Abstract
Aims/hypothesis
Proinsulin is increased in persons at cardiovascular risk. Increased secretion of proinsulin relative to insulin has been suggested as a sign of defective conversion of proinsulin to insulin and C-peptide and is associated with beta cell dysfunction. It has also been suggested that proinsulin has more of a pro-atherogenic effect than insulin, the levels of which are also increased in the insulin resistance state. In this prospective population-based study, we examined whether the proinsulin:insulin ratio (PIR) or insulin:glucose ratio (IGR, an insulin resistance surrogate) predicted carotid plaque size in nondiabetic participants.
Materials and methods
The study included 1,859 men and 1,998 women aged 25–82 years from the Tromsø Study, who were examined with B-mode high resolution ultrasound at baseline in 1994–1995 and at follow-up in 2001–2002. All images were computer processed to yield mm2 measures of plaque. Proinsulin and insulin were measured at baseline. All analyses were stratified for sex.
Results
After adjusting for age, baseline plaque area, BMI, cholesterol, HDL-cholesterol, HbA1c, IGR, albumin:creatinine ratio, fibrinogen, BP and lifestyle factors (tobacco smoking, alcohol consumption, physical activity), PIR was significantly associated with plaque size at follow-up in women but not men. For each SD in the PIR in women, the mean plaque area increased by 0.97 mm2 (95% CI 0.44–1.50). IGR was not associated with carotid plaque size.
Conclusions/interpretation
The PIR is associated with progressive carotid artery plaque size in women.
Similar content being viewed by others
Introduction
Cardiovascular disease (CVD) due to atherosclerosis is one of the main causes of morbidity and mortality in the western world. The pathogenesis of atherosclerosis is multifactorial. Both the so-called metabolic syndrome (high BP, obesity, dyslipidaemia, insulin resistance with hyperinsulinaemia) and increased proinsulin have been reported to predict atherosclerosis and CVD [1, 2]. Whether increased proinsulin is simply an indicator of insulin resistance [3], or if it has other pro-atherogenic actions has been debated [4]. The relationship between proinsulin and CVD has not been fully clarified, and the results of earlier studies are inconsistent [1, 5, 6]. In some studies, the association between proinsulin and CVD was stronger than that between insulin and CVD [1], and proinsulin has been suggested to have a greater pro-atherogenic effect than insulin [7]. Proinsulin predicted first-ever stroke in nondiabetic individuals, independently of insulin sensitivity [8]; and in one study, treatment of diabetic patients with proinsulin appeared to increase mortality compared with treatment with insulin [9]. Population-based studies of subclinical atherosclerotic progression have mainly assessed intima–media thickness (IMT) of the common carotid artery. However, plaque size and growth are probably just as good or even better markers of atherosclerosis and CHD [10, 11]. Most studies that assess proinsulin as a risk factor for CVD include only men [1, 5–7]. Furthermore, the relationship between proinsulin or insulin and carotid atherosclerosis has not been explored with a population-based prospective design. The aim of the present study was to determine whether early signs of beta cell dysfunction, assessed by the proinsulin:insulin ratio (PIR) [12] or insulin:glucose ratio (IGR, a surrogate measurement for insulin sensitivity), were associated with expansion of carotid plaque area (CPA) in a general nondiabetic population.
Materials and methods
Participants
The Tromsø Study is a population-based prospective study with repeated health surveys of inhabitants of the municipality of Tromsø, Norway. In the fourth survey conducted (1994–1995; baseline in the present study), all participants aged 55–74 years and 5–10% of samples in the other age groups >24 years of age were offered an ultrasonographic examination of the right carotid artery. A total of 6,902 (76% of the eligible population) accepted, and ultrasound examination was performed in 6,727 participants [10].
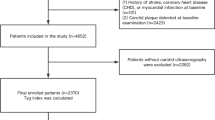
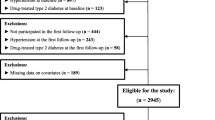
All the participants in the baseline study who were still alive and residing in Tromsø were offered a follow-up ultrasound examination in 2001–2002 (i.e. follow-up in the present study). During the period between the baseline and follow-up screenings, 532 participants died, 271 migrated, 956 did not participate in the follow-up examination and 110 did not receive follow-up scans due to logistical problems. Therefore, there were 4,858 participants at baseline and follow-up. Baseline measures of proinsulin, insulin, HbA1c and glucose were obtained for 4,127 of these participants. Data from participants who met the following criteria were excluded from analyses: self-reported diabetes (n = 96), use of glucose-lowering medication (n = 4), non-fasting plasma glucose >6.1 mmol/l (n = 103) or missing values for any of the analysed variables (n = 67). Thus, the final study population included 3,857 participants (1,859 men, 1,998 women). All the participants gave informed consent and the study was approved by the Regional Ethical Committee.
Cardiovascular risk factors
At the baseline examination, participants completed a self-administered questionnaire that included information about smoking habits, alcohol consumption, drug use, physical activity and time since last meal. We categorised time since last meal >5 h as fasting and time since last meal <5 h as non-fasting. Height, weight and waist circumference were measured in all participants. BP was recorded by a trained nurse using an automated device (Dinamap Vital Signs Monitor 1846; Critikon, Tampa, FL, USA). After a participant had been seated for 2 min, three BP recordings were obtained at 2 min intervals. The mean of the second and the third recordings was used for analysis. Total serum cholesterol was measured in a non-fasting blood sample by enzymatic colorimetric methods with commercial kits (CHOD-PAP for cholesterol and GPO-PAP for triacylglycerol; Boehringer-Mannheim, Mannheim, Germany). Serum HDL-cholesterol was measured after precipitation of LDL with heparin and manganese chloride. Data on fibrinogen and the albumin:creatinine ratio (ACR) have previously been published from the Tromsø Survey [13], and numbers for these variables were included in the present analyses as covariates. Fibrinogen was measured using the PT-Fibrinogen reagent (Instrumentation Laboratory, Milan, Italy). Three separate morning urine samples were analysed for albumin and creatinine [13]. Urinary albumin and creatinine were both measured with commercial kits (ABX Diagnostics, Montpellier, France). The mean albumin and creatinine values were used to calculate the ACR in mg/mmol.
Intact proinsulin was measured with a commercial kit (Dako Diagnostics Ltd, Ely, UK), using mouse monoclonal antibodies, with no insulin cross-reaction, C-peptide or 32–33 split proinsulin. Plasma insulin was measured by ELISA, which was modified from a previously published procedure [14]. The plasma insulin assay had <0.2% cross-reactivity with proinsulin or its primary circulating split form, Des31,31 HPI. Glucose was measured by the hexokinase method, in which NADPH was measured photometrically (Hitachi 917 analyser; Roche Diagnostics, Mannheim, Germany). HbA1c levels were measured in the haemolysate by a latex-enhanced turbidimetric immunoassay on a Cobras Mira Plus instrument (Unimate 5 HbA1c; Hoffmann-La Roche, Basel, Switzerland). PIR and IGR were calculated to reduce the variation inherent in the use of non-fasting insulin values.
Physical activity categories were dichotomised as active (>1 h hard physical activity per week with prominent perspiration or breathlessness, or >3 h light activity without prominent perspiration or breathlessness) or inactive (all others). Physical activity during working hours was not included in the analyses. Participants were classified as current smokers, former smokers or those who had never smoked. Alcohol consumption was divided into three categories: (1) alcohol abstention (no consumption of alcohol); (2) low alcohol consumption (<7 units alcohol per week for men or <3.5 units for women): and (3) high alcohol consumption (≥7 units alcohol per week for men or ≥3.5 units for women). (One unit of alcohol equals 12 g of alcohol).
Ultrasonography and measures of atherosclerosis
As previously described in detail [15], carotid atherosclerosis was assessed with high-resolution B-mode ultrasonography performed with an ultrasound scanner (Acuson Xp10 128 ART-upgraded; Mountain View, CA, USA) that was equipped with a linear array transducer. Identical scans were done at baseline and follow-up. Personnel performing the ultrasound measurements had no knowledge of the results of the other examinations, the questionnaires or laboratory data. The right common, internal and external carotid arteries of participants were identified by combined B-mode and colour Doppler/pulsed-wave Doppler ultrasound. All atherosclerotic plaques from six carotid artery sites were recorded. These included sites in the near and far walls of the internal carotid artery, the bifurcation segment of the common carotid artery, and the common carotid artery from the bifurcation downstream to the supraclavicular region. A plaque was defined as a localised protrusion of the internal part of the vessel wall into the lumen of more than 50% compared with the adjacent IMT. For each plaque, a still image was recorded with the transducer parallel to the vessel wall and perpendicular to the point of maximum plaque thickness. The B-mode images of the plaques were stored on super-VHS videotape. B-mode images were subsequently digitised and transferred to a personal computer using a commercially available video grabber card (Matrox Meteor II) and Matrox Intellicam v2.07 software (High-Tech Digital Inc., Rolling Hills Estates, CA, USA), at a resolution of 768 × 576 pixels. The plaque area measurements were made with Adobe Photoshop software (version 7.0), by tracing the perimeter of each plaque with a cursor. For the resolution used in this study, a plaque area of 167 pixels corresponded to 1 mm2. For each participant, total CPA was defined as the sum of the plaque areas in the six carotid sites.
Between- and within-sonographer agreement on plaque occurrence was substantial, with κ values (95% CI) of 0.67 (0.58–0.76) and 0.80 (0.70–0.91). The mean interobserver arithmetic difference (SD) of plaque area was 1.0 (4.4) mm2, and the limits of agreement were ±8.6 mm2. The mean intraobserver arithmetic difference (SD) for sonographer 1 was 0.2 (3.1) mm2, and the limits of agreement were ±6.1 mm2. The corresponding values for sonographer 2 were 0.01 (3.8) and ±7.5 mm2 [10, 15, 16].
Statistical analyses
Analyses of predictors of carotid artery plaque size were done separately for men and women in univariable and multivariable linear regression analyses. Baseline data are presented as means±SD or proportions and compared by sex using t tests or χ 2 tests as appropriate. ACR was logarithmically transformed because of marked skewed distribution. In the linear regression analyses, total CPA at follow-up (i.e. 2001–2002) was the dependent variable, and baseline variables were used as independent variables. All regression analyses were adjusted for total CPA at baseline. Analyses of each age-adjusted baseline variable and total CPA and multiple analyses with all the variables entered in a single step were performed. Continuous variables were tested for nonlinear associations by including a quadratic term in the analyses. In the multiple linear regression analyses, baseline plaque CPA, age, BMI, cholesterol, HDL-cholesterol, HbA1c, systolic BP, physical activity, alcohol consumption, smoking status, antihypertensive medication and fasting condition were included as covariates. One sex-specific SD was used as a unit for the continuous variables in the regression analyses. All statistical analyses were performed using SPSS software version 13.0 (SPSS, Inc., Chicago, IL, USA). Statistical models were tested by two-tailed analyses. p values <0.05 were considered statistically significant.
Results
General characteristics of the participants are presented in Table 1. Serum levels of total cholesterol, HDL-cholesterol, ACR and fibrinogen were significantly higher in women compared with men, and PIR and IGR were significantly higher in men. Alcohol consumption was higher among men, and more women than men had never smoked. At baseline, 954 men and 818 women had one or more carotid plaques (Table 2). The increase in mean CPA over 7 years was 7.30 mm2 in men and 5.12 mm2 in women. Of the 905 men and 1,180 women who did not have carotid plaque at baseline, 412 men and 421 women had one or more plaques at the follow-up examination. The usage of lipid-lowering agents increased markedly from baseline to follow-up (2.0 to 15.2% among men and 1.8 to 11.5% among women). BMI increased significantly in both sexes (men: 26.0 kg/m2 (SD 3.1) to 26.8 kg/m2 (SD 3.5), p < 0.0005; women: 25.8 kg/m2 (SD 4.0) to 26.9 kg/m2 (SD 4.5), p < 0.0005), as did also the waist circumference (men: 94.2 cm (SD 10.8) to 95.7 cm (SD 10.2), p < 0.0005; women: 83.9 cm (11.5) to 85.4 cm (SD 11.5), p < 0.0005). At baseline 11.8% of the women used contraceptive pills or oestrogen substitution.
Table 3 shows linear regression analyses of pairwise associations between baseline variables and follow-up CPA adjusted for age and baseline CPA. Baseline systolic BP, fibrinogen and ln ACR were positively associated with CPA at follow-up for both sexes, while total cholesterol, proinsulin and PIR were associated with CPA in women only. For men and women, current smokers had larger plaque areas than nonsmokers.
The association between PIR and CPA in women remained significant in the multivariate analysis (β coefficient 0.97, 95% CI 0.44–1.50; Table 4). PIR was significant whether or not IGR was included in the analysis. In contrast, there was no association between IGR and CPA whether or not PIR was included in the analysis. Similar results were obtained when PIR was replaced by proinsulin. Adding the interaction variable, sex × PIR, in a multivariable regression analysis that included men and women showed that the association between PIR and plaque area was sex-dependent (p = 0.006). No sex differences were found for cholesterol (p = 0.357 for the interaction variable sex × total cholesterol). The association between PIR and CPA was further analysed in separate multiple regression analyses for participants with or without carotid plaques at baseline, using the same model. PIR was significant in women without plaques (β coefficient 0.92, 95% CI 0.55–1.30) but not in women with plaques at baseline (β coefficient 1.99, 95% CI 1.58–5.57). Because a substantial proportion of participants had no plaques at baseline or follow-up (493 men, 759 women), the requirement for constant variance of the residuals in the multiple linear regression analyses was not fully satisfied. Therefore, we performed ordinal regression analyses with a dependent variable at four levels that included those with regression of CPA, those with CPA change equal to zero, and those with progression of CPA (divided into two equal groups). The results of those analyses were similar to the linear regression analyses with significant associations between CPA and PIR in women (odds ratio 1.66, 95% CI 1.10–2.52) but not men. Furthermore, multiple linear regression analyses after excluding participants without plaques at both examinations showed results similar to the entire study population.
Discussion
Our population-based prospective study showed that proinsulin was associated with carotid plaque size in nondiabetic women. It has been suggested that high proinsulin concentrations reflect beta cell dysfunction and insufficient insulin processing by pancreatic beta cells [12]. Since our surrogate parameter of insulin sensitivity, IGR, was not related to the development of carotid plaques, our results lend support to earlier findings that proinsulin is an independent pro-atherosclerotic factor [2, 8, 17]. A tentative mechanism explaining this could be altered haemostasis through the association of proinsulin and increased plasminogen activator inhibitor-1 activity and increased fibrinogen levels [4, 18–21].
Many studies have shown a relationship between proinsulin, proinsulin split products, and atherosclerotic lesions, but they were all cross-sectional studies, and the majority of them included only men [1, 5, 22]. In a cross-sectional analysis of 1,456 nondiabetic participants in the Rancho Bernardo Study, proinsulin was positively associated with prevalent CHD in men and women [23]. In the Caerphilly Study, proinsulin, but not insulin, predicted incident CHD during a 14 year follow-up study of 1,181 men, although the association was attenuated when corrections were made for HDL-cholesterol and triacylglycerol [1]. Our finding of sex differences in the relationship between proinsulin and plaque progression lends support to findings of a significant association between proinsulin and stroke among women (but not among men) in a study of 94 nondiabetic patients with first-ever stroke [8].
Cross-sectional studies have looked at proinsulin and carotid IMT as an atherosclerotic marker [2, 22], but to the best of our knowledge this is the first population-based prospective study of proinsulin and atherosclerosis in nondiabetic men and women. IMT primarily represents medial hypertrophy related to hypertension [24, 25], and the association between IMT and CHD was weak in several studies [26, 27]. CPA was claimed to be a better predictor of coronary atherosclerosis than other noninvasive methods [11, 28]. CPA was also shown to be more sensitive than IMT for early detection of atherosclerosis [29].
Significant sex differences in the association between proinsulin and CPA are not surprising. Serum proinsulin tends to increase when type 2 diabetes develops, and several studies have shown that type 2 diabetes increases the risk of CHD in women to a greater extent than in men [30, 31]. A recent meta-analysis of 37 prospective cohort studies showed that the risk of fatal CHD among diabetic individuals was significantly greater in women (relative risk 3.50, CI 2.70–4.53) than men (relative risk 2.06, CI 1.81–2.34) [30]. A cross-sectional study including 1,588 middle-aged participants showed that the effect of the metabolic syndrome on carotid atherosclerosis was more pronounced in women than men [32].
When we stratified for the presence of plaques at baseline, PIR remained significantly associated with plaque size only for women without plaques at baseline (n = 1,190). The association among women with plaques at baseline (n = 823) was positive, but not significant. The CI was quite wide, indicating that the group was rather small to determine an association.
In the present study, CPA increased with age in men and women, and smoking and higher systolic BP were associated with larger plaque areas for both sexes. Similar findings have previously been reported [10, 33]. Cholesterol was strongly associated with CPA in women but not in men, and fibrinogen was associated with CPA in men but not in women. However, analysing sex differences by adding the product variables cholesterol × sex and fibrinogen × sex in analyses that included both sexes did not result in significant sex differences. We previously reported that a high level of HDL-cholesterol was associated with less plaque growth in participants with preexisting carotid atherosclerosis [10]. In the current analysis, which also included individuals without atherosclerotic plaques at baseline, no such effect was demonstrated.
A shortcoming of the present study is that for logistical reasons we had to collect random and mainly non-fasting blood samples. However, the resulting variation in time since the last meal should have increased random measurement error of proinsulin and insulin and led to underestimation of the associations. When we corrected statistically for the non-fasting condition or linearly for hours since last meal intake, the results did not change substantially. Furthermore, proinsulin may be a more reliable measure of beta cell activity due to its longer half-life and lower variability than specific insulin when assessed in plasma samples [5]. This may favour proinsulin compared with insulin in regression analyses, and proinsulin is probably better than insulin for estimating future risk for CVD. Accordingly, the lack of significant association between IGR and CPA in our study could have been caused by the greater variation in non-fasting insulin compared with proinsulin.
On the other hand, the large sample size, the high participation rate and the prospective design of the study strengthen our findings. Moreover, the computerised analysis of CPA may have better reproducibility and is more accurate than visual assessment. The participants lost to follow-up were older, smoked more and had higher systolic BP, fibrinogen and CVD compared with the study population. This may have weakened the true relationship between risk factors at baseline and CPA at follow-up.
In summary, we found that PIR was independently associated with growth of carotid plaques in women but not men. The association was primarily present in the formation of new plaques. The study lends support to the hypothesis, but does not prove, that proinsulin has pro-atherogenic properties, at least in women.
Abbreviations
- ACR:
-
albumin:creatinine ratio
- CPA:
-
carotid plaque area
- CVD:
-
cardiovascular disease
- IGR:
-
insulin:glucose ratio
- IMT:
-
intima–media thickness
- PIR:
-
proinsulin:insulin ratio
References
Yudkin JS, May M, Elwood P, Yarnell JW, Greenwood R, Davey Smith G (2002) Concentrations of proinsulin like molecules predict coronary heart disease risk independently of insulin: prospective data from the Caerphilly Study. Diabetologia 45:327–336
Haffner SM, D’Agostino R, Mykkänen L et al (1998) Proinsulin and insulin concentrations in relation to carotid wall thickness: Insulin Resistance Atherosclerosis Study. Stroke 29:1498–1503
Haffner SM, Mykkänen L, Valdez RA et al (1994) Disproportionately increased proinsulin levels are associated with the insulin resistance syndrome. J Clin Endocrinol Metab 79:1806–1810
Perry IJ, Wannamethee SG, Whincup PH, Shaper AG, Walker MK, Alberti KG (1996) Serum insulin and incident coronary heart disease in middle-aged British men. Am J Epidemiol 144:224–234
Båvenholm P, Proudler A, Tornvall P et al (1995) Insulin, intact and split proinsulin, and coronary artery disease in young men. Circulation 92:1422–1429
Kahn SE, Leonetti DL, Prigeon RL, Boyko EJ, Bergstrom RW, Fujimoto WY (1995) Relationship of proinsulin and insulin with noninsulin-dependent diabetes mellitus and coronary heart disease in Japanese-American men: impact of obesity clinical research center study. J Clin Endocrinol Metab 80:1399–1406
Zethelius B, Lithell H, Hales CN, Berne C (2005) Insulin sensitivity, proinsulin and insulin as predictors of coronary heart disease. A population-based 10-year, follow-up study in 70-year old men using the euglycaemic insulin clamp. Diabetologia 48:862–867
Lindahl B, Dinesen B, Eliasson M, Røder M, Hallmans G, Stegmayr B (2000) High proinsulin levels precede first-ever stroke in a nondiabetic population. Stroke 31:2936–2941
Galloway JA, Hooper SA, Spradlin CT et al (1992) Biosynthetic human proinsulin. Review of chemistry, in vitro and in vivo receptor binding, animal and human pharmacology studies, and clinical trial experience. Diabetes Care 15:666–692
Johnsen SH, Mathiesen EB, Fosse E et al (2005) Elevated high-density lipoprotein cholesterol levels are protective against plaque progression: a follow-up study of 1952 persons with carotid atherosclerosis. The Tromsø study. Circulation 112:498–504
Spence JD (2002) Ultrasound measurement of carotid plaque as a surrogate outcome for coronary artery disease. Am J Cardiol 89:10B–15B, discussion 1
Mykkänen L, Haffner SM, Hales CN, Rönnemaa T, Laakso M (1997) The relation of proinsulin, insulin, and proinsulin-to-insulin ratio to insulin sensitivity and acute insulin response in normoglycemic subjects. Diabetes 46:1990–1995
Jørgensen L, Jenssen T, Johnsen SH et al (2007) Albuminuria as risk factor for initiation and progression of carotid atherosclerosis in non-diabetic persons: the Tromso Study. Eur Heart J 28:363–369
Jorde R, Burhol PG, Schulz TB et al (1981) The effect of a 34-h fast on the meal-induced rises in plasma GIP, serum insulin, and blood glucose in man. Scand J Gastroenterol 16:109–112
Fosse E, Johnsen SH, Stensland-Bugge E et al (2006) Repeated visual and computer-assisted carotid plaque characterization in a longitudinal population-based ultrasound study: the Tromsø study. Ultrasound Med Biol 32:3–11
Joakimsen O, Bønaa KH, Stensland-Bugge E (1997) Reproducibility of ultrasound assessment of carotid plaque occurrence, thickness, and morphology. The Tromsø Study. Stroke 28:2201–2207
Sayer JW, Marchant B, Gelding SV, Cooper JA, Timmis AD (2000) Autonomic dysfunction is related to impaired pancreatic beta cell function in patients with coronary artery disease. Heart 83:210–216
Mohamed-Ali V, Gould MM, Gillies S, Goubet S, Yudkin JS, Haines AP (1995) Association of proinsulin-like molecules with lipids and fibrinogen in non-diabetic subjects—evidence against a modulating role for insulin. Diabetologia 38:1110–1116
Gray RP, Mohamed-Ali V, Patterson DL, Yudkin JS (1995) Determinants of plasminogen activator inhibitor-1 activity in survivors of myocardial infarction. Thromb Haemost 73:261–267
Haffner SM, Mykkänen L, Stern MP, Valdez RA, Heisserman JA, Bowsher RR (1993) Relationship of proinsulin and insulin to cardiovascular risk factors in nondiabetic subjects. Diabetes 42:1297–1302
Festa A, D’Agostino R, Mykkänen L et al (1999) Relative contribution of insulin and its precursors to fibrinogen and PAI-1 in a large population with different states of glucose tolerance. The Insulin Resistance Atherosclerosis Study (IRAS). Arterioscler Thromb Vasc Biol 19:562–568
Bokemark L, Wikstrand J, Wedel H, Fagerberg B (2002) Insulin, insulin propeptides and intima–media thickness in the carotid artery in 58-year-old clinically healthy men. The Atherosclerosis and Insulin Resistance study (AIR). Diabet Med 19:144–151
Oh JY, Barrett-Connor E, Wedick NM (2002) Sex differences in the association between proinsulin and intact insulin with coronary heart disease in nondiabetic older adults: the Rancho Bernardo Study. Circulation 105:1311–1316
Linhart A, Gariepy J, Giral P, Levenson J, Simon A (1996) Carotid artery and left ventricular structural relationship in asymptomatic men at risk for cardiovascular disease. Atherosclerosis 127:103–112
Cuspidi C, Lonati L, Macca G et al (2001) Prevalence of left ventricular hypertrophy and carotid thickening in a large selected hypertensive population: impact of different echocardiographic and ultrasonographic diagnostic criteria. Blood Pressure 10:142–149
Adams MR, Nakagomi A, Keech A et al (1995) Carotid intima–media thickness is only weakly correlated with the extent and severity of coronary artery disease. Circulation 92:2127–2134
Hallerstam S, Larsson PT, Zuber E, Rosfors S (2004) Carotid atherosclerosis is correlated with extent and severity of coronary artery disease evaluated by myocardial perfusion scintigraphy. Angiology 55:281–288
Brook RD, Bard RL, Patel S et al (2006) A negative carotid plaque area test is superior to other noninvasive atherosclerosis studies for reducing the likelihood of having underlying significant coronary artery disease. Arterioscler Thromb Vasc Biol 26:656–662
Aminbakhsh A, Frohlich J, Mancini GB (1999) Detection of early atherosclerosis with B mode carotid ultrasonography: assessment of a new quantitative approach. Clin Invest Med 22:265–274
Huxley R, Barzi F, Woodward M (2006) Excess risk of fatal coronary heart disease associated with diabetes in men and women: meta-analysis of 37 prospective cohort studies. BMJ 332:73–78
Levantesi G, Macchia A, Marfisi R et al (2005) Metabolic syndrome and risk of cardiovascular events after myocardial infarction. J Am Coll Cardiol 46:277–283
Iglseder B, Cip P, Malaimare L, Ladurner G, Paulweber B (2005) The metabolic syndrome is a stronger risk factor for early carotid atherosclerosis in women than in men. Stroke 36:1212–1217
Rajala U, Laakso M, Päivänsalo M, Pelkonen O, Suramo I, Keinänen-Kiukaanniemi S (2002) Low insulin sensitivity measured by both quantitative insulin sensitivity check index and homeostasis model assessment method as a risk factor of increased intima–media thickness of the carotid artery. J Clin Endocrinol Metab 87:5092–5097
Acknowledgements
The present study was financially supported by the Norwegian Research Council, the Norwegian Foundation for Health and Rehabilitation, the Innlandet Hospital Trust and the University Hospital of North Norway Trust. We appreciate the superb technical assistance of Å. L. Bendikssen, J. H. Eikrem and H. Appelbom at the Laboratory of Metabolic Research, University of Tromsø, Norway.
Duality of interest
None of the authors has any duality of interest in connection with this study.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kronborg, J., Johnsen, S.H., Njølstad, I. et al. Proinsulin:insulin and insulin:glucose ratios as predictors of carotid plaque growth: a population-based 7 year follow-up of the Tromsø Study. Diabetologia 50, 1607–1614 (2007). https://doi.org/10.1007/s00125-007-0715-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00125-007-0715-z