharmacokinetic (AUC-Ins(0–10 h); Cmax-Ins) and pharmacodynamic (AUC-GIR(0–10 h); GIRmax) parameters. Time to maximum insulin concentration (Tmax-Ins) and time to maximum glucose infusion rate (TGIRmax) were shorter with inhaled insulin than with subcutaneous administration. The pharmacodynamic system efficiency of inhaled insulin (AUC-GIR(0–6 h)) was 12.7 % (95 % C. I.: 10.2–15.6). Conclusion/interpretation. The inhalation of soluble human insulin using the AERx iDMS is feasible and provides a clear dose response. Further long-term studies are required to investigate safety aspects, HbA1 c values, incidence of hypoglycaemic events and the quality of life. [Diabetologia (2001) 44: 305–308]
Article PDF
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Received: 18 July 2000 and in revised form: 25 October 2000
Rights and permissions
About this article
Cite this article
Brunner, G., Balent, B., Ellmerer, M. et al. Dose-response relation of liquid aerosol inhaled insulin in Type I diabetic patients. Diabetologia 44, 305–308 (2001). https://doi.org/10.1007/s001250051618
Issue Date:
DOI: https://doi.org/10.1007/s001250051618