Abstract
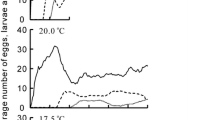
Temperature is a major driver of biological phenomena, from metabolism to ecological interactions and rates of evolutionary diversification. However, species vary greatly in their thermal tolerance, as well as the temperature under which they perform best. This study aimed to investigate the effect of experimental manipulation of environmental temperatures on the individual mortality and phenotypic composition of colonies of Melipona interrupta. To fulfill these objectives, 30 colonies in equivalent developmental conditions were artificially subjected to different temperatures. Temperatures were monitored by thermo-hygrometers, and immature mortality and sex and caste ratios were observed in brood combs during 14 months. A strong effect of external temperature on immatures was detected on deviations from 28 to 30 °C (the natural average temperature inside the colony), causing an increase in mortality. Likewise, a significant effect of temperature on sex ratio was detected, with male:female ratio decreasing at temperatures below and above 28–30 °C. Lastly, there was no clear evidence for an effect of temperature on caste ratio, although queens appeared to become relatively more frequent at warmer temperatures. The results of this study allow us to conclude that anthropogenic changes, whose effect can be extrapolated to the similar natural changes, that modify the environmental temperatures to which M. interrupta colonies are exposed are likely to compromise their survival, mainly through individual mortality.




Similar content being viewed by others
References
Allen AP, Gillooly JF, Savage VM, Brown JH (2006) Kinetic effects of temperature on rates of genetic divergence and speciation. Proc Natl Acad Sci U S A 103(24):9130–9135. https://doi.org/10.1073/pnas.0603587103
Barbosa-Costa K, Carvalho-Zilse GA (2013) Processo de oviposição da abelha da Amazônia ScaptotrigonaxanthotrichaMoure, 1950. In: Bermúdez EGC, Teles BR, Keppler RF (eds) Entomologia na Amazônia brasileira, 1st edn. Editora INPA, Manaus, pp 91–106
Bellard C, Bertelsmeier C, Leadley P, Thuiller W, Courchamp F (2012) Impacts of climate change on the future of biodiversity. Ecol Lett 15:365–377. https://doi.org/10.1111/j.1461-0248.2011.01736.x
Brito DV, Nunes RA, Pequeno PACL, Nunes-Silva CG, Carvalho-Zilse GA (2013) Differential environmental effects on caste allocation in two Amazonian Melipona bees. Apidologie 44(6):666–672. https://doi.org/10.1007/s13592-013-0215-8
Brito DV, Silva CGN, Hasselmann M, Viana LS, Astolfi-Filho S, Carvalho-Zilse GA (2015) Molecular characterization of the gene feminizer in the stingless bee Meliponainterrupta (Hymenoptera: Apidae) reveals association to sex and caste development. Insect Biochem Mol Biol 66:24–30
Brosi BJ, Daily GC, Shih TM, Oviedo F, Durán G (2007) The effects of forest fragmentation on bee communities in tropical countryside. J Appl Ecol 45(3):773–783. https://doi.org/10.1111/j.1365-2664.2007.01412.x
Brosi BJ (2009) The complex responses of social stingless bees (Apidae: Meliponini) to tropical deforestation. For Ecol Manag 258(9):1830–1837. https://doi.org/10.1016/j.foreco.2009.02.025
Brown J, Gillooly J, Allen A, Savage V, West G (2004) Toward a metabolic theory of ecology. Ecology 85(7):1771–1789 Retrieved from:http://www.esajournals.org/doi/abs/10.1890/03-9000
Brown JC, Albrecht C (2001) The effect of tropical deforestation on stingless bees of the genus Melipona (Insecta: Hymenoptera: Apidae: Meliponini) in central Rondonia, Brazil. J Biogeogr 28:623–634. https://doi.org/10.1046/j.1365-2699.2001.00583.x
Carmargo JMF, Kerr WE, Lopes CR (1967) Morfologia externa de Melipona (Melipona) marginata Lepeletier (Hymenoptera, Apidae). Pap Avulsos Zool 20:229–258
Carvalho-Zilse GA, Vilas-Boas HC, Costa KB, Nunes-Silva CG, Souza MT, Fernandes RS (2012) Meliponicultura na Amazônia. INPA, Manaus
Chown S, Hoffmann A, Kristensen T, Angilletta M, Stenseth N, Pertoldi C (2010) Adapting to climate change: a perspective from evolutionary physiology. Clim Res 43(1):3–15. https://doi.org/10.3354/cr00879
Clusella-Trullas S, Blackburn TM, Chown SL (2011) Climatic predictors of temperature performance curve parameters in ectotherms imply complex responses to climate change. Am Nat 177(6):738–751. https://doi.org/10.1086/660021
Couto RM, Camillo E (2007) Influence of temperature on the immatures mortality of Centris (Heterocentris) analis (Hymenoptera, Apidae, Centridini). Iheringia, Sér Zool 97(1):51–55. https://doi.org/10.1590/S0073-47212007000100008
DeFries RS, Rudel T, Uriarte M, Hansen M (2010) Deforestation driven by urban population growth and agricultural trade in the twenty-first century. Nat Geosci 3:178–181. https://doi.org/10.1038/ngeo756
Deutsch C A, Tewksbury J J, Huey R B, Sheldon K S, Ghalambor C K, Haak D C, Martin P R (2008) Impacts of climate warming on terrestrial ectotherms across latitude. Proc Natl Acad Sci U S A. Retrieved from: http://www.pnas.org/content/105/18/6668.short
Engels W, Rosenkranz P, Engels E (1995) Thermoregulation in the nest of the Neotropical stingless bee Scaptotrigonapostiça and a hypothesis on the evolution of temperature homeostasis in highly eusocial bees. Stud Neotrop Fauna E30(4):193–205. https://doi.org/10.1080/01650529509360958
Francini IB, Nunes-Silva CG, Carvalho-ZilseG A (2012) Diploid male production of two Amazonian Melipona bees (Hymenoptera: Apidae). Psyche 1:1–7
Heard TA (1999) The role of stingless bees. Annu Ver Entomol 44:183–206. https://doi.org/10.1146/annurev.ento.44.1.183
INMET (2014) http://www.inmet.gov.br. Accessed 5 Nov 2014
IPCC (2013) Climate change 2013: the physical science basis. Contribution of working group I to the fifth assessment report of the intergovernmental panel on climate change. Stocker T F, Qin D, Plattner G K, Tignor M, Allen S K, Boschung J, Nauels a, Xia Y, Bex V, Midgley P M (ed). Cambridge University press, Cambridge, 1535p
Jones J, Oldroyd B (2006) Nest thermoregulation in social insects. Adv Insect Physiol 33(06):153–191. https://doi.org/10.1016/S0065-2806(06)33003-2
Kerr WE (1950) Genetic determination of castes in the genus Melipona. Genetics 35(2):143–152 Retrieved from: http://www.pubmedcentral.nih.gov/articlerender.fcgi?artid=1209477&tool=pmcentrez&rendertype=abstract
Kerr WE (1974) Sex determination in bees. III Caste determination and genetic control in Melipona. Insect Soc 21:357–368
Kerr WE (1996) Biologia e manejo da tiúba: a abelha do Maranhão. EDUFMA, São Luis
Kerr WE (1997) Sex determination in honey bees (Apinae and Meliponinae) and its consequences. Braz J Genet 20:601–611
Kerr WE, Carvalho GA, Nascimento VA (1996) Abelha Uruçu: Biologia, Manejo e Conservação. Fundação Acangaú, Belo Horizonte
Kerr WE, Carvalho GA, Coletto-Silva A, Assis MGP (2001) Aspectos Pouco Mencionados da Biodiversidade Amazônica.InBiodiversidade, Pesquisa e Desenvolvimento na Amazônia. Parcerias Estratégicas, Ministério da Ciência e Tecnologia 12:20–41
Kerr WE, Stort AC, Montenegro MJ (1966) Importância de alguns fatores ambientais na determinação das castas no gênero Melipona. An Acad Bras Ciênc 38(1):151–168
Lopes MTR, Barbosa AL, Neto JMV, Pereira FM, Camargo RCR, Ribeiro VQ, Souza BA (2011) Alternativas de sombreamento para apiários. Pesquisa Agropecuária Tropical 41(3):299–305. https://doi.org/10.5216/pat.v41i3.8919
Mittelbach GG, Schemske DW, Cornell HV, Allen AP, Brown JM, Bush MB, Harrison SP, Hurlbert AH, Knowlton N, Lessios H, McCain CM, McCune AR, McDade L, McPeek M, Near TJ, Price TD, Ricklefs RE, Roy K, Sax DF, Schluter D, Sobel JM, Turelli M (2007) Evolution and the latitudinal diversity gradient: speciation, extinction and biogeography. Ecol Lett 10(4):315–331. https://doi.org/10.1111/j.1461-0248.2007.01020.x
Nogueira-Neto P (1997) Vida e criação de abelhas indígenas sem ferrão. Editora Nogueirapis, São Paulo
Nunes-Silva CG, Kerr WE, Bonetti AM, Carvalho-Zilse GA (2006) Effect of juvenile hormone III and heat shock in caste determinations in Melipona scutellaris Latreille, 1811 (Hymenoptera, Apidae). Magistra 18(4):277–280
Palmer G, Platts PJ, Brereton T, Chapman JW, Dytham C, Fox R, Pearce-Higgins J, Roy DB, Hill JK, Thomas CD (2017) Climate change, climatic variation and extreme biological responses. Phil Trans R Soc B 372:20160144. https://doi.org/10.1098/rstb.2016.0144
Parmesan C (2006) Ecological and evolutionary responses to recent climate change. Annu Rev Ecol Evol Syst 37(2006):637–669. https://doi.org/10.2307/annurev.ecolsys.37.091305.30000024
Pereboom JJM, Biesmeijer JC (2003) Thermal constraints for stingless bee foragers: the importance of body size and coloration. Oecologia 137(1):42–50. https://doi.org/10.1007/s00442-003-1324-2
Development Core Team R (2013) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna ISBN 3-900051-07-0, http://www.R-project.org/
Roubik DW (2006) Stingless bee nesting biology. Apidologie 37:124–143. https://doi.org/10.1051/apido:2006026
Roubik D, Peralta F (1983) Thermodynamics in nests of two Melipona species in Brasil. Acta Amazon 13(2):453–466 Retrieved from: http://agris.fao.org/agris-search/search.do?recordID=US201302048790
Settele J, Bishop J, Potts SG (2016) Climate change impacts on pollination. Nature plants 2(7):16092. https://doi.org/10.1126/science.aaa4984
Urban MC (2015) Accelerating extinction risk from climate change. Science 348(6234):571–573. https://doi.org/10.1038/nplants.2016.9210.1038/nplants.2016.92
Whiting, P W (1943) Multiple Alleles in complementary sex determination of Habrobracon. Genetics 28(5):365–382
Wood SN (2006) Generalized additive models: an introduction with R. Chapman and Hall/CRC
Zuur AF, Ieno EM, Walker NJ, Saveliev AA, Smith GM (2009) Mixed effects models and extensions in ecology with R. Springer, New York
Acknowledgments
We are grateful to the anonymous referees for the constructive suggestions. We acknowledge the Coordenação de Aperfeiçoamento de Pessoal de Nível Superior (CAPES) for a MSc scholarship to the first author and the Instituto Nacional de Pesquisas da Amazônia (INPA), Graduate Program in Entomology (PPG-Entomologia) for the logistical support.
Funding
We thank the sponsoring agencies FAPEAM and CNPq and CENBAM (Instituto Nacional de Ciência e Tecnologia-Centro de Estudos Integrados da Biodiversidade Amazônica) for the financial support.
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by: Sven Thatje
Electronic supplementary material
ESM 1
(DOCX 43 kb)
Rights and permissions
About this article
Cite this article
Becker, T., Pequeno, P.A.C.L. & Carvalho-Zilse, G.A. Impact of environmental temperatures on mortality, sex and caste ratios in Melipona interrupta Latreille (Hymenoptera, Apidae). Sci Nat 105, 55 (2018). https://doi.org/10.1007/s00114-018-1577-6
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00114-018-1577-6