Abstract
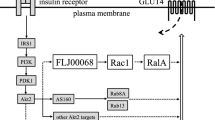
Insulin-stimulated glucose uptake in adipose tissue and striated muscle is critical for reducing post-prandial blood glucose concentrations and the dysregulation of this process is one hallmark of Type II (non-insulin-dependent) diabetes mellitus. It has been well established that the insulin-stimulated redistribution of the insulin responsive glucose transporter, GLUT-4, from intracellular storage sites to the plasma membrane depends on the production of phosphoinositide 3,4,5 trisphosphate by the Class IA Phosphatidylinositol 3' kinase. Recent discoveries however, have shown the presence of a second insulin signalling pathway leading to GLUT-4 translocation, a pathway dependent on insulin receptor signalling emanating from caveolae or lipid rafts at the plasma membrane. This pathway begins with the phosphorylation of the adaptor protein Cbl by the insulin receptor, and results in the activation of a small GTP binding protein, TC10, a member of the Rho family. TC10 is able to modulate actin structure in 3T3L1 adipocytes, and its overexpression inhibits insulin-stimulated GLUT-4 translocation, an inhibition completely dependent on localization of TC10 to the caveolae or lipid rafts. The spatial compartmentalization of insulin signalling from caveolae or lipid rafts provides a novel signalling pathway that functions in concert with general signalling mechanisms in the control of actin dynamics regulating insulin-dependent GLUT-4 translocation.
Article PDF
Similar content being viewed by others
Author information
Authors and Affiliations
Additional information
Electronic Publication
Rights and permissions
About this article
Cite this article
Khan, .A., Pessin, .J. Insulin regulation of glucose uptake: a complex interplay of intracellular signalling pathways. Diabetologia 45, 1475–1483 (2002). https://doi.org/10.1007/s00125-002-0974-7
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/s00125-002-0974-7