Abstract
Key message
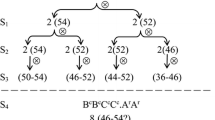
Different digenomic Brassica autoallohexaploids were produced from the crosses of three allotetraploids and ancestral diploids and characterized for the cytological behavior of two subgenomes with two and four copies.
Abstract
Interspecific hybridization and allopolyploidization present an important pathway for plant evolution and breeding. In this study, different types of digenomic autoallohexaploids with two or four copies of two subgenomes (AAAACC, AACCCC, AAAABB, BBBBCC, BBCCCC) were synthesized by the crosses between three Brassica allotetraploids and their diploid progenitors and the chromosome doubling, and their meiotic behaviors were analyzed by fluorescence in situ hybridization (FISH). These autoallohexaploids showed some variations in pollen fertility and seed-sets and produced both euploid and aneuploid progenies with some chromosomes lost. Two subgenomes in these autoallohexaploids showed some aberrant pairings and segregations, and the degrees of meiotic regularity were negatively associated with the genome affinities. The chromosomes of the subgenome with four copies formed few quadrivalents with the average number < 2, and mainly paired as bivalents, and majority of the chromosomes from the subgenome with two copies gave the expected bivalents. The different extents of the equal and unequal segregations corresponded to the chromosome pairings. The development and cytological investigation of these autoallohexaploids provide not only the new germplasm for genetic research and breeding but also the new clues for the genome behavior and interplay between these subgenomes with different copies.




Similar content being viewed by others
Data availability
The hybridization and chromosome behavior data used to support the findings of this study are included within the article.
References
Attia T, Röbbelen G (1986) Cytogenetic relationship within cultivated Brassica analyzed in amphihaploids from the three diploid ancestors. Can J Genet Cytol. https://doi.org/10.1139/g86-048
Bock DG, Kane NC, Ebert DP, Rieseberg LH (2014) Genome skimming reveals the origin of the jerusalem artichoke tuber crop species: neither from jerusalem nor an artichoke. New Phytol 201:1021–1030. https://doi.org/10.1111/nph.12560
Cai HW, Yuyama N, Tamaki H, Yoshizawa A (2003) Isolation and characterization of simple sequence repeat markers in the hexaploid forage grass timothy (Phleum pratense L.). Theor Appl Genet 107:1337–1349. https://doi.org/10.1007/s00122-003-1386-x
Chalhoub B, Denoeud F, Liu S et al (2014) Early allopolyploid evolution in the post-neolithic Brassica napus oilseed genome. Science 345:950–953. https://doi.org/10.1126/science.1253435
Chatterjee D, Banga S, Gupta M et al (2016) Resynthesis of Brassica napus through hybridization between. Theor Appl Genet 129:977–990. https://doi.org/10.1007/s00122-016-2677-3
Chen S, Nelson MN, Chèver AM et al (2011) Trigenomic bridges for Brassica improvement. Crit Rev Plant Sci 30:524–547. https://doi.org/10.1080/07352689.2011.615700
Comai L (2005) The advantages and disadvantages of being polyploid. Nat Rev Genet 6:836–846. https://doi.org/10.1038/nrg1711
Cui C, Ge X, Gautam M et al (2012) Cytoplasmic and genomic effects on meiotic pairing in Brassica hybrids and allotetraploids from pair crosses of three cultivated diploids. Genetics 191:725–738. https://doi.org/10.1534/genetics.112.140780
Ferreira de Carvalho J, Stoeckel S, Eber F et al (2021) Untangling structural factors driving genome stabilization in nascent Brassica napus allopolyploids. New Phytol 230:2072–2084. https://doi.org/10.1111/nph.17308
Gaebelein R, Mason AS (2018) Allohexaploids in the Genus Brassica. Crit Rev Plant Sci 37:422–437. https://doi.org/10.1080/07352689.2018.1517143
Gaeta RT, Pires JC, Iniguez-Luy F et al (2007) Genomic changes in resynthesized Brassica napus and their effect on gene expression and phenotype. Plant Cell 19:3403–3417. https://doi.org/10.1105/tpc.107.054346
Ge XH, Li ZY (2007) Intra- and intergenomic homology of B-genome chromosomes in trigenomic combinations of the cultivated Brassica species revealed by GISH analysis. Chromosome Res 15:849–861. https://doi.org/10.1007/s10577-007-1168-4
Ge XH, Ding L, Li ZY (2013) Nucleolar dominance and different genome behaviors in hybrids and allopolyploids. Plant Cell Rep 32:1661–1673. https://doi.org/10.1007/s00299-013-1475-5
Gorgidze A D (1970). Synthetic reproduction of primary hexaploid cultivated wheat Triticum macha Dek. et Men. Soobshch An Gruzssr 58:685–688. https://www.cabdirect.org/cabdirect/abstract/19721605385
Gupta M, Gupta S, Kumar H et al (2015) Population structure and breeding value of a new type of Brassica juncea created by combining A and B genomes from related allotetraploids. Theor Appl Genet 128:221–234. https://doi.org/10.1007/s00122-014-2423-7
Harberd DJ (1972) A contribution to the cyto-taxonomy of Brassica (Cruciferae) and its allies. Bot J Linn Soc 65:1–25
Hardberg DJ (1976) Cytotaxonomic studies of Brassica and related genera. In: Vaughan JG, MacLeod AJ, Jones BM (eds) The biology and Chemistry of the Crucifereae. Academic Press, London, pp 47–68
Jenczewski E, Eber F, Manzanares-Dauleux MJ, Chevre AM (2002) A strict diploid-like pairing regime is associated with tetrasomic segregation in induced autotetraploids of kale. Plant Breed 121:177–179. https://doi.org/10.1046/j.1439-0523.2002.00672.x
Jenczewski E, Eber F, Grimaud A et al (2003) PrBn, a major gene controlling homeologous pairing in oilseed rape (Brassica napus) haploids. Genetics 164:645–653. https://doi.org/10.1093/genetics/164.2.645
Katche E, Gaebelein R, Idris Z et al (2021) Stable, fertile lines produced by hybridization between allotetraploids Brassica juncea (AABB) and Brassica carinata (BBCC) have merged the A and C genomes. New Phytol 230:1242–1257. https://doi.org/10.1111/nph.17225
Lagercrantz U, Lydiate DJ (1996) Comparative genome mapping in Brassica. Genetics 144:1903–1910. https://doi.org/10.1093/genetics/144.4.1903
Li Z, Liu HL, Luo P (1995) Production and cytogenetics of intergeneric hybrids between Brassica napus and Orychophragmus violaceus. Theor Appl Genet 91:131–136. https://doi.org/10.1007/BF00220869
Li Q, Mei J, Zhang Y et al (2013) A large-scale introgression of genomic components of Brassica rapa into B. napus by the bridge of hexaploid derived from hybridization between B. napus and B. oleracea. Theor Appl Genet 126:2073–2080. https://doi.org/10.1007/s00122-013-2119-4
Liu Z, Adamczyk K, Manzanares-Dauleux M et al (2006) Mapping PrBn and other quantitative trait loci responsible for the control of homeologous chromosome pairing in oilseed rape (Brassica napus L.) haploids. Genetics 174:1583–1596. https://doi.org/10.1534/genetics.106.064071
Lloyd A, Bomblies K (2016) Meiosis in autopolyploid and allopolyploid Arabidopsis. Curr Opin Plant Biol 30:116–122. https://doi.org/10.1016/j.pbi.2016.02.004
Mason AS, Huteau V, Eber F et al (2010) Genome structure affects the rate of autosyndesis and allosyndesis in AABC, BBAC and CCAB Brassica interspecific hybrids. Chromosome Res 18:655–666. https://doi.org/10.1007/s10577-010-9140-0
Mason AS, Nelson MN, Takahira J et al (2014) The fate of chromosomes and alleles in an allohexaploid Brassica population. Genetics 197:273–283. https://doi.org/10.1534/genetics.113.159574
McNaughton IH (1973a) Brassica napocampestris L. (2n=58). 1. Synthesis, cytology, fertility and general considerations. Euphytica 22:301–309. https://doi.org/10.1007/BF00022638
McNaughton IH (1973b) Synthesis and sterility of Raphanobrassica. Euphytica 22:70–88. https://doi.org/10.1007/BF00021558
Mei J, Liu Y, Wei D et al (2015) Transfer of sclerotinia resistance from wild relative of Brassica oleracea into Brassica napus using a hexaploidy step. Theor Appl Genet 128:639–644. https://doi.org/10.1007/s00122-015-2459-3
Mizushima U (1950). Karyogenetic studies of species and genus hybrids in the tribe Brassicae. - Tohoku J Agr Res I: 1–14.
Murat F, Louis A, Maumus F, Armero A, Cooke R, Quesneville H, Roest Crollius H, Salse J (2015) Understanding Brassicaceae evolution through ancestral genome reconstruction. Genome Biol 16:262. https://doi.org/10.1186/s13059-015-0814-y
Prakash S, Hinata K (1980) Taxonomy, cytogenetics and origin of crop Brassicas, a review. Opera Bot 55:1–57
Ramsey J, Schemske DW (1998) Pathways, mechanisms, and rates of polyploid formation in flowering plants. Ann Rev Ecol Syst 29:467–501. https://doi.org/10.1146/annurev.ecolsys.29.1.467
Ramsey J, Schemske DW (2002) Neopolyploidy in flowering plants. Ann Rev Ecol Syst 33:589–639. https://doi.org/10.1146/annurev.ecolsys.33.010802.150437
Schelfhout CJ, Snowdon R, Cowling WA, Wroth JM (2004) A PCR based B-genome-specific marker in Brassica species. Theor Appl Genet 109:917–921. https://doi.org/10.1007/s00122-004-1713-x
Snowdon RJ (2007) Cytogenetics and genome analysis in Brassica crops. Chromosome Res 15:85–95. https://doi.org/10.1007/s10577-006-1105-y
Soltis DE, Albert VA, Leebens-Mack J et al (2009) Polyploidy and angiosperm diversification. Am J Bot 96:336–348. https://doi.org/10.3732/ajb.0800079
Stebbins GL (1947) Types of polyploids: Their classification and significance. Adv Genet 1:403–429. https://doi.org/10.1016/S0065-2660(08)60490-3
Szadkowski E, Eber F, Huteau V et al (2010) The first meiosis of resynthesized Brassica napus, a genome blender. New Phytol 186:102–112. https://doi.org/10.1111/j.1469-8137.2010.03182.x
Tang H, Woodhouse MR, Cheng F, Schnable JC, Pedersen BS, Conant G, Wang X, Freeling M, Pires JC (2012) Altered patterns of fractionation and exon deletions in Brassica rapa support a two-step model of paleohexaploidy. Genetics 190(4):1563–1574. https://doi.org/10.1534/genetics.111.137349
Tank DC, Eastman JM, Pennell MW et al (2015) Nested radiations and the pulse of angiosperm diversification: Increased diversification rates often follow whole genome duplications. New Phytol 207:454–467. https://doi.org/10.1111/nph.13491
U N, (1935) Genome analysis in Brassica with special reference to the experimental formation of B. napus and peculiar mode of fertilization. Jpn J Bot 7:389–452
Van De Peer Y, Maere S, Meyer A (2009) The evolutionary significance of ancient genome duplications. Nat Rev Genet 10:725–732. https://doi.org/10.1038/nrg2600
Weitz AP, Dukic M, Zeitler L, Bomblies K (2021) Male meiotic recombination rate varies with seasonal temperature fluctuations in wild populations of autotetraploid Arabidopsis arenosa. Mol Ecol 30:4630–4641. https://doi.org/10.1111/mec.16084
Xiong Z, Gaeta RT, Pires JC (2011) Homoeologous shuffling and chromosome compensation maintain genome balance in resynthesized allopolyploid Brassica napus. Proc Natl Acad Sci U S A 108:7908–7913. https://doi.org/10.1073/pnas.1014138108
Zhang K, Mason AS, Farooq MA et al (2021) Challenges and prospects for a potential allohexaploid Brassica crop. Theor Appl Genet 134:2711–2726. https://doi.org/10.1007/s00122-021-03845-8
Zhou J, Tan C, Cui C et al (2016) Distinct subgenome stabilities in synthesized Brassica allohexaploids. Theor Appl Genet 129:1257–1271. https://doi.org/10.1007/s00122-016-2701-7
Zhu B, Tu Y, Zeng P et al (2016) Extraction of the constituent subgenomes of the natural allopolyploid rapeseed (Brassica napus L.). Genetics 204:1015–1027. https://doi.org/10.1534/genetics.116.190967
Acknowledgements
We thank Dr. Jing Wang for her valuable suggestions in data analysis and contributions in laboratory management.
Funding
This work was supported by Natural Science Foundation of China (Grant No. 31771827).
Author information
Authors and Affiliations
Contributions
XG: conceived and designed the experiments. BC, TW, FY and AH: performed the experiments. BC. and BZ. analyzed the data, BC., XG, WQ. and ZL. wrote the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Annaliese S. Mason.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Cai, B., Wang, T., Yue, F. et al. Production and cytology of Brassica autoallohexaploids with two and four copies of two subgenomes. Theor Appl Genet 135, 2641–2653 (2022). https://doi.org/10.1007/s00122-022-04136-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00122-022-04136-6