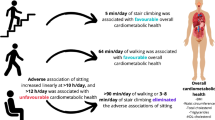
Abstract
Aims/hypothesis
The associations of sitting, standing, physical activity and sleep with cardiometabolic health and glycaemic control markers are interrelated. We aimed to identify 24 h time-use compositions associated with optimal metabolic and glycaemic control and determine whether these varied by diabetes status.
Methods
Thigh-worn activPAL data from 2388 participants aged 40–75 years (48.7% female; mean age 60.1 [SD = 8.1] years; n=684 with type 2 diabetes) in The Maastricht Study were examined. Compositional isometric log ratios were generated from mean 24 h time use (sitting, standing, light-intensity physical activity [LPA], moderate-to-vigorous physical activity [MVPA] and sleeping) and regressed with outcomes of waist circumference, fasting plasma glucose (FPG), 2 h plasma glucose, HbA1c, the Matsuda index expressed as z scores, and with a clustered cardiometabolic risk score. Overall analyses were adjusted for demographics, smoking, dietary intake and diabetes status, and interaction by diabetes status was examined separately. The estimated difference when substituting 30 min of one behaviour with another was determined with isotemporal substitution. To identify optimal time use, all combinations of 24 h compositions possible within the study footprint (1st–99th percentile of each behaviour) were investigated to determine those cross-sectionally associated with the most-optimal outcome (top 5%) for each outcome measure.
Results
Compositions lower in sitting time and with greater standing time, physical activity and sleeping had the most beneficial associations with outcomes. Associations were stronger in participants with type 2 diabetes (p<0.05 for interactions), with larger estimated benefits for waist circumference, FPG and HbA1c when sitting was replaced by LPA or MVPA in those with type 2 diabetes vs the overall sample. The mean (range) optimal compositions of 24 h time use, considering all outcomes, were 6 h (range 5 h 40 min–7 h 10 min) for sitting, 5 h 10 min (4 h 10 min–6 h 10 min) for standing, 2 h 10 min (2 h–2 h 20 min) for LPA, 2 h 10 min (1 h 40 min–2 h 20 min) for MVPA and 8 h 20 min (7 h 30 min–9 h) for sleeping.
Conclusions/interpretation
Shorter sitting time and more time spent standing, undergoing physical activity and sleeping are associated with preferable cardiometabolic health. The substitutions of behavioural time use were significantly stronger in their associations with glycaemic control in those with type 2 diabetes compared with those with normoglycaemic metabolism, especially when sitting time was balanced with greater physical activity.
Graphical Abstract

Similar content being viewed by others
Avoid common mistakes on your manuscript.

Introduction
Guidelines for 24 h movement [1, 2] are based on evidence that behaviours composing a day (sitting, standing, physical activity and sleep) can have interrelated contributions to health. Changing time spent in one of these behaviours will necessarily change the time spent in another. While the 24 h guidelines have been informed by a broad body of evidence [3], a commonly referenced limitation is a lack of relevant findings from studies employing a compositional analytic approach [2, 4]. Identifying the optimal balance of 24 h behavioural time-use compositions (sitting, standing, physical activity and sleeping) and the relationships of indicators of cardiometabolic health and glycaemic control with compositional techniques, can further inform 24 h guidelines and provide more precise targets for the improvement of disease risk and management of diseases such as type 2 diabetes.
Continuous measurement approaches, such as those collected via thigh-worn accelerometers, facilitate the investigation of 24 h free-living behaviours. Compositional data analysis (CoDA) appropriately considers the time spent in these behaviours as relative to one another and as having interrelated influence on health outcomes. Although well established in other fields, such as geochemistry [5] and nutrition [6], the application of CoDA is relatively new to physical activity and sedentary behaviour fields [7]. There has been limited application of this methodology in understanding different risk profiles, including in people with, or at risk of, type 2 diabetes [4, 8]. There is a need to evaluate the health risks of excess sedentary behaviour [9], low physical activity [10, 11] or inadequate sleep [12] as having interrelated implications for disease risk and disease management.
To address the evidence gaps, we examined associations of compositions of sitting, standing, light-intensity physical activity (LPA), moderate-to-vigorous physical activity (MVPA) and sleep time with cardiometabolic risk and glycaemic control markers in a large sample of adults using thigh-worn accelerometers. Associations were examined overall, as well as by diabetes status (normoglycaemia, impaired glucose metabolism [IGM], type 2 diabetes) and sex. The compositions associated with more-optimal benefits, for all cardiometabolic and glycaemic control markers, were also investigated. It was hypothesised that compositions with longer sitting time would be adversely associated with cardiometabolic parameters, while longer standing and physical activity time would be beneficially associated with cardiometabolic parameters, and that these associations would be stronger in people with type 2 diabetes and IGM than in those with normoglycaemia.
Methods
Study population
Data were obtained from The Maastricht Study [13], which is an ongoing observational study of adults aged between 40 and 75 years old living in the Southern Netherlands. The rationale and study methodology have been described previously [13]. In brief, recruitment was conducted through mass media campaigns, municipal registries and the regional diabetes patient registry. Participants were recruited and stratified according to diabetes status to investigate the aetiology and pathophysiology of diabetes, with an oversampling of those with known type 2 diabetes status. The current report includes cross-sectional data from 3451 participants recruited between November 2010 and September 2013; all examinations were performed on each participant within 3 months of consent. Exclusions were applied if they had missing data on the following covariates: sex; age; education category; smoking status; and adherence to the Dutch Healthy Diet index (n=334) [14]. Participants who had invalid activPAL activity monitor data (n=100) or did not wear the device (n=601) were excluded. Participants with type 1 diabetes or latent autoimmune or steroid-induced diabetes, or diabetes following pancreatectomy, were also excluded (n=28), leaving 2388 participants for the present analyses. Ethnicity data were collected; based on self-report, nearly all participants were of European descent [13].
The Maastricht Study was approved by the institutional medical ethical committee (NL31329.068.10) and the Minister of Health Welfare and Sports of the Netherlands (permit no. 131088-105234-PG). Written informed consent was obtained from all participants. The manuscript was written in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) reporting guidelines [15].
Sitting, standing, physical activity and sleeping
Daily behaviours (sitting, standing, stepping and sleeping) were measured using the activPAL3 inclinometer (version 6.4.1; PAL Technologies, Glasgow, UK). The device was attached directly to the skin on the front of the right thigh with transparent tape and was waterproofed with a nitrile sleeve. Participants were instructed to wear the monitor continuously for eight consecutive days without removal. To avoid inaccurately identifying non-wear time, participants were asked not to replace the device once removed. Data were uploaded using the activPAL software and processed using customised software written in MATLAB R2018b (MathWorks, Natick, MA, USA). The first measured wear day was excluded from analyses because it coincided with the clinical assessment and was therefore not a true representation of typical-day behaviours. Data from the final wear day, containing ≤14 h information, were also excluded for each participant. Data were included for analyses if there was at least one valid wear day that constituted ≥14 h of waking time. Stepping minutes were further categorised into LPA (<100 steps/min) and MVPA (≥100 steps/min) according to common cadence definitions [16]. The total amount of time spent sitting, standing, and in LPA and MVPA was divided by the number of valid wear days to derive average daily totals. Average daily sleeping time was estimated by subtracting the average waking time use from 24 h. An automated algorithm was used to determine sleep and waking time, as described elsewhere [17], therefore misclassification was possible (e.g. time lying in bed in some instances may have been incorrectly classified as sleeping time).
Cardiometabolic risk-marker outcomes
Outcome measures were waist circumference, fasting plasma glucose (FPG), 2 h post-load glucose (2hPLG), HbA1c, Matsuda index (ISI-M) and a clustered cardiometabolic risk score (CMR). The normality of residuals for FPG, 2hPLG and HbA1c improved following natural logarithm transformation. Waist circumference was measured manually with a tape measure midway between the lower rib margin and the peak of the iliac crest to the nearest 0.5 cm. Fasting samples were assessed using a standard enzymatic hexokinase reference method for plasma glucose. HbA1c was measured with ion-exchange HPLC. All included participants underwent a standardised 2 h OGTT, as described previously [13], where blood draws subsequent to fasting were collected at 15, 30, 45, 60, 90 and 120 min. The 2hPLG was informed by the OGTT and all six time points of glucose and insulin concentrations informed the calculation of the ISI-M, with a higher index indicating higher insulin sensitivity [18]. The ISI-M was calculated using fasting and mean glucose and insulin values, as follows (where FPI is fasting plasma insulin):
The clustered CMR was calculated using five cardiometabolic markers, including waist circumference, FPG, triacylglycerol, HDL-cholesterol and average BP, as per previously devised methods [19, 20]. Fasting blood samples were used for HDL-cholesterol and triacylglycerol analyses, which were assessed in laboratory with enzymatic and/or colorimetric methods by an automatic analyser (Beckman Synchron LX20; Beckman Coulter, Brea, CA, USA). Average systolic and diastolic BP was calculated from three office measurements of the right arm after a 10 min rest period using a non-invasive BP monitor (OMROW 705IT; OMRON, Kyoto, Japan). The average BP outcome was calculated by adding the systolic and diastolic measures together and dividing the value by two. Triacylglycerol, HDL-cholesterol and FPG measures were log-transformed. All variables were then standardised according to the mean [z=(value–mean)/SD]. The risk score was then calculated by summing all the scores (with HDL-cholesterol added in inverse) and dividing the sum by five. Higher CMR is relative to the sample mean and is indicative of higher cardiometabolic disease risk [19].
Covariates
Covariates were extracted from questionnaires administered during baseline assessment and included sex (self-reported), age, education (low, medium, high), smoking history (never, former, current smoker) and diet quality score. Education was ascribed as follows: low if the participant’s highest education was no education, primary education or lower vocational education; medium if it was general secondary education, general vocational education or higher secondary and pre-university education; and high if it was higher vocational education or university. Diet quality was measured with a validated Food Frequency Questionnaire [21] and scored as 0–140 using a Dutch Health Diet Index as a measure of adherence to the Dutch dietary guidelines, with higher scores indicating greater adherence [22]. Linear regression models featured adjustment for waist circumference, except where the independent variable was waist circumference or CMR. Diabetes status was assessed according to WHO 2006 criteria [23] using results from a 2 h OGTT and if the participant were using glucose-lowering medication. IGM was defined as impaired glucose tolerance (FPG <7.0 mmol/l and 2hPLG between ≥7.8 and <11.1 mmol/l) and/or impaired fasting glucose (FPG between 6.1 and 6.9 mmol/l and 2hPLG <7.8 mmol/l). Type 2 diabetes was defined as FPG ≥7.0 mmol/l and 2hPLG ≥11.1 mmol/l. Normal glucose metabolism was defined as below the IGM and type 2 diabetes cut points.
Statistical analyses
All analyses were conducted using R statistical analysis software version 4.0 (R Foundation for Statistical Computing, Vienna, Austria). In CoDA [7], the outcome is dependent on compositional isometric log ratios (ilrs) multiplied by their coefficients and then summed with covariates in a linear regression model. Within this model, ilrs map compositional data into real space. There were no behaviour counts equalling zero for sitting, standing, physical activity or sleeping. Behaviours were first transformed into a finite composition using the ‘acomp’ function in R package Compositions [24]. To investigate the associations between time use (expressed as ilrs) and the chosen outcome variables, linear regression was performed. A five-part composition can be expressed as a set of four ilrs (i.e. ilr1, ilr2, ilr3, ilr4), which were included in all analyses. To test the association of increasing sitting relative to remaining behaviours, the first ilr (corresponding to the β1 coefficient in the regression model) was constructed to reflect the effect of time sitting relative to the other three behaviours. Therefore, ilr1 is equal to:
The behaviours in the ilrs can be reordered in regression modelling as per the permutation principle [7] and give the same fit regardless of order. This allows each behaviour to be explored relative to the remaining behaviours. The composition (the set of ilrs in the regression model) was investigated for interaction by sex and diabetes status and the moderation on ilr1 coefficient was reported. The ilr models were used to perform isotemporal substitution (with R package: deltacomp [25]) whereby 30 min in one behaviour were substituted for 30 min in another. This method, explained in detail elsewhere [26], produces an estimated difference in the risk-marker value when time is reallocated to/from the geometric mean.
The optimal composition associated with each outcome was predicted by feeding a range of simulated compositions rounded to the nearest 10 min into the regression models. The simulated compositions were restricted to be within the empirical footprint, varying from the first to the 99th percentile of each behaviour. The full range of simulated compositions was created by producing every combination of these behaviours with one another that summed to 24 h. This resulted in 142,938 possible composition permutations for the overall sample. The compositions associated with the best 5% of z scores (i.e. lowest cardiometabolic risk) were chosen as the range of optimal compositions. The area at which the optimal compositions (by health outcome) overlapped in compositional space was taken as the unanimous overlapping optimal composition of 24 h time use. In many instances the overlapping compositions required that the optimal compositions be extended beyond the top 5% (i.e. to top 10%) to have a mutually overlapped area between outcomes in compositional space. Tetrahedrons were used to visualise the behavioural dynamics and overlapping optimal space, with sleep fixed at 8 h and the remaining behaviours preserved in four dimensions to represent waking time only. These were produced using the R package: rgl [27].
Results
Table 1 shows the characteristics of the 2388 participants, overall and stratified by diabetes status. Overall, the sample was evenly balanced between male (51.3%) and female sex (48.7%). In addition, a former smoking history was most common (52%), as was high education level (38.3%) in the sample. Sitting time occupied the greatest proportion of the day, and sleeping time was similar across all strata. The IGM and type 2 diabetes groups had higher sitting levels, and lower standing and physical activity levels compared with the normoglycaemic group.
Compositional isotemporal substitution modelling
Figure 1 depicts the estimated difference from the mean for risk markers when substituting 30 min of time spent sitting with other behaviours; all pairwise substitutions are depicted in electronic supplementary material (ESM) Tables 1–6. In the overall sample, higher LPA or MVPA levels with lower levels of sitting had beneficial associations with all risk markers, with variation as to whether the marker favoured higher compositional LPA (e.g. FPG) or MVPA (e.g. waist circumference). Higher standing time with lower levels of sitting time was significantly beneficially associated with waist circumference (z score [95% CI]: −0.04 [−0.06, −0.03]) and CMR (z score [95% CI]: −0.02 [−0.03, −0.01]) only. More time use spent sleeping was associated with benefit, for HbA1c when replacing standing time (z score [95% CI]: −0.02 [−0.04, −0.00]), otherwise it had adverse associations when replacing LPA, MVPA or non-significant associations. The estimated difference in risk markers was most pronounced in the type 2 diabetes group, especially when compared with the normoglycaemic group, for FPG and HbA1c and when LPA or MVPA replaced sitting. When MVPA replaced LPA this was associated with higher FPG in the type 2 diabetes group only. Basic compositional regression modelling (ESM Table 7) was additionally performed to investigate interactions by sex (ESM Table 8) and diabetes status (ESM Tables 9 and 10), and adjustment by waist circumference (ESM Table 11), on the relationships. Only CMR differed significantly by sex, suggesting that the beneficial association of standing at the expense of other behaviours was stronger in female participants. Adjustment by waist circumference resulted in the attenuation of all effect estimates, though they remained statistically significant for 2hPLG, HbA1c and ISI-M outcomes.
Estimating the difference incurred to glycaemic control and cardiometabolic risk markers with isotemporal substitution of 30 min from sitting to standing, physical activity and sleeping. Models are adjusted for age, sex, education, smoking status and dietary intake score. Overall sample analysis additionally adjusted for diabetes status. NGM, n=1341; IGM, n=363; type 2 diabetes, n=684. T2D, type 2 diabetes
Optimal compositions of time use
The optimal compositions of time use associated with optimal glycaemic control and cardiometabolic risk markers are depicted in Table 2. Across the markers, the optimal sitting time was consistently lower than the sample mean, the optimal standing time was mostly higher, and the optimal LPA and MVPA time was also higher for most markers. Optimal sleeping time had more variation across health markers, with 2hPLG and HbA1c having more beneficial associations with sleep duration. The optimal compositions of time use are visualised in multidimensional space in Fig. 2, with Fig. 2a–d featuring the same tetrahedron rotated at different perspectives. Figure 2a demonstrates the optimal compositions for FPG, favouring more time spent in LPA, and optimal compositions for waist circumference, favouring more time spent in MVPA. Figure 2b demonstrates the overlapping space between waist circumference and FPG forming the overlapped optimal zone. Figure 2c shows that the higher end of the optimal sitting range (i.e. 7 h and 10 min) must be accompanied by higher levels of physical activity, whereas the lower end of the sitting range comprises higher standing time and lower levels of physical activity. An interactive version of this plot can be viewed separately in ESM Fig. 1.
Optimal compositions of time use visualised in multidimensional space, adjusted for age, sex, education, smoking status, dietary intake score and diabetes status. Blue, waist circumference; pink, FPG; red, zone where all markers (including 2hPLG, HbA1c, ISI-M and CMR) share mutual overlap. Sleeping time was fixed to 8 h. Each corner of the tetrahedron depicts 100% of time use, and a composition data point directly in the middle of the tetrahedron would depict equal, 25% (4 h) of time use in sitting, standing, LPA and MVPA. Images in (a–d) depict the same four-axis quaternary tetrahedron rotated in different ways. Only FPG and waist circumference are displayed here for clarity, with the mutually shared overlap calculated using all health markers. PA, physical activity
There were small differences in optimal time-use composition by diabetes status (ESM Tables 12–14), which were potentially attributable to the innate differences between the optimal levels in each of the strata. For example, the top 5% of individuals with normoglycaemic metabolism (NGM) had substantially lower absolute waist circumference compared with those in the top 5% of people with type 2 diabetes. To summarise, the NGM group and the optimal compositions favoured much greater levels of standing and lower levels of sitting relative to the remaining groups. In the IGM group, standing time was less influential on the outcomes, as indicated by an expansive range. The type 2 diabetes group and optimal compositions on average favoured greater levels of sleeping time.
Discussion
These are novel findings from compositional analyses of free-living sitting, standing, physical activity and sleeping time in a large cross-sectional sample of middle-aged and older adults recruited to oversample people with type 2 diabetes. We show that optimal compositions of time use involved substantially less time spent sitting, a greater time spent standing and a substantially greater time being physically active than the times being achieved on average for each of these activities by the participants in our study. Optimal sleeping time aligned with the sample mean. The optimal time-use zone did not substantially differ by diabetes status, although compositions with greater physical activity and less sitting were associated more strongly in both the IGM and type 2 diabetes group. This highlights the importance of considering all the behaviours that compose 24 h time use when managing cardiometabolic disorders. The mean time-use composition that universally covered the optimal association of all cardiometabolic risk and glycaemic markers was as follows: sitting, 6 h (range: 5 h 40 min–7 h 10 min); standing, 5 h 10 min (range: 4 h 10 min–6 h 10 min); LPA, 2 h 10 min (range: 2 h–2 h 20 min); MVPA, 2 h 10 min (range: 1 h 40 min–2 h 20 min); sleeping, 8 h 20 min (range: 7 h 30 min–9 h).
Our investigation builds upon previous non-compositional analyses conducted with The Maastricht Study data by van der Berg et al [28, 29], extending these observational works by investigating optimal compositions and incorporating sleep time. The findings corroborate those of other observational analyses in populations with IGM and diabetes. Sedentary behaviour is adversely associated with cardiometabolic health [30, 31]. Less time spent being sedentary and more time spent participating in physical activity is associated with improved plasma glucose [32], insulin sensitivity [32,33,34], insulin levels, fat percentage, and triacylglycerol and cholesterol levels [34]. These studies largely suggest that MVPA is beneficial for cardiometabolic health, while acknowledging that reduction of sedentary time through the adoption of regular LPA is an important consideration irrespective of MVPA levels [35].
In alignment with the previous isotemporal analyses of data from The Maastricht Study [29], the findings suggest the viability of standing as a distinct alternative (along with physical activity and sleeping) to sitting, albeit with a greater amount required than for LPA and MVPA. These findings are in line with other compositional investigations that indicate that MVPA and stepping have the strongest associations with favourable cardiometabolic risk markers [7], including glucose and insulin [32]. Standing in CoDA has been less studied, with some evidence suggesting weak or mixed associations with health outcomes [36]. Optimal levels of standing time (4 h 10 min–6 h 10 min) have been demonstrated to be feasible in sedentary behaviour intervention settings [37]. Optimal sleep time (7 h 30 min–9 h) findings are aligned with current guidelines, which recommend a minimum of 7 h per day [38]. Interestingly, optimal sleeping levels differed slightly by health marker, especially for ISI-M, a marker of insulin sensitivity. Prolonged sleeping durations are associated with insulin resistance [39]; however, the optimal sleep duration must also be considered alongside beneficial associations of the ISI-M with standing and physical activity time. CoDA in this instance has balanced the benefit of sleeping with the benefit of longer time spent partaking in physical activity and longer standing time. Lastly, the inclusion of CMR provides clinical relevance to the findings. In the current study, the mean±SD difference between the average CMR estimate of the sample and the CMR estimate at the optimal composition was ~0.3±1.5. In a previous study, a similar CMR difference was found to be prospectively associated with significantly higher risk of cardiovascular events [40].
Experimental studies in people with type 2 diabetes that have acutely substituted sitting time with LPA have reported improvements in incremental AUC (iAUC) of glucose, triacylglycerol levels, insulin and insulin sensitivity [41]. Reducing sitting time through a combination of LPA and standing time has also been demonstrated to have positive effects on insulin sensitivity in postmenopausal women [42]. A review of field-based sedentary behaviour interventions determined that reductions in sitting corresponded with modest decreases in waist circumference, improvements to cardiometabolic risk (through systolic BP and HDL-cholesterol) and improved insulin sensitivity [43]. Across all reviewed trials, there were no changes in fasting glucose and HbA1c, possibly because there were limited studies featuring people with type 2 diabetes. These trials predominantly replaced sedentary time with standing, potentially leading to only modest associations observed with glycaemic outcomes [43]. Our findings were in line with those reported from experimental settings, where people with IGM benefited in terms of glucose and insulin from reductions in sedentary behaviour [44]. Further prospective evidence is required in free-living settings. Overall, current sedentary behaviour evidence suggests that, in addition to replacing sitting with standing time, sedentary behaviour interventions may need to incorporate more ambulatory behaviours to facilitate greater benefits in glucose metabolism, including those of higher intensity [45]. In line with this, it has been suggested that a ‘staircase’ approach could be considered when attempting to improve daily composition of waking behaviours, starting with replacing sedentary time with standing time, and then substituting in behaviours that are light intensity before more moderate-to-vigorous-intensity activities [46].
The findings from the present analysis could be used to further inform future iterations of time-use activity guidelines. Current 24 h activity guidelines [2] recommend specific quantities of time to be spent in MVPA (150 min/week), sedentary behaviour (<8 h/day) and sleep (7–9 h/day), but are less defined in their recommendations on how exactly sedentary behaviour should be replaced. The optimal zone upper limit of sedentary behaviour (7 h 10 min/day) supports these sedentary behaviour recommendations. Beneficial associations with cardiometabolic risk and glycaemic control were optimised as low as 4 h 10 min of standing per day and 2 h of LPA per day. These findings could help to inform future 24 h guidelines and provide evidence to inform recommendations pertaining to LPA and standing.
A key strength of this study is the use of CoDA in a large sample including those with type 2 diabetes, IGM and normoglycaemia for comparison purposes. Findings can be generalised to both sexes. All analyses were informed by data from a posture-sensing activity monitor that was able to accurately collect continuous measurements over multiple days. Notably, the current study is one of the few [29, 32, 36] to consider standing in a composition of time use. Few studies [32, 47] have ascertained the relationship between composition of daily behaviours with an array of risk markers indicative of subsequent disease risk, such as 2hPLG and ISI-M, which are resource-intensive to collect. The same is true for the sophisticated phenotyping that allows for appropriate adjustment of relevant confounders necessary in observational research. Observational studies have the potential to address novel hypotheses in the absence of more sophisticated prospective studies. However, limitations need to be considered. First, the sample includes individuals mainly of European descent and participants with well-controlled diabetes, therefore limiting generalisability of the findings to other populations. Second, the analyses are cross-sectional in nature, therefore precluding causal inference about the potential for composition changes to benefit risk markers. Similarly, the findings may be driven by reverse causation whereby poor glycaemic control, cardiometabolic ill-health or other comorbidities may be causing an increase in sedentary behaviours and a decrease in physical activity. Third, while the current analyses consider intensity of physical activity, this was based on stepping cadence cut points, warranting further investigation with more sophisticated measures of relative intensity such as heart rate. Finally, bout length (e.g. sedentary behaviours accumulated in prolonged bouts or activity accumulated in short bouts) was not considered, which might have distinct implications for cardiometabolic health that are potentially independent of total sitting time [48,49,50].
We provide novel observational evidence on compositional 24 h time use and an optimal balance of sitting time with standing, LPA, MVPA and sleeping. The optimal composition associated with all cardiometabolic risk and glycaemic control markers was as follows: sitting, 6 h (range: 5 h 40 min–7 h 10 min); standing, 5 h 10 min (range: 4 h 10 min–6 h 10 min); LPA, 2 h 10 min (range: 2 h–2 h 20 min); MVPA, 2 h 10 min (range: 1 h 40 min–2 h 20 min); and sleeping, 8 h 20 min (range: 7 h 30 min–9 h). These findings can help to inform future 24 h guidelines on sitting, standing, physical activity and sleep to improve cardiometabolic health and glycaemic control. For those with IGM or type 2 diabetes, our findings support recommendations to limit daily sedentary behaviour. However, longer-term and prospective study evidence, and intervention trials that change sedentary behaviour in daily time-use compositions, are needed to corroborate our findings.
Abbreviations
- CMR:
-
Clustered cardiometabolic risk score
- CoDA:
-
Compositional data analysis
- FPG:
-
Fasting plasma glucose
- 2hPLG:
-
2 h post-load glucose
- IGM:
-
Impaired glucose metabolism
- ilr:
-
Isometric log ratio
- ISI-M:
-
Matsuda index
- LPA:
-
Light-intensity physical activity
- MVPA:
-
Moderate-to-vigorous physical activity
- NGM:
-
Normoglycaemic metabolism
References
Tremblay MS, Chaput J-P, Adamo KB et al (2017) Canadian 24-hour movement guidelines for the early years (0–4 years): an integration of physical activity, sedentary behaviour, and sleep. BMC Public Health 17(5):874. https://doi.org/10.1186/s12889-017-4859-6
Ross R, Chaput JP, Giangregorio LM et al (2020) Canadian 24-hour movement guidelines for adults aged 18–64 years and adults aged 65 years or older: an integration of physical activity, sedentary behaviour, and sleep. Appl Physiol Nutr Metab 45(10):S57–S102. https://doi.org/10.1139/apnm-2020-0467
Grgic J, Dumuid D, Bengoechea EG et al (2018) Health outcomes associated with reallocations of time between sleep, sedentary behaviour, and physical activity: a systematic scoping review of isotemporal substitution studies. Int J Behav Nutr Phys Act 15(1):69. https://doi.org/10.1186/s12966-018-0691-3
Janssen I, Clarke AE, Carson V et al (2020) A systematic review of compositional data analysis studies examining associations between sleep, sedentary behaviour, and physical activity with health outcomes in adults. Appl Physiol Nutr Metab 45(10 (Suppl. 2)):S248–S257. https://doi.org/10.1139/apnm-2020-0160
Buccianti A, Nisi B, Martín-Fernández JA, Palarea-Albaladejo J (2014) Methods to investigate the geochemistry of groundwaters with values for nitrogen compounds below the detection limit. J Geochemical Explor 141:78–88. https://doi.org/10.1016/j.gexplo.2014.01.014
Ros-Freixedes R, Estany J (2014) On the compositional analysis of fatty acids in pork. J Agric Biol Environ Stat 19(1):136–155. https://doi.org/10.1007/s13253-013-0162-x
Chastin SFM, Palarea-Albaladejo J, Dontje ML, Skelton DA (2015) Combined effects of time spent in physical activity, sedentary behaviors and sleep on obesity and cardio-metabolic health markers: a novel compositional data analysis approach. PLoS One 10(10):e0139984. https://doi.org/10.1371/journal.pone.0139984
Brakenridge CJ, Healy GN, Sethi P et al (2021) Contrasting compositions of sitting, standing, stepping, and sleeping time: associations with glycaemic outcome by diabetes risk. Int J Behav Nutr Phys Act 18(1):155. https://doi.org/10.1186/s12966-021-01209-5
Ekelund U, Tarp J, Steene-Johannessen J et al (2019) Dose-response associations between accelerometry measured physical activity and sedentary time and all cause mortality: systematic review and harmonised meta-analysis. BMJ 366:l4570. https://doi.org/10.1136/bmj.l4570
Stamatakis E, Gale J, Bauman A, Ekelund U, Hamer M, Ding D (2019) Sitting time, physical activity, and risk of mortality in adults. J Am Coll Cardiol 73(16):2062–2072. https://doi.org/10.1016/j.jacc.2019.02.031
Hall KS, Hyde ET, Bassett DR et al (2020) Systematic review of the prospective association of daily step counts with risk of mortality, cardiovascular disease, and dysglycemia. Int J Behav Nutr Phys Act 17(1):78. https://doi.org/10.1186/s12966-020-00978-9
Shan Z, Ma H, Xie M et al (2015) Sleep duration and risk of type 2 diabetes: a meta-analysis of prospective studies. Diabetes Care 38(3):529–537. https://doi.org/10.2337/dc14-2073
Schram MT, Sep SJS, van der Kallen CJ et al (2014) The Maastricht Study: an extensive phenotyping study on determinants of type 2 diabetes, its complications and its comorbidities. Eur J Epidemiol 29(6):439–451. https://doi.org/10.1007/s10654-014-9889-0
Slingerland AS, van Lenthe FJ, Jukema JW et al (2007) Aging, retirement, and changes in physical activity: prospective cohort findings from the GLOBE study. Am J Epidemiol 165:1356–1363. https://doi.org/10.1093/aje/kwm053
Cuschieri S (2019) The STROBE guidelines. Saudi J Anaesth 13(Suppl 1):S31–S34. https://doi.org/10.4103/sja.SJA_543_18
Tudor-Locke C, Ducharme SW, Aguiar EJ et al (2020) Walking cadence (steps/min) and intensity in 41 to 60-year-old adults: the CADENCE-adults study. Int J Behav Nutr Phys Act 17(1):137. https://doi.org/10.1186/s12966-020-01045-z
van der Berg JD, Willems PJB, van der Velde JHPM et al (2016) Identifying waking time in 24-h accelerometry data in adults using an automated algorithm. J Sports Sci 34(19):1867–1873. https://doi.org/10.1080/02640414.2016.1140908
Matsuda M, DeFronzo RA (1999) Insulin sensitivity indices obtained from oral glucose tolerance testing: comparison with the euglycemic insulin clamp. Diabetes Care 22(9):1462–1470. https://doi.org/10.2337/diacare.22.9.1462
Wijndaele K, Duvigneaud N, Matton L et al (2009) Sedentary behaviour, physical activity and a continuous metabolic syndrome risk score in adults. Eur J Clin Nutr 63(3):421–429. https://doi.org/10.1038/sj.ejcn.1602944
Dempsey PC, Hadgraft NT, Winkler EAH et al (2018) Associations of context-specific sitting time with markers of cardiometabolic risk in Australian adults. Int J Behav Nutr Phys Act 15(1):114. https://doi.org/10.1186/s12966-018-0748-3
van Dongen MC, Wijckmans-Duysens NEG, den Biggelaar LJ et al (2019) The Maastricht FFQ: development and validation of a comprehensive food frequency questionnaire for the Maastricht study. Nutrition 62:39–46. https://doi.org/10.1016/j.nut.2018.10.015
Looman M, Feskens EJ, de Rijk M et al (2017) Development and evaluation of the Dutch Healthy Diet index 2015. Public Health Nutr 20(13):2289–2299. https://doi.org/10.1017/S136898001700091X
World Health Organization (2006) Definition and diagnosis of diabetes mellitus and intermediate hyperglycaemia: report of a WHO/IDF consultation. Available from: www.who.int/publications/i/item/definition-and-diagnosis-of-diabetes-mellitus-and-intermediate-hyperglycaemia. Accessed: 16 September 2021
van den Boogaart KG, Tolosana-Delgado R, Bren M (2021) Compositions: compositional data analysis. R package version 2.0-2. Available from: https://CRAN.R-project.org/package=compositions. Accessed: 23 December 2021
Stanford TE (2021) Deltacomp: functions to analyse compositional data and produce confidence intervals for relative increases and decreases in the compositional components. Available from: https://rdrr.io/github/tystan/deltacomp/. Accessed: 16 September 2021
Dumuid D, Pedišić Ž, Stanford TE et al (2019) The compositional isotemporal substitution model: a method for estimating changes in a health outcome for reallocation of time between sleep, physical activity and sedentary behaviour. Stat Methods Med Res 28(3):846–857. https://doi.org/10.1177/0962280217737805
Murdoch D, Adler D (2021) rgl: 3D visualization using OpenGL. R package version 0.106.8. Available from: https://CRAN.R-project.org/package=rgl. Accessed: 24 July 2023
van der Berg JD, Stehouwer CD, Bosma H et al (2016) Associations of total amount and patterns of sedentary behaviour with type 2 diabetes and the metabolic syndrome: The Maastricht Study. Diabetologia 59(4):709–718. https://doi.org/10.1007/s00125-015-3861-8
van der Berg JD, Van Der Velde JHPM, De Waard EAC et al (2017) Replacement effects of sedentary time on metabolic outcomes: The Maastricht Study. Med Sci Sports Exerc 49(7):1351–1358. https://doi.org/10.1249/MSS.0000000000001248
Rossen J, Von Rosen P, Johansson U-B, Brismar K, Hagströmer M (2020) Associations of physical activity and sedentary behavior with cardiometabolic biomarkers in prediabetes and type 2 diabetes: a compositional data analysis. Phys Sportsmed 48(2):222–228. https://doi.org/10.1080/00913847.2019.1684811
Ryan DJ, Wullems JA, Stebbings GK, Morse CI, Stewart CE, Onambele-Pearson GL (2019) The difference in sleep, sedentary behaviour, and physical activity between older adults with ‘healthy’ and ‘unhealthy’ cardiometabolic profiles: a cross-sectional compositional data analysis approach. Eur Rev Aging Phys Act 16(1):25. https://doi.org/10.1186/s11556-019-0231-4
Biddle GJH, Edwardson CL, Henson J et al (2018) Associations of physical behaviours and behavioural reallocations with markers of metabolic health: a compositional data analysis. Int J Environ Res Public Health 15(10):2280. https://doi.org/10.3390/ijerph15102280
Edwardson CL, Henson J, Bodicoat DH et al (2017) Associations of reallocating sitting time into standing or stepping with glucose, insulin and insulin sensitivity: a cross-sectional analysis of adults at risk of type 2 diabetes. BMJ Open 7(1):e014267. https://doi.org/10.1136/bmjopen-2016-014267
Swindell N, Rees P, Fogelholm M et al (2020) Compositional analysis of the associations between 24-h movement behaviours and cardio-metabolic risk factors in overweight and obese adults with pre-diabetes from the PREVIEW study: cross-sectional baseline analysis. Int J Behav Nutr Phys Act 17(1):29. https://doi.org/10.1186/s12966-020-00936-5
Madden KM, Feldman B, Chase J (2020) Sedentary time and metabolic risk in extremely active older adults. Diabetes Care 44(1):194–200. https://doi.org/10.2337/dc20-0849
Powell C, Browne LD, Carson BP et al (2020) Use of compositional data analysis to show estimated changes in cardiometabolic health by reallocating time to light-intensity physical activity in older adults. Sports Med 50(1):205–217. https://doi.org/10.1007/s40279-019-01153-2
Healy GN, Eakin EG, Owen N et al (2016) A cluster randomized controlled trial to reduce office workers’ sitting time: effect on activity outcomes. Med Sci Sports Exerc 48(9):1787–1797. https://doi.org/10.1249/MSS.0000000000000972
Watson NF, Badr MS, Belenky G et al (2023) Recommended amount of sleep for a healthy adult: a joint consensus statement of the American Academy of Sleep Medicine and Sleep Research Society. J Clin Sleep Med 11(06):591–592. https://doi.org/10.5664/jcsm.4758
Brady EM, Bodicoat DH, Hall AP et al (2018) Sleep duration, obesity and insulin resistance in a multi-ethnic UK population at high risk of diabetes. Diabetes Res Clin Pract 139:195–202. https://doi.org/10.1016/j.diabres.2018.03.010
Agarwal S, Jacobs DR Jr, Vaidya D et al (2012) Metabolic syndrome derived from principal component analysis and incident cardiovascular events: the Multi Ethnic Study of Atherosclerosis (MESA) and Health, Aging, and Body Composition (Health ABC). Cardiol Res Pract 2012:919425. https://doi.org/10.1155/2012/919425
Duvivier BM, Schaper NC, Hesselink MK et al (2017) Breaking sitting with light activities vs structured exercise: a randomised crossover study demonstrating benefits for glycaemic control and insulin sensitivity in type 2 diabetes. Diabetologia 60(3):490–498. https://doi.org/10.1007/s00125-016-4161-7
Remie CME, Janssens GE, Bilet L et al (2021) Sitting less elicits metabolic responses similar to exercise and enhances insulin sensitivity in postmenopausal women. Diabetologia 64(12):2817–2828. https://doi.org/10.1007/s00125-021-05558-5
Hadgraft NT, Winkler E, Climie RE et al (2021) Effects of sedentary behaviour interventions on biomarkers of cardiometabolic risk in adults: systematic review with meta-analyses. Br J Sports Med 55:144–154. https://doi.org/10.1136/bjsports-2019-101154
Dempsey PC, Larsen RN, Winkler EAH, Owen N, Kingwell BA, Dunstan DW (2018) Prolonged uninterrupted sitting elevates postprandial hyperglycaemia proportional to degree of insulin resistance. Diabetes, Obes Metab 20(6):1526–1530. https://doi.org/10.1111/dom.13254
Yates T, Edwardson CL, Henson J, Zaccardi F, Khunti K, Davies MJ (2020) Prospectively reallocating sedentary time: associations with cardiometabolic health. Med Sci Sports Exerc 52(4):844–850. https://doi.org/10.1249/MSS.0000000000002204
Dogra S, Copeland JL, Altenburg TM, Heyland DK, Owen N, Dunstan DW (2021) Start with reducing sedentary behavior: a stepwise approach to physical activity counseling in clinical practice. Patient Educ Couns 105(6):1353–1361. https://doi.org/10.1016/j.pec.2021.09.019
Farrahi V, Kangas M, Walmsley R et al (2021) Compositional associations of sleep and activities within the 24-h cycle with cardiometabolic health markers in adults. Med Sci Sports Exerc 53(2):324–332. https://doi.org/10.1249/MSS.0000000000002481
Dempsey PC, Strain T, Winkler EAH et al (2022) Association of accelerometer-measured sedentary accumulation patterns with incident cardiovascular disease, cancer, and all-cause mortality. J Am Heart Assoc 11(9):e023845. https://doi.org/10.1161/JAHA.121.023845
Bellettiere J, Winkler EAH, Chastin SFM, Kerr J, Owen N, Dunstan DW (2017) Associations of sitting accumulation patterns with cardio-metabolic risk biomarkers in Australian adults. PLoS One 12(6):e0180119. https://doi.org/10.1371/journal.pone.0180119
Glazer NL, Lyass A, Esliger DW et al (2013) Sustained and shorter bouts of physical activity are related to cardiovascular health. Med Sci Sports Exerc 45(1):109–115. https://doi.org/10.1249/MSS.0b013e31826beae5
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Acknowledgements
We acknowledge the people who participated in this study, without whom this work would not be possible. Preliminary analyses of these data appear in a PhD thesis by CJB.
Data availability
Datasets generated and analysed are not publicly available but are available from the corresponding author upon reasonable request.
Funding
The Maastricht Study was supported by the European Regional Development Fund via OP-Zuid, the Province of Limburg, the Dutch Ministry of Economic Affairs (grant 31O.041), Stichting De Weijerhorst (Maastricht, the Netherlands), the Pearl String Initiative Diabetes (Amsterdam, the Netherlands), the Cardiovascular Center (CVC, Maastricht, the Netherlands), CARIM School for Cardiovascular Diseases (Maastricht, the Netherlands), CAPHRI Care and Public Health Research Institute (Maastricht, the Netherlands), NUTRIM School for Nutrition and Translational Research in Metabolism (Maastricht, the Netherlands), Stichting Annadal (Maastricht, the Netherlands), Health Foundation Limburg (Maastricht, the Netherlands), and by unrestricted grants from Janssen-Cilag BV (Tilburg, the Netherlands), Novo Nordisk Farma BV (Alphen aan den Rijn, the Netherlands), and Sanofi-Aventis Netherlands BV (Gouda, the Netherlands). DD was funded by an Australian Research Council (ARC) Discovery Early Career Award (DE230101174). DWD and NO are supported by the NHMRC Fellowships scheme and the Victorian Government’s OIS Program. GNH was supported by an MRFF-NHMRC Emerging Leadership Investigator Grant (no. 1193815). The funders of this study and The Maastricht Study were not involved in the design of the study; the collection, analysis, and interpretation of data; writing the report; and did not impose any restrictions regarding the publication of the report.
Authors’ relationships and activities
BEdG is a member of the editorial board of Diabetologia. The remaining authors declare that there are no relationships or activities that might bias, or be perceived to bias, their work.
Contribution statement
CJB contributed to the conception and design, analysed, and interpreted data, drafted and revised the manuscript critically for important intellectual content. DWD, GNH, NO, AC and FQSD were involved in the conception of the study design, drafted, and revised the manuscript critically for important intellectual content. DD made substantial contributions to the analysis and interpretation of the data, and reviewed the text critically for important intellectual content. AK, BEdG and NCS provided substantial contributions to the conception of the design, acquisition of the data, and provided critical review and drafting of the manuscript. SJPME, HHCMS and HB provided substantial contributions to the acquisition of the data and provided critical review for important intellectual content. All authors provided final approval of the version to be published. DWD is the guarantor of this work.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Brakenridge, C.J., Koster, A., de Galan, B.E. et al. Associations of 24 h time-use compositions of sitting, standing, physical activity and sleeping with optimal cardiometabolic risk and glycaemic control: The Maastricht Study. Diabetologia 67, 1356–1367 (2024). https://doi.org/10.1007/s00125-024-06145-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00125-024-06145-0