Abstract
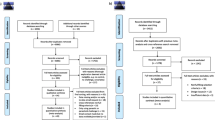
Immune response is evolutionary costly, but it is not clear whether these costs affect energetic expenditure (short-term cost), growth (medium-term cost), or reproduction (long-term cost). We tested the costs of immune memory in Tenebrio molitor against Metarhizium brunneum. To do this, we used two groups of T. molitor larvae: (a) the control group, which was injected first with Tween solution and 10 days later with M. brunneum and (b) the memory group, which was first injected with M. brunneum and 10 days later with M. brunneum. Compared to controls, larvae of the memory group were more likely to survive, but they also had an increased metabolic rate (CO2 production), spent a long time before becoming pupae, and had a shorter time from pupae to adulthood. In the adult stage, control females preferred control males, but there was no significant difference in the preference of memory females. Finally, control and memory males preferred control females. These results confirm that immune memory has costs in terms of energetic expenditure, growth, and reproduction. To the best of our knowledge, this is the first experimental demonstration that immune memory in larvae is traded-off with adult sexual selection involving mate choice.





Similar content being viewed by others
References
Ardia DR, Gantz JE, Strebel S (2012) Costs of immunity in insects: an induced immune response increases metabolic rate and decreases antimicrobial activity. Funct Ecol 26:732–739. https://doi.org/10.1111/j.1365-2435.2012.01989.x
Best A, Tidbury H, White A, Boots M (2013) The evolutionary dynamics of within-generation immune priming in invertebrate hosts. J R Soc Interfce 10:20120887. https://doi.org/10.1098/rsif.2012.0887
Bryning GP, Chambers J, Wakefield ME (2005) Identification of a sex pheromone from male yellow mealworm beetles, Tenebrio molitor. J Chem Ecol 31:2721–2730. https://doi.org/10.1007/s10886-005-7622-x
Carazo P, Sanchez E, Font E, Desfilis E (2004) Chemosensory cues allow male Tenebrio molitor beetles to assess the reproductive status of potential mates. Anim Behav 68:123–129. https://doi.org/10.1016/j.anbehav.2003.10.014
Castro-Vargas C, Linares-López C, López-Torres A, Wrobel K, Torres-Guzmán JC, Hernández GAG, Wrobel K, Lanz-Mendoza H, Contreras-Garduño J (2017) Methylation on RNA: a potential mechanism related to immune priming within but not across generations. Front Microbiol 8:473. https://doi.org/10.3389/fmicb.2017.00473
Cole TJ, Eggleston P, Hurd H (2003) Juvenile hormone titre and egg production in Tenebrio molitor infected by Hymenolepis diminuta: effect of male and/or female infection, male age and mating. J Insect Physiol 49:583–590. https://doi.org/10.1016/S0022-1910(03)00030-1
Contreras-Garduño J, Buzatto BA, Serrano-Meneses MA, Nájera-Cordero K, Córdoba-Aguilar A (2008) The size of the red wing spot of the American rubyspot as a heightened condition-dependent ornament. Behav Ecol 19:724–732. https://doi.org/10.1093/beheco/arn026
Contreras-Garduño J, Lanz H, Franco B, Pedraza-Reyes M, Nava A, Canales-Lazcano J (2016) Insect immune priming: ecology and experimental evidence. Ecol Entomol 41:351–366. https://doi.org/10.1111/een.12300
Contreras-Garduño J, Rodríguez MC, Martínez SH, Barnetche JM, Alvarado A, Izquierdo J, Herrera-Ortiz A, Moreno-García M, Velazquez-Meza ME, Valverde V, Argotte-Ramos R, Rodríguez MH, Lanz-Mendoza H (2015) Plasmodium berghei induced priming in Anopheles albimanus independently of bacterial co-infection. Dev Comp Immunol 52:172–181. https://doi.org/10.1016/j.dci.2015.05.004
Contreras-Garduño J, Rodríguez MC, Rodríguez MH, Alvarado-Delgado A, Lanz-Mendoza H (2014) Cost of immune priming within generations: trade-off between infection and reproduction. Microbes Infect 16:261–267. https://doi.org/10.1016/j.micinf.2013.11.010
Demas GE, Greives T, Chester E, French S (2012) The energetics of immunity: mechanisms mediating trade-offs in ecoimmunology. In: Demas GE, Nelson RJ (eds.). Ecoimmunology. Oxford, New York: Oxford University Press. pp 259-296
Deruytter D, Coudron CL, Teerlinck S (2019) Influence of crate size, oviposition time, number of adults and cannibalism on the reproduction of Tenebrio molitor. J Insects Food Feed 5:1–10. https://doi.org/10.3920/JIFF2019.0018
Dhinaut J, Chogne M, Moret Y (2018) Immune priming specificity within and across generations reveals the range of pathogens affecting evolution of immunity in an insect. J Anim Ecol 87:448–463. https://doi.org/10.1111/1365-2656.12661
Duneau DF, Lazzaro BP (2018) Persistence of an extracellular systemic infection across metamorphosis in a holometabolous insect. Biol Lett 14:20170771. https://doi.org/10.1098/rsbl.2017.0771
Freitak D, Schmidtberg H, Dickel F, Lochnit G, Vogel H, Vilcinskas A (2014) The maternal transfer of bacteria can mediate trans-generational immune priming in insects. Virulence 5:547–554. https://doi.org/10.4161/viru.28367
Haine ER, Pollitt LC, Moret Y, Siva-Jothy MT, Rolff J (2008) Temporal patterns in immune responses to a range of microbial insults (Tenebrio molitor). J Insect Physiol 54:1090–1097. https://doi.org/10.1016/j.jinsphys.2008.04.013
Hurd H, Parry G (1991) Metacestode-induced depression of the production of, and response to, sex pheromone in the intermediate host Tenebrio molitor. J Invertebr Pathol 58:82–87. https://doi.org/10.1016/0022-2011(91)90165-M
Ichikawa T, Kurauchi T (2009) Larval cannibalism and pupal defense against cannibalism in two species of Tenebrionid beetles. Zool Sci 26:525–530. https://doi.org/10.2108/zsj.26.525
Johnston PR, Makarova O, Rolff J (2014) Inducible defenses stay up late: temporal patterns of immune gene expression in Tenebrio molitor. G3 4:947–955. https://doi.org/10.1534/g3.113.008516
Khan I, Prakash A, Agashe D (2019) Pathogen susceptibility and fitness costs explain variation in immune priming across natural populations of flour beetles. J Anim Ecol 88:1332–1342. https://doi.org/10.1111/1365-2656.13030
Kivleniece I, Krams I, Daukšte J, Krama T, Rantala MJ (2010) Sexual attractiveness of immune-challenged male mealworm beetles suggests terminal investment in reproduction. Anim Behav 80:1015–1021. https://doi.org/10.1016/j.anbehav.2010.09.004
Knorr E, Schmidtberg H, Arslan D, Bingsohn L, Vilcinskas A (2015) Translocation of bacteria from the gut to the eggs triggers maternal transgenerational immune priming in Tribolium castaneum. Biol Lett 11:20150885. https://doi.org/10.1098/rsbl.2015.0885
Koella JC, Boëte C (2002) A genetic correlation between age at pupation and melanization immune response of the yellow fever mosquito Aedes aegypti. Evolution 56:1074–1079. https://doi.org/10.1111/j.0014-3820.2002.tb01419.x
Krams IA, Krama T, Moore FR, Rantala MJ, Mänd R, Mierauskas P, Mänd M (2015) Resource availability as a proxy for terminal investment in a beetle. Oecologia 178:339–345. https://doi.org/10.1007/s00442-014-3210-5
Krams I, Kivleniece I, Kuusik A, Krama T, Freeberg TM, Mänd R, Sivacova L, Rantala MJ, Mänd M (2014) High repeatability of anti-predator responses and resting metabolic rate in a beetle. J Insect Behav 27:57–66. https://doi.org/10.1007/s10905-013-9408-2
Krams I, Daukšte J, Kivleniece I, Krama T, Rantala MJ, Ramey G, Šauša L (2011) Female choice reveals terminal investment in male mealworm beetles, Tenebrio molitor, after a repeated activation of the immune system. J Insect Sci 11:56. https://doi.org/10.1673/031.011.5601
Kurtz J, Franz K (2003) Innate defence: evidence for memory in invertebrate immunity. Nature 425:37–38. https://doi.org/10.1038/425037a
Lanz-Mendoza H, Contreras-Garduño J (2018) Insect innate immune memory. In: Cooper EL (ed) Advances in comparative immunology. Springer, LA CA USA, pp 193–211. https://doi.org/10.1007/978-3-319-76768-0_9
Lawniczak MKN, Barnes AI, Linklater JR, Boone JM, Wigby S, Chapman T (2007) Mating and immunity in invertebrates. Trends Ecol Evol 22:48–55. https://doi.org/10.1016/j.tree.2006.09.012
Li L, Stasiak M, Li L, Xie B, Fu Y, Gidzinski D, Dixon M, Liu H (2016) Rearing Tenebrio molitor in BLSS: Dietary fiber affects larval growth, development, and respiration characteristics. Acta Astronaut. 118:130–136. https://doi.org/10.1016/j.actaastro.2015.10.003
Little TJ, Kraaijeveld AR (2004) Ecological and evolutionary implications of immunological priming in invertebrates. Trends Ecol Evol 19:58–60. https://doi.org/10.1016/j.tree.2003.11.011
Lopez S (1998) Acquired resistance affects male sexual display and female choice in guppies. Proc R Soc B 265:717–723. https://doi.org/10.1098/rspb.1998.0352
Márquez-García A, Canales-Lazcano J, Rantala M, Contreras-Garduño J (2016) Is juvenile hormone a potential mechanism that underlay the “branched Y-model”? Gen Comp Endocrinol 230:170–176. https://doi.org/10.1016/j.ygcen.2016.03.027
Medina-Gómez H, Adame G, Hernández-Quintero A, González A, Guzmán JCT, Mendoza HL, Contreras-Garduño J (2018a) The occurrence of immune priming can be species-specific in entomopathogens. Microb Pathol 118:361–364. https://doi.org/10.1016/j.micpath.2018.03.063
Medina-Gómez H, Farriols M, Santos F, González-Hernández A, Torres-Guzmán JC, Lanz H, Contreras-Garduño J (2018b) Pathogen-produced catalase affects immune priming: a potential pathogen strategy. Microb Pathol 125:93–95. https://doi.org/10.1016/j.micpath.2018.09.012
Metcalfe NB, Monaghan P (2001) Compensation for a bad start: grow now, pay later? Trends Ecol Evol 16:254–260. https://doi.org/10.1016/S0169-5347(01)02124-3
Milutinović B, Kurtz J (2016) Immune memory in invertebrates. Semin Immunol 28:328–342. https://doi.org/10.1016/j.smim.2016.05.004
Nielsen ML, Holman L (2012) Terminal investment in multiple sexual signals: immune challenged males produce more attractive pheromones. Funct Ecol 26:20–28. https://doi.org/10.1111/j.1365-2435.2011.01914.x
Rantala MJ, Jokinen I, Kortet R, Vainikka A, Suhonen J (2002) Do pheromones reveal immunocompetence? Proc R Soc B 269:1681–1685. https://doi.org/10.1098/rspb.2002.2056
Rantala MJ, Kortet R, Kotiaho JS, Vainikka A, Suhonen J (2003a) Condition dependence of pheromones and immune function in the grain beetle Tenebrio molitor. Funct Ecol 17:534–540. https://doi.org/10.1046/j.1365-2435.2003.00764.x
Rantala MJ, Vainikka A, Kortet R (2003b) The role of juvenile hormone in immune function and pheromone production trade-offs: a test of the immunocompetence handicap principle. Proc R Soc B 270:2257–2261. https://doi.org/10.1098/rspb.2003.2472
Rohner GB, Snape MD, Kelly DF, John T, Morant A, Yu LM, Borkowski A, Ceddia F, Borrow R, Siegrist CA, Pollard AJ (2008) The magnitude of the antibody and memory B cell responses during priming with a protein-polysaccharide conjugate vaccine in human infants is associated with the persistence of antibody and the intensity of booster response. J Immunol 180:2165–2173. https://doi.org/10.4049/jimmunol.180.4.2165
Roth O, Sadd BM, Schmid-Hempel P, Kurtz J (2009) Strain-specific priming of resistance in the red flour beetle, Tribolium castaneum. Proc R Soc B 276:145–151. https://doi.org/10.1098/rspb.2008.1157
Sadd BM, Schmid-Hempel P (2009) A distinct infection cost associated with trans-generational priming of antibacterial immunity in bumble-bees. Biol Lett 5:798–801. https://doi.org/10.1098/rsbl.2009.0458
Sadd B, Holman L, Armitage H, Lock F, Marland R, Siva-Jothy MT (2006) Modulation of sexual signalling by immune challenged male mealworm beetles (Tenebrio molitor, L.): evidence for terminal investment and dishonesty. J Evol Biol 19:321–325. https://doi.org/10.1111/j.1420-9101.2005.01062.x
Schmid-Hempel P (2011) Evolutionary parasitology: the integrated study of infections, immunology, ecology, and genetics. Oxford, New York: Oxford University Press
Stearns SC (2000) Life history evolution: successes, limitations, and prospects. Naturwissenschaften 87:476–486. https://doi.org/10.1007/s001140050763
Stearns SC (1992) The Evolution of Life Histories. Oxford, New York: Oxford University Press
Stearns SC (1989) Trade-offs in life-history evolution. Funct Ecol 3:259–268. https://doi.org/10.2307/2389364
Tanaka Y, Honda H, Ohsawa K, Yamamoto I (1986) A sex attractant of the yellow mealworm, Tenebrio molitor L., and its role in the mating behavior. J Pest Sci 11:49–55. https://doi.org/10.1584/jpestics.11.49
Tate AT (2016) The interaction of immune priming with different modes of disease transmission. Front Microbiol 7:1102. https://doi.org/10.3389/fmicb.2016.01102
Tate AT, Rudolf VH (2012) Impact of life stage specific immune priming on invertebrate disease dynamics. Oikos 121:1083–1092. https://doi.org/10.1111/j.1600-0706.2011.19725.x
Thomas AT, Rudolf VHW (2012) Immune priming across life stages and generations: implications for infectious disease dynamics in insects. Oikos 121:1083–1092. https://doi.org/10.1111/j.1600-0706.2011.19725.x
Trauer U, Hilker M (2013) Parental legacy in insects: variation of transgenerational immune priming during offspring development. PLoS One 8:e63392. https://doi.org/10.1371/journal.pone.0063392
Tschinkel W, Willson C, Bern HA (1967) Sex pheromone of the mealworm beetle (Tenebrio molitor). J Exp Zool 164:81–85. https://doi.org/10.1002/jez.1401640108
Vainikka A, Rantala MJ, Seppälä O, Suhonen J (2007) Do male mealworm beetles, Tenebrio molitor, sustain the honesty of pheromone signals under immune challenge? Acta Ethol 10:63–72. https://doi.org/10.1007/s10211-007-0031-0
Vainikka A, Seppälä O, Löytynoja K, Rantala MJ (2006) Fitness consequences of female preference for male pheromones in Tenebrio molitor. Evol Ecol Res 8:943–957
Valtonen TM, Rantala MJ (2012) Poor early nutrition reveals the rrade-off between immune defense and mating success. Ecol Parasitol Immunol 1:1–7. https://doi.org/10.4303/epi/235523
Vanderwel D, Bacala CH, Bacala R, Currie RW (2017) Clarification of the role of 4-methylnonanol, female-produced sex pheromone of the yellow mealworm beetle, Tenebrio molitor (Coleoptera: Tenebrionidae). J Stored Prod Res 70:60–64. https://doi.org/10.1016/j.jspr.2016.12.001
Worden BD, Parker PG, Pappas PW (2000) Parasites reduce attractiveness and reproductive success in male grain beetles. Anim Behav 59:543–550. https://doi.org/10.1006/anbe.1999.1368
Worden BD, Parker PG (2005) Females prefer noninfected males as mates in the grain beetle Tenebrio molitor: evidence in pre-and postcopulatory behaviours. Anim Behav 70:1047–1053. https://doi.org/10.1016/j.anbehav.2005.01.023
Zanchi C, Troussard J-P, Martinaud G, Moreau J, Moret Y (2011) Differential expression and costs between maternally and paternally derived immune priming for offspring in an insect. J Anim Ecol 80:1174–1183. https://doi.org/10.1111/j.1365-2656.2011.01872.x
Acknowledgments
Three anonymous reviewers and editors substantially improved this paper. Ana Guerrero Garcia y Guillermo Hernández Robles provided help with some experiments. T.T.M.L acknowledges the support by the Posgrado en Ciencias Biológicas, UNAM, México.
Funding
The study received funding from PAPIIT (IA207716 and IA205318), UNAM.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The work has been carried out under the ethical standards of the Ethical commission in research of the ENES, UNAM, Morelia, México.
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by: Paula Roig Boixeda
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Contreras-Garduño, J., Méndez-López, T.T., Patiño-Morales, A. et al. The costs of the immune memory within generations. Sci Nat 106, 59 (2019). https://doi.org/10.1007/s00114-019-1657-2
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00114-019-1657-2