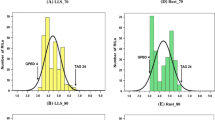
Abstract
Gray leaf spot (GLS) is a serious fungal disease caused by Magnaporthe grisea, recently reported on perennial ryegrass (Lolium perenne L.), an important turfgrass and forage species. This fungus also causes rice blast and many other grass diseases. Rice blast is usually controlled by host resistance, but durability of resistance is a problem. Little GLS resistance has been reported in perennial ryegrass. However, greenhouse inoculations in our lab using one ryegrass isolate and one rice-infecting lab strain suggest presence of partial resistance. A high density linkage map of a three generation Italian × perennial ryegrass mapping population was used to identify quantitative trait loci (QTL) for GLS resistance. Potential QTL of varying effect were detected on four linkage groups, and resistance to the ryegrass isolate and the lab strain appeared to be controlled by different QTL. Of three potential QTL detected using the ryegrass isolate, the one with strongest effect for resistance was located on linkage group 3 of the MFB parent, explaining between 20% and 37% of the phenotypic variance depending on experiment. Another QTL was detected on linkage group 6 of the MFA parent, explaining between 5% and 10% of the phenotypic variance. The two QTL with strongest effect for resistance to the lab strain were located on linkage groups MFA 2 and MFB 4, each explaining about 10% of the phenotypic variance. Further, the QTL on linkage groups 3 and 4 appear syntenic to blast resistance loci in rice. This work will likely benefit users and growers of perennial ryegrass, by setting the stage for improvement of GLS resistance in perennial ryegrass through marker-assisted selection.




Similar content being viewed by others
References
Armstead IP, Turner LB, Farrell M, Skøt L, Gomez P, Montoya T, Donnison, IS, King IP, Humphreys MO (2004) Synteny between a major heading-date QTL in perennial ryegrass (Lolium perenne L.) and the Hd3 heading-date locus in rice. Theor Appl Genet 108:822–828
Atkins JG, Robert AL, Adair CR, Goto K, Kozaka K, Yanagida R, Yamada M, Matsumoto S (1967) An international set of rice varieties for differentiating races of Piricularia oryzae. Phytopathology 57:297–301
Bert PF, Charmet G, Sourdille P, Hayward MD, Balfourier F (1999) A high-density molecular map for ryegrass (Lolium perenne L.) using AFLP markers. Theor Appl Genet 99:445–452
Bonman JM (1992) Blast. In: Webster RK, Gunnell PS (eds) Compendium of rice diseases. APS Press, St. Paul, pp 14–17
Bonos SA, Kubik C, Clarke BB, Meyer WA (2004) Breeding perennial ryegrass for resistance to gray leaf spot. Crop Sci 44:575–580
Calenge F, Faure A, Goerre M, Gebhardt C, Van de Weg WE, Parisi L, Durel C-E (2004) Quantitative trait loci (QTL) analysis reveals both broad-spectrum and isolate-specific QTL for scab resistance in an apple progeny challenged with eight isolates of Venturia inaequalis. Phytopathology 94:370–379
Causse M, Fulton T, Cho Y, Ahn S (1994) Saturated molecular map of the rice genome based on an interspecific backcross population. Genetics 138:1251–1274
Chauhan R, Farman ML, Zhang H-B, Leong SA (2002) Genetic and physical mapping of a rice blast resistance locus, Pi-CO39(t), that corresponds to the avirulence gene AVR1-CO39 of Magnaporthe grisea. Mol Gen Genomics 267:603–612
Chen D-H, dela Viña M, Inukai T, Mackill DJ, Ronald PC, Nelson RJ (1999) Molecular mapping of the blast resistance gene, Pi44(t), in a line derived from a durably resistant rice cultivar. Theor Appl Genet 98:1046–1053
Chen H, Wang S, Xing Y, Xu C, Hayes PM, Zhang Q (2003) Comparative analyses of genomic locations and race specificities of loci for quantitative resistance to Pyricularia grisea in rice and barley. Proc Natl Acad Sci USA 100:2544–2549
Fukuoka S, Okuno K (2001) QTL analysis and mapping of pi21, a recessive gene for field resistance to rice blast in Japanese upland rice. Theor Appl Genet 103:185–190
Hannaway D, Fransen S, Cropper J, Teel M, Chaney M, Griggs T, Halse R, Hart J, Cheeke P, Hansen D, Klinger R, Lane W (1999) Perennial ryegrass (Lolium perenne L.). Oregon State University Extension Publication PNW503
Hittalmani S, Parco A, Mew TV, Zeigler RS, Huang N (2000) Fine mapping and DNA marker-assisted pyramiding of the three major genes for blast resistance in rice. Theor Appl Genet 100:1121–1128
Hoffman N, Hamblin A (2001) Reaction of perennial ryegrass to gray leaf spot following inoculation in the field, 2000. Biol Cult Tests 16:T56
Jones ES, Mahoney NL, Hayward MD, Armstead HI, Jones JG, Humphreys MO, King IP, Kishida T, Yamada T, Balfourier F, Charmet G, Forster JW (2002) An enhanced molecular marker based genetic map of perennial ryegrass (Lolium perenne) reveals comparative relationships with other Poaceae genomes. Genome 45:282–295
Jung G, Coyne D, Skroch P, Nienhuis J, Arnaud-Santana E, Bokosi J, Ariyarathne H, Steadman J, Beaver J, Kaeppler S (1996) Molecular markers associated with plant architecture and resistance to common blight, web blight and rust in common beans (Phaseolus vulgaris L.). J Am Soc Hort Sci 121(5):794–803
Kellogg E (2000) The grasses: a case study in macroevolution. Ann Rev Ecol Syst 31:217–238
Lander E, Botstein D (1989) Mapping Mendelian factors underlying quantitative traits using RFLP linkage maps. Genetics 121:185–199
Landschoot P, Hoyland B (1992) Gray leaf spot of perennial ryegrass turf in Pennsylvania. Plant Dis 76:1280–1282
Melchinger AE (1990) Use of molecular markers in breeding for oligogenic disease resistance. Plant Breeding 104:1–19
Moss MA, Trevathan LE (1987) Environmental conditions conducive to infection of ryegrass by Pyricularia grisea. Phytopathology 77:863–866
Sallaud C, Lorieux M, Roumen E, Tharreau D, Berruyer R, Svestasrani P, Garsmeur O, Ghesquire A, Notteghem J-L (2003) Identification of five new blast resistance genes in the highly blast-resistant rice variety IR64 using a QTL mapping strategy. Theor Appl Genet 106:794–803
Sato K, Inukai T, Hayes PM (2001) QTL analysis of resistance to the rice blast pathogen in barley (Hordeum vulgare). Theor Appl Genet 102:916–920
Seebold KW, Kucharek TA, Datnoff LE, Correa-Victoria FJ, Marchetti MA (2001) The influence of silicon on components of resistance to blast in susceptible, partially resistant, and resistant cultivars of rice. Phytopathology 91:63–69
Sim S, Chang T, Curley J, Warnke S, Barker R, Jung G (2005) Chromosomal rearrangements differentiating the ryegrass genome from the Triticeae, oat, and rice genomes using common heterologous RFLP probes. Theor Appl Genet 110:1011–1019
Tabien RE, Li Z, Paterson AH, Marchetti MA, Stansel JW, Pinson SRM (2000) Mapping of four major rice blast resistance genes from ‘Lemont’ and ‘Teqing’ and evaluation of their combinatorial effect for field resistance. Theor Appl Genet 101:1215–1225
Tabien R, Li Z, Paterson AH, Marchetti MA, Stansel JW, Pinson SRM (2002) Mapping QTLs for field resistance to the rice blast pathogen and evaluating their individual and combined utility in improved varieties. Theor Appl Genet 105:313–324
Tredway LP, Stevenson KL, Burpee LL (2003) Components of resistance to Magnaporthe grisea in ‘Coyote’ and ‘Coronado’ tall fescue. Plant Dis 87:906–912
Trevathan LE, Moss MA, Blasingame D (1994) Ryegrass blast. Plant Dis 78:113–117
Uddin W, Viji G, Vincelli P (2003a) Gray leaf spot (blast) of perennial ryegrass turf-an emerging problem for the turfgrass industry. Plant Dis 87:880–889
Uddin W, Serlemitsos K, Viji G (2003b) A temperature and leaf wetness duration-based model for prediction of gray leaf spot of perennial ryegrass turf. Phytopathology 93:336–343
Van Ooijen JW, Voorrips RE (2001) JoinMap® 3.0: software for the calculation of genetic linkage maps. Plant Research International, Wageningen
Van Ooijen JW, Boer MP, Jansen RC, Maliepaard C (2002) MapQTL® version 4.0: software for the calculation of QTL positions on genetic maps. Plant Research International, Wageningen
Viji G, Wu B, Kang S, Uddin W, Huff DR (2001) Pyricularia grisea causing gray leaf spot of perennial ryegrass turf: population structure and host specificity. Plant Dis 85:817–826
Vincelli P, Dixon E (2002) Resistance to QoI (Strobilurin-like) fungicides in isolates of Pyricularia grisea from perennial ryegrass. Plant Dis 86:235–240
Wang G-L, Mackill DJ, Bonman JM, McCouch SR, Champoux MC, Nelson RJ (1994) RFLP mapping of genes conferring complete and partial resistance to blast in a durably resistant rice cultivar. Genetics 136:1421–1434
Warnke SE, Barker RE, Jung G, Sim S, Rouf Mian MA, Saha MC, Brilman LA, Dupal MP, Forster JW (2004) Genetic linkage mapping of an annual x perennial ryegrass population. Theor Appl Genet 109:294–304
Williams DW, Burrus PB, Vincelli P (2001) Severity of gray leaf spot in perennial ryegrass as influenced by mowing height and nitrogen level. Crop Sci 41:1207–1211
Yaneshita M, Ohmura T, Sasakua T, Ogihara Y (1993) Phylogenetic relationships of turfgrasses as revealed by restriction fragment analysis of chloroplast DNA. Theor Appl Genet 87:129–135
Zenbayashi K, Ashizawa T, Tani T, Koizumi S (2002) Mapping of the QTL (quantitative trait locus) conferring partial resistance to leaf blast in rice cultivar Chubu 32. Theor Appl Genet 104:547–552
Zhu M, Wang L, Pan Q (2004) Identification and characterization of a new blast resistance gene located on rice chromosome 1 through linkage and differential analyses. Phytopathology 94:515–519
Acknowledgements
We thank Drs. Mark Farman and Andy Hamblin for providing us the perennial ryegrass isolates, and Drs. Michael Havey and Michael Casler for critical reviews for the manuscript. We thank Jung lab members for their excellent technical assistance. Funding for this project was provided by the United States Golf Association and partially by the Wisconsin Turfgrass Association.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by T. Lübberstedt
Rights and permissions
About this article
Cite this article
Curley, J., Sim, S.C., Warnke, S. et al. QTL mapping of resistance to gray leaf spot in ryegrass. Theor Appl Genet 111, 1107–1117 (2005). https://doi.org/10.1007/s00122-005-0036-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00122-005-0036-x