Abstract
Aims/hypothesis
Rosiglitazone, an insulin sensitiser, not only improves insulin sensitivity but also enhances insulin secretory capacity by ameliorating gluco- and lipotoxicity in beta cells. Rosiglitazone can stimulate insulin secretion at basal and high glucose levels via a phosphatidylinositol 3-kinase (PI3K)-dependent pathway. We hypothesised that regulation of phosphorylation of the ATP-sensitive potassium (KATP) channel might serve as a key step in the regulation of insulin secretion.
Methods
Insulin secretory responses were studied in an isolated pancreas perfusion system, cultured rat islets and MIN6 and RINm5F beta cells. Signal transduction pathways downstream of PI3K were explored to link rosiglitazone to KATP channel conductance with patch clamp techniques and insulin secretion measured by ELISA.
Results
Rosiglitazone stimulated AMP-activated protein kinase (AMPK) activity and induced inhibition of the KATP channel conductance in islet beta cells; both effects were blocked by the PI3K inhibitor LY294002. Following stimulation of AMPK by 5-aminoimidazole-4-carboxamide ribonucleoside (AICAR), a pharmacological activator, both AICAR-stimulated insulin secretion and inhibition of KATP channel conductance were unaffected by LY294002, indicating that AMPK activation occurs at a site downstream of PI3K activity. The serine residue at amino acid position 385 of Kir6.2 was found to be the substrate phosphorylation site of AMPK when activated by rosiglitazone or AICAR.
Conclusions/interpretation
Our data indicate that PI3K-dependent activation of AMPK is required for rosiglitazone-stimulated insulin secretion in pancreatic beta cells. Phosphorylation of the Ser385 residue of the Kir6.2 subunit of the KATP channel by AMPK may play a role in insulin secretion.
Similar content being viewed by others
Type 2 diabetes is characterised by peripheral insulin resistance, impaired insulin secretion and enhanced hepatic glucose production [1]. Pancreatic beta cell function has been shown to deteriorate as the duration of diabetes increases despite the use of existing therapies, as demonstrated in the UK Prospective Diabetes Study [2]. The thiazolidinediones, on the other hand, are a new class of glucose-lowering agent targeted at peroxisome proliferator-activated receptor γ (PPARγ) [3] and have a beneficial effect on beta cell functions [4, 5]. This beneficial effect on beta cell function is partly mediated by alleviating gluco- and lipotoxicity [5, 6] and partly by a protective effect against apoptosis [5–11] of beta cells. In one human study, it was reported that glucose-entrained high-frequency insulin pulsatility in response to an oscillatory glucose infusion improved after treatment with rosiglitazone for 3 months, despite the absence of a change in beta cell secretory capacity [12].
Few studies have addressed the acute action of thiazolidinediones on beta cell function. In one early study [4], it was demonstrated that short-term treatment (10 or 60 min) with troglitazone induced insulin secretion. This insulin secretory response was accompanied by an increase in cytoplasmic free Ca2+ concentrations in both isolated islets and in HIT insulin-secreting beta cells. Furthermore, thiazolidinediones can inhibit ATP-sensitive potassium (KATP) channels in CRI-G1 insulin-secreting cells [13,14], although the mechanism underlying this action has not been well studied. We have previously reported that rosiglitazone, in addition to its stimulatory effect on insulin secretion at a basal glucose concentration, also enhanced the first and second phases of insulin secretion at a high glucose concentration [15]. This stimulating effect of rosiglitazone is dependent on phosphatidylinositol 3-kinase (PI3K) activity [15]. However, the mechanism by which PI3K activation causes an insulin secretory response in beta cells remains unknown.
Recently, 5-aminoimidazole-4-carboxamide ribonucleoside (AICAR), an allosteric activator of AMP-dependent protein kinase (AMPK), has been shown to stimulate insulin release in both isolated islets and perfused pancreatic beta cells [16, 17]. The effects of AICAR on insulin secretion are mediated by both KATP channel-dependent and -independent pathways [18]. KATP channels are hetero-octamers which are composed of four sulfonylurea receptor (SUR1) subunits and four inwardly rectifying K+ channel (Kir6.2) subunits [19], which are regulated by allosteric activation with various nucleotides [20]. KATP channel activity can also be regulated by phosphorylation events of the constituent proteins, such as phosphorylation of Ser372 of the Kir6.2 subunit and Ser1571 of the SUR1 subunit by protein kinase A [21] and phosphorylation of the Thr180 residue of the Kir6.2 subunit by protein kinase C [22]. In this study, we investigated the function of AMPK in the rosiglitazone-potentiated insulin secretory response, and the potential site of phosphorylation of the Kir6.2 subunit.
Methods
Chemicals
All chemicals were purchased from Sigma Chemical (St Louis, MO, USA), except for rosiglitazone (BRL49653), LY294002 and compound C, which were provided by GlaxoSmithKline Pharmaceuticals (Brentford, UK), Eli Lilly (Indianapolis, IN, USA) and Merck Research Laboratories (Rahway, NJ, USA), respectively. Bis-(1,3-dibutylbarbituric acid)pentamethine oxonol [DiBAC4(5)] was purchased from Invitrogen (Carlsbad, CA, USA).
Experimental animals
Male Sprague–Dawley rats weighing 250–350 g, obtained from the Animal Center of the National Science Council of the Republic of China, were used in these experiments. Animal studies were approved by the institutional review board. The principles of laboratory animal care outlined in NIH publication No. 85–23 (revised 1985) were followed in all experiments.
Pancreatic perfusion
In situ rat pancreatic perfusion was performed using an open system at 37°C, as described previously [15, 23]. After a 10 min baseline perfusion, insulin secretory responses to AICAR (50 μmol/l) in the presence of either 5.5 or 10 mmol/l glucose were assessed as indicated in the experimental procedures. Effluent fluid was collected from the portal vein cannula at intervals of 1 min for either 50 or 140 min. The collected effluent was kept at 4°C and subsequently assayed for insulin, as described previously [15, 24].
Rat islet isolation and culture
Rat pancreas islet cells were isolated from male Sprague–Dawley rats weighing 250–300 g by collagenase digestion, as described previously [25]. Islet cells were cultured at 37°C, 5% CO2, pH 7.4, in RPMI 1640 containing 10% fetal bovine serum, 10 mmol/l glucose, 180 μmol/l penicillin and 68.6 μmol/l streptomycin and used 2–3 days after isolation.
Cell line and cell culture
MIN6 cells, kindly provided by S. Seino (Kobe University, Japan), were used between passages 15 and 25 and grown in DMEM containing 10% (vol/vol) fetal calf serum at 37°C with 5% CO2. RINm5F cells were used between passage 15 and 25 and grown in RPMI containing 10% (vol./vol.) fetal calf serum at 37°C with 5% CO2.
MIN6 or RINm5F cells were seeded and incubated for about 24 h before the experiment. Next, the previous medium was removed and replaced with DMEM (MIN6) or RPMI (RINm5F) containing 0.2% BSA at 37°C for 2 h. This medium was then removed and replaced with KRB buffer, 5 mmol/l sodium bicarbonate and 0.2% (wt/vol.) BSA containing either 3.3 or 16.7 mmol/l glucose with or without drug treatment.
Cell extraction and assay of AMPK activity
Rat islets, MIN6 cells or RINm5F cells were lysed and immunoprecipitated with anti-AMPK α subunit antibody (Cell Signaling Technology, Beverly, MA, USA) as described previously [26]. The AMPK activity in the immunoprecipitate was assessed as a function of phosphorylation of a SAMS-containing peptide (HMRSAMSGLHLVKRR) [26] or an FSIS-containing peptide (KFSISPDSLS) (amino acids 381–390 of the Kir6.2 subunit of the KATP channel).
Antibodies, immunoprecipitation and western blot analyses
Polyclonal antibodies against Kir6.2 were generated by immunising rabbits with KLH-conjugated APSEGTAEPC. The antibody was purified by peptide-specific affinity column chromatography. In addition, mouse monoclonal anti-phosphoserine385 of the Kir6.2 antibody (clone 6B3) was produced by immunising BALB/c mice with KLH-conjugated PKFSI(S*)PDSLC. The antibodies were produced in ascites and purified with a protein G column. Anti-AMPK α subunit and anti-AMPK phospho-threonine172 antibodies were purchased from Cell Signaling Technology.
Equal amounts (1 mg) of cell lysates from rat islets, MIN6 cells or RINm5F cells were immunoprecipitated with anti-Kir6.2 antibody and then analysed by western blotting with either rabbit anti-Kir6.2 antibody or 6B3 monoclonal anti-phosphoserine385 Kir6.2 antibody [27]. For direct immunoblotting, equal amounts (50 μg) of MIN6 or RINm5F cell lysate were analysed by western blotting with an anti-AMPK α subunit antibody and an anti-AMPK phosphothreonine172 antibody (Cell Signaling Technology).
Measurement of KATP current
The KATP current was recorded by a whole-cell voltage clamp technique at room temperature (∼25°C) in single islets 10–15 μm in diameter. The whole-cell patch clamp technique was used to record ionic currents with a patch clamp amplifier (WPC-100; ESF Electronic, Goettingen, Germany). Heat-polished glass electrodes, with tip resistances between 2 and 5 MΩ when filled with pipette internal solution, were used. Signals were acquired with a Digidata 1322 analogue-to-digital interface controlled by pClamp software (Axon Instruments, Foster City, CA, USA). Recordings were low-pass filtered at 10 kHz and stored for further analysis. The pipette internal solution contained 130 mmol/l potassium aspartate, 10 mmol/l KCl, 10 mmol/l NaCl, 2 mmol/l MgCl2, 10 mmol/l HEPES, 0.3 mmol/l ATP, 0.3 mmol/l ADP, 11 mmol/l EGTA and 1 mmol/l CaCl2, pH 7.2. The bath KRP solution contained 109 mmol/l NaCl, 4.6 mmol/l KCl, 0.4 mmol/l KH2PO4, 1.15 mmol/l NaH2PO4, 1.1 mmol/l MgCl2, 3.2 mmol/l CaCl2, 10 mmol/l HEPES and 3.3 mmol/l glucose, pH 7.4. Currents at −40 mV were recorded continuously in response to the different treatments, and ramp currents were elicited from −80 to 0 mV with slope 80 V/s to establish a current–voltage relationship at the time points indicated in the results. The current of the KATP channel (IKATP) was gradually activated and developed to a steady amplitude within 5 min after intracellular dialysis under the condition of a low ATP/ADP ratio; the developed current was stable and could be kept at the same level (less than 5% run-down) for 20 min in the control condition.
Silencing of α subunit of AMPK in RINm5F cells
RINm5F cells were cultured in RPMI containing 10% fetal calf serum without antibiotics until 50% confluence. Then, scrambled short interfering RNA (siRNA; Stealth RNAi negative control with low GC content; Invitrogen) or siRNA of the α1 subunit of AMPK (5′-UUAAGGCUUCAUCAUCAAUCAUGGU-3′, 5′-ACCAUGAUUGAUGAUGA AGCCUUAA-3′; Invitrogen) was transfected into RINm5F cells using Lipofectamine 2000 (Invitrogen) according to the manufacturer’s instructions.
Exchanging S385 for Ala in human Kir6.2 subunit
A pCMV6c-hKir6.2 (human Kir6.2), kindly provided by S. Seino (Kobe University, Japan), was used to construct wild-type pCMS-EGFP-hKir6.2. The Ser385Ala mutant type of Kir6.2 was then generated using the QuickChange II site-directed mutagenesis kit (Stratagene, La Jolla, CA, USA) to replace serine at the 385th amino acid with alanine. The forward primer was 5′-TTCAGCATCGCTCCAGATTCCCTGTCCTGA-3′ and the reverse primer was 5′-TCAGGACAGGGAATCTGGAGCGATGCTGAA-3′. The wild-type or Ser385Ala mutant plasmid was transfected into RINm5F cells using Lipofectamine 2000 (Invitrogen) according to the manufacturer’s instructions.
Flow cytometric measurement of the change in membrane potential
Flow cytometric measurement of the change in membrane potential in response to rosiglitazone or AICAR in RINm5F cells transfected with wild-type or Ser385Ala mutant Kir6.2 subunit was carried out using a FACSCalibur cytometer (Becton Dickinson, Mountain View, CA, USA). RINm5F cells were resuspended in 1 ml KRB solution containing 3.3 mmol/l glucose and 400 nmol/l DiSBAC4(5) with or without drug treatment. DiSBAC4(5) is a slow-response, potential-sensitive probe that can enter depolarised cells, where it binds to intracellular proteins or membranes and exhibits enhanced fluorescence and a red spectral shift (excitation maximum 590 nm, emission maximum 616 nm). Emission was detected through a 515–545 nm broadband interference filter for EGFP (FL1) and 564–606 nm for DiBAC4(5) (FL2). Cytofluorimetric data were analysed using Cellquest software (Becton Dickinson). The geometric means of the FL2 fluorescence histograms were calculated from the EGFP-positive cells, which were gated by FL1 at the region in the presence of increased FL1 intensity, as indicated in Fig. 6, on the basis of 2 × 104 cells. For cells in the control group (without transfection), the FL2 geometric mean was calculated directly from 2 × 104 cells.
Data expression and statistical analysis
Effluent insulin concentration was recorded as the percentage of the baseline insulin level (calculated as the mean of 12 baseline values), and the data are presented as mean ± SE. ANOVA was used to determine the statistical significance of treatment and time data. Tukey’s honestly significant difference post hoc test was used to determine the statistical significance of differences between treatments for which the ANOVA analysis showed significance. Statistical significance was accepted at p < 0.05. The statistical significances of differences in other data between or among different experimental conditions were analysed with Student’s t test, the paired t test, ANOVA or non-parametric analysis, using the Statistical Package for the Social Sciences version 8 (SPSS, Chicago, IL, USA).
Results
Rosiglitazone-stimulated AMPK phosphorylation depends on PI3K activity in MIN6 beta cells
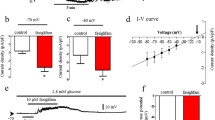
Rosiglitazone significantly increased phosphorylation of the Thr172 of the α subunit of AMPK in MIN6 beta cells in a concentration- and time-dependent manner (Electronic supplementary material [ESM] Fig. 1a, b). This effect was independent of glucose concentration. LY294002, a PI3K inhibitor, markedly inhibited both rosiglitazone-induced AMPK phosphorylation (Fig. 1a) and its kinase activity (Fig. 1b). These effects were observed at both low and high glucose concentrations.
Effect of PI3K activity on rosiglitazone-stimulated AMPK activation. a Western blot with anti-AMPK and anti-Thr172-phospho-AMPK in the absence or presence of rosiglitazone (RSG; 4.5 µmol/l) and/or LY294002 (3.9 µmol/l) in low (3.3 mmol/l) and high (16.7 mmol/l) glucose (Glu) in MIN6 cells. pAMPK/AMPK density relative to that in glucose 3.3 mmol/l is shown below (n = 3). White columns, control; black columns, rosiglitazone 4.5 µmol/l; light grey columns, rosiglitazone 4.5 µmol/l and LY294002 3.9 µmol/l. *p < 0.05, non-parametric analysis. b AMPK enzyme activity in the absence or presence of rosiglitazone (4.5 µmol/l) and/or LY294002 (3.9 µmol/l) in low (3.3 mmol/l) and high (16.7 mmol/l) glucose in MIN6 cells. Data are shown as relative AMPK activity (mean % and SE [bar]) compared with that in glucose at 3.3 mmol/l (100% as referent) (n = 4). The experimental condition for each column is the same as that for the corresponding column in a. *p < 0.05. c Insulin secretory response in the absence or presence of rosiglitazone (4.5 µmol/l) and/or LY294002 (3.9 µmol/l) and wortmannin (50 nmol/l) in low (3.3 mmol/l) and high (16.7 mmol/l) glucose in MIN6 cells. Data are relative insulin responses (mean % and SE [bar]) compared with that in glucose 3.3 mmol/l (100% as referent) (n = 4). White columns, control; black columns, rosiglitazone 4.5 µmol/l; light grey columns, rosiglitazone 4.5 µmol/l and LY294002 3.9 µmol/l; dark grey columns, rosiglitazone 4.5 µmol/l and wortmannin 50 nmol/l. *p < 0.05
Pharmacological activator of AMPK stimulates insulin secretion in isolated rat pancreas perfusion and in cultured MIN6 beta cells
At basal glucose concentration (5.5 mmol/l), perfusion with 50 μmol/l AICAR resulted in a 45% increase in insulin release within 2–3 min in the isolated rat pancreas. Insulin secretion increased progressively as perfusion continued, reached an increase of ∼100% at 30 min, and then declined gradually after AICAR was removed (Fig. 2a). Consistently, AMPK activator enhanced both first- and second-phase insulin secretion during perfusion with a high glucose concentration (10 mmol/l) (Fig. 2b).
Effect of AICAR on insulin secretion and AMPK activity. In pancreatic perfusion experiments (see Methods), an equilibration period of 15 min preceded time 0. a After a baseline period of 10 min AICAR (50 µmol/l) was administered for 30 min. Black circles, basal control group; white circles, AICAR 50 µmol/l. b Glucose (Glu; 10 mmol/l) with or without AICAR (50 µmol/l) was administered for 30 min, followed by perfusion with basal medium. Horizontal line indicates the presence of AICAR. Values are mean and SE (n = 4). Black circles, basal control group; white circles, AICAR 50 µmol/l in the presence of glucose 10 mmol/l; black triangles, glucose 10 mmol/l. c Insulin secretory response in the absence or presence of AICAR (50 µmol/l) and/or LY294002 (3.9 µmol/l) and wortmannin (50 nmol/l) in low (3.3 mmol/l) and high (16.7 mmol/l) glucose in MIN6 cells. Data are relative insulin responses (mean % and SE [bar]) compared with that in glucose 3.3 mM (100% as referent) (n = 4). White columns, control, black columns, AICAR 50 µmol/l, light grey columns, AICAR 50 µmol/l and LY294002 3.9 µmol/l; dark grey columns, AICAR 50 µmol/l and wortmannin 50 nmol/l. *p < 0.05. d Western blot with anti-AMPK and anti-Thr172-phospho-AMPK antibodies in the absence or presence of AICAR (50 µmol/l) and/or LY294002 (3.9 µmol/l) in low (3.3 mmol/l) and high (16.7 mmol/l) glucose in MIN6 cells. pAMPK/AMPK density relative to that in glucose 3.3 mmol/l is shown below (n = 3). White columns, control; black columns, AICAR 50 μmol/l; light grey columns, AICAR 50 μmol/l and LY294002 3.9 μmol/l. *p < 0.05, non-parametric analysis. e AMPK enzyme activity in the absence or presence of AICAR (50 μmol/l) and/or LY294002 (3.9 μmol/l) in low (3.3 mmol/l) and high (16.7 mmol/l) glucose in MIN6 cells. Data are relative AMPK activities (mean % and SE [bar]) compared with that in glucose 3.3 mmol/l (100% as referent) (n = 4). The experimental condition for each column is the same as that for the corresponding column in d
In MIN6 cells, AICAR increased insulin secretory responses by 35% and 47% at low and high glucose concentrations (3.3 and 16.7 mmol/l), respectively (Fig. 2c). The stimulatory effect of AICAR on insulin secretion was not inhibited by pretreatment with a PI3K inhibitor (LY294002 or wortmannin) (Fig. 2c). In both low and high glucose concentrations, the responses in the presence of AICAR and LY294002 or wortmannin were still significantly different from those of the controls, as tested by ANOVA (Fig. 2c). Consistently, AICAR stimulated Thr172 phosphorylation of the α subunit of AMPK (Fig. 2d) and its kinase activity (Fig. 2e), which were not inhibited by pretreatment with the PI3K inhibitor LY294002.
Rosiglitazone inhibits KATP channels through a PI3K-dependent AMPK signalling pathway
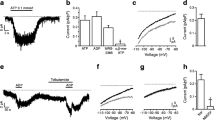
The effects of rosiglitazone and AICAR on IKATP were examined in primary cultures of rat islet cells. The holding currents at a membrane potential of −40 mV were recorded continuously in the absence or presence of the indicated treatments (Fig. 3a). Because the experimental conditions reached a steady state, a ramp voltage protocol (with slope 80 V/s) was employed to examine the currents elicited from −80 mV to 0 mV (Fig. 3a) and to construct the current–voltage curves in response to the respective treatments (Fig. 3b). Figure 3a showed that IKATP was gradually activated and developed to steady amplitude after intracellular dialysis in a low ATP condition; the effect was completely abolished by glibenclamide. Rosiglitazone significantly inhibited glibenclamide-sensitive IKATP, but did not alter the residual currents in the presence of glibenclamide. When the cells were pretreated with 10 μmol/l LY294002 for 5 min before addition of rosiglitazone, the inhibitory effect of rosiglitazone on the IKATP was abolished (Fig. 3c, d), whereas inhibition at a high glucose concentration (16.7 mmol/l) was not affected (Fig. 3c, d)
Rosiglitazone-induced inhibition of KATP channel current in rat islets is mediated by PI3K. a Currents at −40 mV in response to the respective treatment were recorded continuously, and ramp currents [indicated as (1)–(6)]were elicited from −80 to 0 mV with slope 80 V/s. The cell was bathed in external solution containing glucose 3.3 mmol/l. IKATP was gradually activated and reached a steady amplitude after intracellular dialysis in low ATP (ATP 0.3 mmol/l, ADP 0.3 mmol/l, EGTA 11 mmol/l, CaCl2 1 mmol/l). (1), break-in; (2), IKATP at steady state; (3), treatment with rosiglitazone (RSG) 9 µmol/l for 5 min; (4), after washing; (5), treatment with glibenclamide 30 µmol/l to block IKATP; (6), after rosiglitazone 9 µmol/l for 5 min. Zero current on the vertical scale bar indicates the baseline of zero current. I(A/F), current density of KATP channel. b Current–voltage curves for ramp currents elicited in a. c LY294002 (10 µmol/l) pretreatment abolished the inhibitory effect of rosiglitazone at −40 mV and the ramp currents elicited with the same protocol as in a. IKATP was decreased after exposure to glucose 16.7 mmol/l. d K+ current density at −40 mV in response to rosiglitazone (RSG; 9 µmol/l) in the absence (−) or presence (+) of LY294002 (10 µmol/l) pretreatment. Significant difference between the presence and absence of LY294002 pretreatment (n = 5, *p < 0.05, Student’s t test). Significant inhibition of current by rosiglitazone under control condition; n = 5, † p < 0.05, paired t test. Black columns, control; white columns, LY294002 10 µmol/l
Figure 4 shows the effect of AMPK activation by AICAR on IKATP in the absence or presence of LY294002. AICAR inhibited IKATP (Fig. 4a, b), and the effect was unaffected by LY294002 (Fig. 4c, d).
Inhibition of KATP channel current by AMPK activator AICAR in rat islets is independent of PI3K activity. a Effect of AICAR on currents at −40 mV and ramp currents [indicated as (1)–(3)] elicited from −80 mV to 0 mV with slope 80 V/s. The cell was bathed in solution containing glucose 3.3 mmol/l. IKATP was gradually activated and reached a steady amplitude after intracellular dialysis with low ATP (ATP 0.3 mmol/l, ADP 0.3 mmol/l, EGTA 11 mmol/l/CaCl2 1 mmol/l). (1), break-in; (2), IKATP developed to a steady state condition; (3), after treatment with AICAR 100 µmol/l for 5 min. The zero current was indicated at the vertical scale bar. b Current–voltage curves of the ramp currents elicited in a. c Effect of LY294002 (LY; 10 µmol/l) pretreatment on the inhibitory effect of AICAR in the currents at −40 mV and ramp currents elicited with the same protocol as that used in panel a. d K+ current density at −40 mV in response to AICAR (100 µmol/l) with or without LY294002 (10 µmol/l) pretreatment. Black columns, control; white columns, AICAR 100 µmol/l. *p < 0.05, control vs AICAR; n = 4, Student’s t test
Both rosiglitazone and AICAR phosphorylate the Ser385 of the Kir6.2 subunit of the KATP channel in rat islets
According to the proteomic professional website (ExPASY proteomic server, http://tw.expasy.org, accessed 4 January 2006), a FSIS 385-containing motif (KFSISPDSLS, amino acids 381–390 of the Kir6.2 subunit of the KATP channel) might be a potential AMPK phosphorylation site. Using immunoprecipitation with anti-Kir6.2 antibodies followed by immunoblotting with anti-phospho-Ser385 Kir6.2 antibodies, we found that rosiglitazone stimulated Ser385 phosphorylation of the Kir6.2 subunit (Fig. 5a). Rosiglitazone-stimulated Ser385 phosphorylation was inhibited by the AMPK inhibitor compound C and the PI3K inhibitor LY294002 (Fig. 5a, b). The AMPK activator AICAR also stimulated Ser385 phosphorylation of the Kir6.2 subunit (Fig. 5c). We used the FSIS 385-containing peptide as a substrate in the AMPK kinase assay and found that AICAR stimulated a threefold increase in the phosphorylation of the FSIS 385-containing peptide of the Kir6.2 subunit at both low and high glucose concentrations (Fig. 5d). Consistently, rosiglitazone also potentiated the effect of AMPK on Ser385 phosphorylation four- to sixfold, and this phosphorylation was blocked by approximately 50% by treatment of cells with the AMPK inhibitor compound C (Fig. 5e). In further experiments, silencing the expression of AMPK resulted in abolishment of Ser385 phosphorylation of Kir6.2 and insulin secretion in RINm5F rat insulinoma cells (ESM Fig. 2a–c).
Phosphorylation of Ser385 of the Kir6.2 subunit of the KATP channel is stimulated by rosiglitazone through PI3K-dependent AMPK activation. a Immunoprecipitation with anti-Kir6.2 peptide antibody followed by Western blotting with anti-phosphoserine385 Kir6.2 peptide antibody or anti-Kir6.2 peptide antibody in the absence or presence of rosiglitazone (RSG; 9 µmol/l) and/or compound C (C.C; 40 µmol/l, pretreatment for 1 h) in low (3.3 mmol/l) and high (16.7 mmol/l) glucose in rat islets. b Immunoprecipitation with anti-Kir6.2 peptide antibody followed by Western blotting with anti-phosphoserine385 Kir6.2 peptide antibody or anti-Kir6.2 peptide antibody in the absence or presence of rosiglitazone (9 µmol/l) and/or pretreatment with different concentrations of LY294002 (10 or 50 µmol/l). c Immunoprecipitation with anti-Kir6.2 peptide antibody followed by Western blotting with anti-phosphoserine385 Kir6.2 peptide antibody or anti-Kir6.2 peptide antibody in the absence or presence of AICAR (100 µmol/l) in rat islets. d Phosphorylation of the FSIS-containing peptide of Kir6.2 in the i.p. AMPK kinase assay from rat islets in the absence or presence of AICAR (50 µmol/l). White columns, control; black columns, AICAR 100 µmol/l. Glu, glucose. e Phosphorylation of the FSIS-containing peptide of Kir6.2 in the immunoprecipitated AMPK kinase assay from rat islets in the absence or presence of rosiglitazone (9 µmol/l) and/or compound C (40 µmol/l, pretreatment for 1 h) in rat islets. Data are relative AMPK activities (mean % and SE [bar]) compared with that in glucose (Glu) 3.3 mmol/l (100% as referent). White columns, control; black columns, rosiglitazone 9 µmol/l; light grey columns, rosiglitazone 9 µmol/l and compound C 40 µmol/l; dark grey columns, compound C 40 µmol/l. *p < 0.05, control vs treatment; n = 4, Student’s t test
Replacing Ser385 with Ala in the Kir6.2 subunit abolishes rosiglitazone- and AICAR-stimulated depolarisation of membrane potential in RINm5F cells
Flow cytometric measurement of the change in membrane potential was employed to examine differences in pharmacological responses to rosiglitazone and AICAR in RINm5F cells transfected with either wild-type Kir6.2 or the Ser385Ala mutant of the Kir6.2 subunit. Cells with high FL1 signal were identified as transfected cells for the expression of EGFP. RINm5F cells were incubated in KRB solutions containing DiBCA4(5) 400 nmol/l to measure the change in membrane potential from the FL2 signal. Figure 6b−d shows that the geometric mean of FL2 was increased after exposure to glibenclamide, rosiglitazone or AICAR in the control group (no transfection). In RINm5F cells transfected with either wild-type Kir6.2 or Ser385Ala mutant Kir6.2, the FL2 signal gated from the same cells in either the R1 or the R2 region of FL1 (Fig. 6e & j) was used to measure the changes in membrane potential in response to the indicated treatments. The membrane potential was significantly depolarised by adding rosiglitazone and AICAR in RINm5F cells transfected with the wild-type human Kir6.2 subunit (Fig. 6h & i), but not in cells transfected with the Ser385Ala mutant Kir6.2 subunit (Fig. 6m & n). Glibenclamide significantly depolarised the membrane potential of RINm5F cells transfected with either the wild-type Kir6.2 subunit or the Ser385Ala mutant of Kir6.2 (Fig. 6g & l). The percentage increments, compared with controls, in different experimental models are shown in Fig. 6o.
Depolarisation of membrane potentials by either rosiglitazone or AICAR in non-transfected and wild-type Kir6.2-transfected RINm5F cells but not in Ser385Ala Kir6.2 mutant-transfected RINm5F cells. Membrane potential was measured by flow cytometry and geometric means of FL2 fluorescence were compared. a–d FL2 fluorescence in non-transfected RINm5F cells in the absence (a) or presence of glibenclamide (b; 30 µmol/l), rosiglitazone (c; 9 µmol/l) and AICAR (d; 300 µmol/l). e–i FL2 fluorescence in wild-type Kir6.2 transfected RINm5F cells in the absence (f) or presence of glibenclamide (g; 30 µmol/l), rosiglitazone (h; 9 µmol/l) and AICAR (i; 300 µmol/l). The FL2 signal gated from the same cells in the R1 region (e; purple, non-transfected cells; green, cells transfected with Kir6.2 wild-type) of FL1 was used to measure the change in membrane potential in response to treatment. j–n FL2 fluorescence in Ser385Ala Kir6.2 mutant-transfected RINm5F cells in the absence (k) or presence of glibenclamide (l; 30 µmol/l), rosiglitazone (m; 9 µmol/l) and AICAR (n; 300 µmol/l). The FL2 signal gated from the same cells in the R2 region (j; purple, non-transfected cells; red, cells transfected with Ser385Ala Kir6.2 mutant) of FL1 was used to measure the change in membrane potential in response to treatment. FL1-H: FL1 histogram; FL2-H: FL2 histogram. o Percentage increments compared with controls in different cell condition. White columns, control; black columns, glibenclamide 30 µmol/l; light grey columns, rosiglitazone 9 µmol/l; dark grey columns, AICAR 300 µmol/l. *p < 0.05, n = 4
Discussion
In this study we provide the first evidence showing the role of AMPK in rosiglitazone-induced and PI3K-dependent insulin secretion in pancreatic beta cells. The results also indicate that AMPK-mediated Ser385 phosphorylation of the Kir6.2 subunit of the KATP channel is critical in modulating KATP channel activity.
Previously, we used an isolated perfused pancreas preparation to demonstrate that rosiglitazone acutely stimulates insulin secretion at either a basal or a high glucose level [15]. More interestingly, both first- and second-phase insulin secretion induced by high glucose was potentiated by rosiglitazone in a PI3K-dependent manner [15]. Thiazolidinediones have been shown to activate AMPK in various tissues [28–30], and we also found that rosiglitazone acutely stimulates Thr172 phosphorylation of the α subunit of AMPK, which leads to a two- to fourfold increase in enzyme activity of AMPK in pancreatic beta cells. These effects were abolished by LY294002 and wortmannin, suggesting that PI3K is an upstream regulator of AMPK activation. A similar pathway has been reported in bovine aortic endothelial cells [31].
The activity of membrane KATP channels is decreased by the increased intracellular ATP/ADP ratio that occurs during the physiological response to glucose stimulation [20]. Certain pharmacological agents that increase insulin secretion, such as sulfonylureas and meglitinides, block KATP channel activity by binding directly to the SUR1 subunit, and lead to depolarisation of the membrane potential; this depolarisation results in opening of voltage-dependent calcium channels, which leads to elevation of intracellular calcium levels and ultimately stimulates insulin secretion [32, 33]. Although thiazolidinediones have been shown to stimulate insulin secretion in insulin-secreting cells [13, 14], the pathway leading to closure of KATP channels is not clear. Using patch clamp studies, we showed that rosiglitazone acutely blocks KATP channel activity, an effect dependent on PI3 kinase activity. In contrast, glucose-induced closure of KATP channels is independent of PI3 kinase activity. These data are consistent with our previous findings showing that rosiglitazone-stimulated insulin secretion is blocked by the PI3K inhibitor LY294002, whereas glucose-stimulated insulin secretion is not affected by LY294002 [15].
Protein phosphorylation is an important biological process that is a possible mechanism for the regulation of the activities of ion channels and a wide variety of cellular functions [34]. In addition to being regulated by various nucleotides, the activity of the KATP channel can be further modulated by phosphorylation events induced by activated protein kinase A and protein kinase C [21, 22]. These phosphorylation events occur at selective amino acid residues within specific peptide motifs. These phosphorylation events lead to enhanced channel activity. In this study, we demonstrated that the Ser385 residue within the peptide motif of KFSISPDSLS was phosphorylated by activation of AMPK. On the other hand, silencing the α subunit of AMPK inhibited rosiglitazone-stimulated phosphorylation of Ser385 of the Kir6.2 subunit and subsequent insulin secretion, indicating that rosiglitazone-stimulated insulin secretion acts at least partly through an AMPK-dependent pathway. To establish unequivocally the role of Ser385 in the Kir6.2 subunit, we demonstrated that both rosiglitazone- and AICAR-induced depolarisation of membrane potential in RINm5F cells was markedly attenuated in cells overexpressing the Ser385Ala mutant of the Kir6.2 subunit. These data suggest that Ser385 phosphorylation is essential and necessary for rosiglitazone-stimulated insulin secretion. It is worthy of note that the 15 amino acid peptide sequence (AKAKPRFSISPDSLS) at the C terminus of Kir6.2 is highly conserved across different species, suggesting that this region has an important biological function in the regulation of KATP channel activity.
The effects of thiazolidinediones on pancreatic beta cell function are not consistent among different reports, mostly because of differences in experimental design [4, 13, 14, 35–42]. More studies need to be done to substantiate our understanding of the signalling pathway involved in rosiglitazone-induced insulin secretion through regulation of KATP channel activity by AMPK. For example, an attempt should be made to replicate our results using islets from AMPK−/− mice to verify the role of this important molecule in physiological and pharmacological stimulation of insulin secretion. Previous reports have shown different relationships between the activation of AMPK and glucose-stimulated insulin secretion [16–18, 43–45]. However, these differences may be explained in large part by the different glucose concentrations and experimental systems employed in the different studies. Forced increases in AMPK activity by AICAR treatment or by adenoviral overexpression of a truncated, constitutively active form of the enzyme (AMPKα1.T172D) have been shown to block glucose-stimulated insulin secretion in MIN6 cells and isolated rat islets [44] and lead to apoptosis of beta cells [46]. By contrast, inactivation of AMPK by expression of a dominant-negative form of the enzyme (AMPKα1.D157A) increases insulin secretion at a low glucose concentration (3 mmol/l) but not at a high glucose concentration [44]. By the use of another pharmacological agent, metformin, which has been shown to stimulate AMPK activation [45], results were even more conflicting when metformin was used alone [45] or in combination of glibenclamide [47]. In the present study, we found that short-term treatment with the AMPK activator AICAR stimulated insulin secretion, an effect that was independent of glucose concentration and was mediated by inhibition of KATP channel activity. Stimulation of insulin secretion in response to AMPK activation by AICAR at physiological glucose concentrations [16–18, 48] is in agreement with our present findings.
In conclusion, we report that rosiglitazone-stimulated insulin secretion occurs through PI3K-dependent activation of AMPK and subsequent inhibition of the KATP channel current (see model in Fig. 7). KATP channel activity is probably regulated by phosphorylation of the Ser385 residue of the Kir6.2 subunit by activation of AMPK in pancreatic beta cells.
Pathways leading to insulin release in pancreatic beta cells. ER, endoplasmic reticulum; RSG, rosiglitazone; SG, secretory granules; VDCC, voltage-dependent calcium channel. The pathway from glucose-stimulated insulin secretion is depicted according to Straub and Sharp [20]
Abbreviations
- KATP :
-
ATP-sensitive potassium
- PI3K:
-
Phosphatidylinositol 3-kinase
- AMPK:
-
AMP-dependent protein kinase
- AICAR:
-
5-Aminoimidazole-4-carboxamide ribonucleoside
- SUR1:
-
Sulfonylurea receptor
- Kir6.2:
-
Inwardly rectifying K+ channel subunit
- IKATP :
-
Current of KATP channel
References
DeFronzo RA (1998) The triumvirate: β-cell, muscle, liver: a collusion responsible for NIDDM. Diabetes 37:667–687
Matthews D, Cull C, Stratton IM, Holman RR, Turner RC (1998) UKPDS 26: Sulphonylurea failure in non-insulin-dependent diabetic patients over six years. UK Prospective Diabetes Study (UKPDS) Group. Diabet Med 15:297–303
Lebovitz HE (1993) Insulin mimetic and insulin-sensitizing drugs. Diabetes Res Clin Pract 20:89–91
Masuda K, Okamoto Y, Tsuura Y et al (1995) Effects of troglitazone (CS-045) on insulin secretion in isolated rat pancreatic islets and HIT cells: an insulinotropic mechanism distinct from glibenclamide. Diabetologia 38:24–30
Shimabukuro M, Zhou YT, Lee Y, Unger RH (1998) Troglitazone lowers islet fat and restores beta cell function of Zucker diabetic fatty rats. J Biol Chem 273:3547–3550
Higa M, Zhou YT, Ravazzola M, Baetens D, Orci L, Unger RH (1999) Troglitazone prevents mitochondrial alterations, β cell destruction, and diabetes in obese prediabetic rats. Proc Natl Acad Sci USA 96:11513–11518
Lupi R, Guerra SD, Marselli L et al (2004) Rosiglitazone prevents the impairment of human islet function induced by fatty acids: evidence for a role of PPARγ2 in the modulation of insulin secretion. Am J Physiol Endocrinol Metab 286:E560–E567
Lee Y, Hirose H, Ohneda M, Johnson JH, McGarry JD, Unger RH (1994) Beta-cell lipotoxicity in the pathogenesis of non-insulin-dependent diabetes mellitus of obese rats: impairment in adipocyte-beta-cell relationship. Proc Natl Acad Sci USA 91:10878–10882
Lee Y, Hirose H, Zhou YT, Esser V, McGarry J, Unger R (1997) Increased lipogenic capacity of the islets of obese rats: a role in the pathogenesis of NIDDM. Diabetes 46:408–413
Shimabukuro M, Koyama K, Lee Y, Unger RH (1997) Leptin- or troglitazone-induced lipopenia protects islets from interleukin 1 beta cytotoxicity. J Clin Invest 100:1750–1754
Unger RH (1995) Lipotoxicity in the pathogenesis of obesity-dependent NIDDM: genetics and clinical implications. Diabetes 44:863–870
Juhl CB, Hollingdal M, Porksen N, Prange A, Lonnqvist F, Schmitz O (2003) Influence of rosiglitazone treatment on beta-cell function in type 2 diabetes: evidence of an increased ability of glucose to entrain high-frequency insulin pulsatility. J Clin Endocrinol Metab 88:3794–3800
Lee K, Ibbotson T, Richardson PJ, Boden PR (1996) Inhibition of KATP channel activity by troglitazone in CRI-G1 insulin-secreting cells. Eur J Pharmacol 313:163–167
Rowe ICM, Lee K, Khan RN, Ashford MLJ (1997) Effect of englitazone on KATP and calcium-activated non-selective cation channels in CRI-G1 insulin-secreting cells. Br J Pharmacol 121:531–539
Yang C, Chang TJ, Chang JC et al (2001) Rosiglitazone (BRL 49653) enhances insulin secretory response via phosphatidylinositol 3-kinase pathway. Diabetes 50:2598–2602
Akkan AG, Malaisse WJ (1994) Insulinotropic action of AICA riboside. I. Insulin release by isolated islets and the perfused pancreas. Diabetes Res 25:13–23
Malaisse WJ, Conget I, Sener A, Rorsman P (1994) Insulinotropic action of AICA riboside. II. Secretory, metabolic and cationic aspects. Diabetes Res 25:25–37
Wang CZ, Wang Y, Di A (2005) 5-Amino-imidazole carboxamide riboside acutely potentiates glucose-stimulated insulin secretion from mouse pancreatic islets by KATP channel-dependent and -independent pathways. Biochem Biophys Res Commun 330:1073–1079
Seino S (1999) ATP-sensitive potassium channels. A model of heteromultimeric potassium channel/receptor assemblies. Annu Rev Physiol 61:337–362
Straub SG, Sharp GW (2002) Glucose-stimulated signaling pathways in biphasic insulin secretion. Diabetes Metab Res Rev 18:451–463
Béguin P, Nagashima K, Nishimura M, Gonoi T, Seino S (1999) PKA-mediated phosphorylation of the human KATP channel: separate roles of Kir6.2 and SUR1 subunit phosphorylation. EMBO J 18:4722–4732
Light PE, Bladen C, Winkfein RJ, Walsh MP, French RJ (2000) Molecular basis of protein kinase C-induced activation of ATP-sensitive potassium channels. Proc Natl Acad Sci USA 97:9058–9063
Grodsky GM, Fanska RE (1975) The in vitro perfused pancreas. In: Hardman JG, O’Malley BW (eds) Methods in enzymology. Vol. 39, Hormone action, Part D. Isolated cells, tissues, and organ systems. Elsevier/Academic Press, New York, pp 364–372
Hale CN, Randle PJ (1963) Immunoassay of insulin with insulin antibody precipitate. Biochem J 88:137–146
Aspinwall CA, Qian WJ, Roper MG, Kulkarni RN, Kahn CR, Kennedy RT (2000) Roles of insulin receptor substrate-1, phosphatidylinositol 3-kinase, and release of intracellular Ca2+ stores in insulin-stimulated insulin secretion in beta-cells. J Biol Chem 275:22331–22338
Davies SP, Carling D, Hardie DG (1989) Tissue distribution of the AMP-activated protein kinase, and lack of activation by cyclic-AMP-dependent protein kinase, studied using a specific and sensitive peptide assay. Eur J Biochem 186:123–128
Chuang LM, Myers MG Jr, Seidner GA, Birnbaum MJ, White MF, Kahn CR (1993) Insulin receptor substrate 1 mediates insulin and insulin-like growth factor 1-stimulated maturation of Xenopus oocytes. Proc Natl Acad Sci USA 90:5172–5175
Fryer LG, Parbu-Patel A, Carling D (2002) The anti-diabetic drugs rosiglitazone and metformin stimulate AMP-activated protein kinase through distinct signaling pathways. J Biol Chem 277:25226–25232
Saha AK, Avilucea PR, Ye JM, Assifi MM, Kraegen EW, Ruderman NB (2004) Pioglitazone treatment activates AMP-activated protein kinase in rat liver and adipose tissue in vivo. Biochem Biophys Res Commun 314:580–585
Bergeron R, Russell RR 3rd, Young LH et al (1999) Effect of AMPK activation on muscle glucose metabolism in conscious rats. Am J Physiol Endocrinol Metab 276:E938–E944
Zou MH, Hou XY, Shi CM et al (2003) Activation of 5′-AMP-activated kinase is mediated through c-Src and phosphoinositide 3-kinase activity during hypoxia-reoxygenation of bovine aortic endothelial cells. Role of peroxynitrite. J Biol Chem 278:34003–34010
Lawrence CL, Proks P, Rodrigo GC et al (2001) Gliclazide produces high-affinity block of KATP channels in mouse isolated pancreatic beta cells but not rat heart or arterial smooth muscle cells. Diabetologia 44:1019–1025
Meyer M, Chudziak F, Schwanstecher C, Schwanstecher M, Panten U (1999) Structural requirements of sulphonylureas and analogues for interaction with sulphonylurea receptor subtypes. Br J Pharmacol 128:27–34
Levitan IB (1994) Modulation of ions channel by protein phosphorylation and dephosphorylation. Annu Rev Physiol 56:193–212
Ovalle F, Bell D (2004) Effect of rosiglitazone vs insulin on the pancreatic beta cell function of subjects with type 2 diabetes mellitus. Diabetes Care 27:2585–2589
Cavaghan MK, Ehrmann DA, Byme MM, Polonsky KS (1997) Treatment with the oral antidiabetic agent troglitazone improves beta cell responses to glucose in subjects with impaired glucose tolerance. J Clin Invest 100:530–537
Ovalle F, Bell DS (2002) Clinical evidence of thiazolidinedione-induced improvement of pancreatic β-cell function in patients with type 2 diabetes mellitus. Diabetes Obes Metab 4:56–59
Kim HI, Ahn YH (2004) Role of peroxisome proliferator-activated receptor-γ in the glucose-sensing apparatus of liver and β-cells. Diabetes 53:S60–S65
Diani AR, Sawada G, Wyse B, Murray FT, Khan M (2004) Pioglitazone preserves pancreatic islet structure and insulin secretory function in three murine models of type 2 diabetes. Am J Physiol Endocrinol Metab 286:E116–E122
Ohtani KI, Shimizu H, Sato N, Mori M (1998) Troglitazone (CS-045) inhibits β-cell proliferation rate following stimulation of insulin secretion in HIT-T15 cells. Endocrinology 139:172–178
Zawalich WS, Tesz G, Zawalich KC (2003) Contrasting effects of nateglinide and rosiglitazone on insulin secretion and phospholipase C activation. Metabolism 52:1393–1399
Dubois M, Pattou F, Kerr-Conte J (2000) Expression of peroxisome proliferator-activated receptor γ (PPARγ) in normal human pancreatic islet cells. Diabetologia 43:1165–1169
da Silva Xavier G, Leclerc I, Salt IP et al (2000) Role of AMP-activated protein kinase in the regulation by glucose of islet beta cell gene expression. Proc Natl Acad Sci USA 97:4023–4028
da Silva Xavier G, Leclerc I, Varadi A, Tsubol T, Moule SK, Rutter GA (2003) Role for AMP-activated protein kinase in glucose-stimulated insulin secretion and preproinsulin gene expression. Biochem J 371:761–774
Isabelle L, Woltersdorf WW, da Silva Xavier G et al (2004) Metformin, but not leptin, regulates AMP-activated protein kinase in pancreatic islets: impact on glucose-stimulated insulin secretion. Am J Physiol Endocrinol Metab 286:E1023–E1031
Richards SK, Parton LE, Leclerc I, Rutter GA, Smith RM (2005) Over-expression of AMP-activated protein kinase impairs pancreatic β-cell function in vivo. J Endocrinol 187:225–235
Lupi R, Marchetti P, Giannarelli R et al (1997) Effects of glibenclamide and metformin (alone or in combination) on insulin release from isolated human pancreatic islets. Acta Diabetol 34:46–48
Gleason CE, Lu D, Witters LA, Newgard CB, Birnbaum MJ (2007) The role of AMPK and mTOR in nutrient sensing in pancreatic β-cells. J Biol Chem 282:10341–10351
Acknowledgements
The authors thank H.-M. Chang (Department of Internal Medicine, National Taiwan University Hospital) and M.-J. Chen (Department of Internal Medicine, National Taiwan University Hospital) for technical support. This work was supported by grants (NSC 91-2314-B002-342, 92-2314-B002-183, 93-2314-B002-137 and 95-2314-B002-191) from the National Science Council of the Republic of China.
Duality of interest statement
The authors declare that there is no duality of interest associated with this manuscript.
Author information
Authors and Affiliations
Corresponding author
Additional information
T.-J. Chang and W.-P. Chen contributed equally to this study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
ESM Fig. 1
Effect of rosiglitazone on AMPK activation. a Western blot with anti-AMPK and anti-Thr172-phospho-AMPK antibodies in the presence of different concentrations of rosiglitazone (RSG; 0, 0.5, 1.5 and 4.5 μmol/l) in low (3.3 mmol/l) and high (16.7 mmol/l) glucose in MIN6 cells. pAMPK/AMPK density relative to that in glucose 3.3 mmol/l is shown below (n = 3). White columns, control; black columns, rosiglitazone 0.5 μmol/l; right diagonal hatching, rosiglitazone 1.5 μmol/l; left diagonal hatching, rosiglitazone 4.5 μmol/l. *p < 0.05 for comparisons between different concentrations of rosiglitazone and control; non-parametric analysis. b Western blot with anti-AMPK and anti-Thr172-phospho-AMPK antibodies in the absence or presence of rosiglitazone (4.5 μmol/l) in low (3.3 mmol/l) and high (16.7 mmol/l) glucose at different time points (0, 10, 30, 60 min) in MIN6 cells. pAMPK/AMPK density relative to that in glucose 3.3 mmol/l is shown below (n = 3). White columns, control; black columns, rosiglitazone 4.5 μmol/l for 10 min; right diagonal hatching, rosiglitazone 4.5 μmol/l for 30 min; left diagonal hatching, rosiglitazone 4.5 μmol/l for 1 h. *p < 0.05 for comparisons between different rosiglitazone exposure times and control; non-parametric analysis (PDF 106 kb)
ESM Fig. 2
Phosphorylation of Ser385 of the Kir6.2 subunit of the KATP channel and stimulatory effect of rosiglitazone on GSIS is inhibited by knocking down the α subunit of AMPK. a Western blot of AMPK α-subunit and β-actin in corresponding control, transfected with scrambled siRNA or siRNA of the AMPK α-subunit in RINm5F cells. b Immunoprecipitation with anti-Kir6.2 peptide antibody followed by Western blot with anti-phosphoserine385 Kir6.2 peptide antibody or anti-Kir6.2 peptide antibody in the absence or presence of rosiglitazone (RSG; 9 μmol/l) in corresponding control, transfected with scrambled siRNA or siRNA of the AMPK α-subunit in RINm5F cells. c Insulin secretory response in the absence or presence of rosiglitazone (9 μmol/l) in low (3.3 mmol/l) and high (16.7 mmol/l) glucose in RINm5F cells. Data are relative insulin responses (mean % and SE [bar]) compared with that of glucose 3.3 mmol/l (100% as reference) (n = 3). White columns, control without transfection; black columns, rosiglitazone 9 μmol/l without transfection; right diagonal hatching, control transfected with scrambled siRNA; left diagonal hatching, rosiglitazone 9 μmol/l transfected with scrambled siRNA; dotted columns, control transfected with AMPK siRNA; horizontal hatching, rosiglitazone 9 μmol/l transfected with AMPK siRNA. *p < 0.05; n.s., not significant (PDF 77 kb)
Rights and permissions
About this article
Cite this article
Chang, TJ., Chen, WP., Yang, C. et al. Serine-385 phosphorylation of inwardly rectifying K+ channel subunit (Kir6.2) by AMP-dependent protein kinase plays a key role in rosiglitazone-induced closure of the KATP channel and insulin secretion in rats. Diabetologia 52, 1112–1121 (2009). https://doi.org/10.1007/s00125-009-1337-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00125-009-1337-4