Abstract
The selective forces imposed by primary receivers and unintended eavesdroppers of animal signals often act in opposite directions, constraining the development of conspicuous coloration. Because iridescent colours change their chromatic properties with viewer angle, iridescence offers a potential mechanism to relax this trade-off when the relevant observers involved in the evolution of signal design adopt different viewer geometries. We used reflectance spectrophotometry and visual modelling to test if the striking blue head coloration of males of the lizard Lacerta schreibeiri (1) is iridescent and (2) is more conspicuous when viewed from the perspective of conspecifics than from that of the main predators of adult L. schreibeiri (raptors). We demonstrate that the blue heads of L. schreiberi show angle-dependent changes in their chromatic properties. This variation allows the blue heads to be relatively conspicuous to conspecific viewers located in the same horizontal plane as the sender, while simultaneously being relatively cryptic to birds that see it from above. This study is the first to suggest the use of angle-dependent chromatic signals in lizards, and provides the first evidence of the adaptive function of iridescent coloration based on its detectability to different observers.





Similar content being viewed by others
References
Andersson S, Prager M (2006) Quantification of coloration. In: Hill GE, McGraw KJ (eds) Bird Coloration, Vol. 1. Mechanisms and Measurements. Harvard University Press, Cambridge, pp 41–89
Bajer K, Molnár O, Török J, Herczeg G (2010) Female European green lizards (Lacerta viridis) prefer males with high ultraviolet throat reflectance. Behav Ecol Sociobiol 64:2007–2014. doi:10.1007/s00265-010-1012-2
Bajer K, Molnár O, Török J, Herczeg G (2011) Ultraviolet nuptial colour determines fight success in male European green lizards (Lacerta viridis). Biol Lett 7:866–868. doi:10.1098/rsbl.2011.0520
Bradbury JW, Vehrencamp SL (2011) Principles of animal communication, 2nd edn. Sinauer Assoc Press, Sunderland
Csermely D, Bonati B, Romani R (2009) Predatory behaviour of common kestrels (Falco tinnunculus) in the wild. J Ethol 27:461–465. doi:10.1007/s10164-008-0143-7
Cummings ME, Rosenthal GG, Ryan MJ (2003) A private ultraviolet channel in visual communication. P Roy Soc Lond B 270:897–904. doi:10.1098/rspb.2003.2334
Doucet SM, Meadows MG (2009) Iridescence: a functional perspective. J R Soc Interface 6:S115–S132. doi:10.1098/rsif.2008.0395.focus
Eliason CM, Shawkey MD (2011) Decreased hydrophobicity of iridescent feathers: a potential cost of shiny plumage. J Evol Biol 214:2157–2163. doi:10.1242/jeb.055822
Endler JA (1991) Variation in the appearance of guppy color patterns to guppies and their predators under different visual conditions. Vision Res 31:587–608
Endler JA, Mielke PW (2005) Comparing entire colour patterns as birds see them. Biol J Linn Soc 86:405–431. doi:10.1111/j.1095-8312.2005.00540.x
Endler JA, Théry M (1996) Interacting effects of lek placement, display behavior, ambient light, and color patterns in three neotropical forest-dwelling birds. Am Nat 148:421–452. doi:10.1086/285934
Fleishman LJ (1992) The influence of the sensory system and the environment on motion patterns in the visual displays of anoline lizards and other vertebrates. Am Nat 139:S36–S61
Fleishman LJ, Leal M, Sheehan J (2006) Illumination geometry, detector position and the objective determination of animal signal colours in natural light. Anim Behav 71:463–474. doi:10.1016/j.anbehav.2005.06.005
Fleishman LJ, Loew ER, Whiting MJ (2011) High sensitivity to short wavelengths in a lizard and implications for understanding the evolution of visual systems in lizards. Proc R Soc B 278:2891–2899. doi:10.1098/rspb.2011.0118
Font E, Rome LC (1990) Functional morphology of dewlap extension in the lizard Anolis equestris (Iguanidae). J Morph 206:245–258. doi:10.1002/jmor.1052060210
Font E, Pérez i de Lanuza G, Sampedro C (2009) Ultraviolet reflectance and cryptic sexual dichromatism in the ocellated lizard, Lacerta (Timon) lepida (Squamata: Lacertidae). Biol J Linn Soc 97:766–780. doi:10.1111/j.1095-8312.2009.01251.x
Griggio M, Serra L, Licheri D, Campomori C, Pilastro A (2009) Moult speed affects structural feather ornaments in the blue tit. J Evol Biol 22:782–792. doi:10.1111/j.1420-9101.2009.01700.x
Guilford T, Dawkins MS (1991) Receiver psychology and the evolution of animal signals. Anim Behav 42:1–14
Hamilton DG, Whiting MJ, Pryke SR (2013) Fiery frills: carotenoid-based coloration predicts contest success in frillneck lizards. Behav Ecol 24:1138–1149. doi:10.1093/beheco/art041
Kemp DJ (2008) Female mating biases for bright ultraviolet iridescence in the butterfly Eurema hecabe (Pieridae). Behav Ecol 19:1–8. doi:10.1093/beheco/arm094
Kemp DJ, Rutowski RL (2007) Condition dependence, quantitative genetics, and the potential signal content of iridescent ultraviolet butterfly coloration. Evolution 61:168–183. doi:10.1111/j.1558-5646.2007.00014.x
Kemp DJ, Herberstein ME, Grether GF (2012) Unraveling the true complexity of costly color signaling. Behav Ecol 23:233–236. doi:10.1093/beheco/arr153
Kemp DJ, Jones D, Macedonia JM, Krockenberger AK (2014) Female mating preferences and male signal variation in iridescent Hypolimnas butterflies. Anim Behav 87:221–229. doi:10.1016/j.anbehav.2013.11.001
Keyser AJ, Hill GE (1999) Condition-dependent variation in the blue-ultraviolet coloration of a structurally based plumage ornament. P Roy Soc Lond B 266:771–777
Land MF (1972) The physics and biology of animal reflectors. Prog Biophys Mol Biol 24:75–106
Lind O, Mitkus M, Olsson P, Kelber A (2013) Ultraviolet sensitivity and colour vision in raptor foraging. J Exp Biol 216:1819–1826. doi:10.1242/jeb.082834
Martín J, López P (2009) Multiple color signals may reveal multiple messages in male Schreiber’s green lizards, Lacerta schreiberi. Behav Ecol Sociobiol 63:1743–1755. doi:10.1007/s00265-009-0794-6
Mäthger LM, Denton EJ, Marshall NJ, Hanlon RT (2009) Mechanisms and behavioural functions of structural colouration in cephalopods. J R Soc Interface 6:S149–S163. doi:10.1098/rsif.2008.0366.focus
Meadows MG, Butler MW, Morehouse NI, Taylor LA, Toomey MB, McGraw KJ, Rutowski RL (2009) Iridescence: views from many angles. J R Soc Interface 6:S107–S113. doi:10.1098/rsif.2009.0013.focus
Meadows MG, Morehouse NI, Rutowski RL, Douglas JM, McGraw KJ (2011) Quantifying iridescent coloration in animals: a method for improving repeatability. Behav Ecol Sociobiol 65:1317–1327. doi:10.1007/s00265-010-1135-5
Meadows MG, Roudybush TE, McGraw KJ (2012) Dietary protein level affects iridescent coloration in Anna’s hummingbirds, Calypte anna. J Evol Biol 215:2742–2750. doi:10.1242/jeb.069351
Morrison RL (1995) A transmission electron microscopic (TEM) method for determining structural colors reflected by lizard iridophores. Pigm Cell Res 8:28–36
Noh H, Liew SF, Saranathan V, Mochrie SGJ, Prum RO, Dufresne ER, Cao H (2010) How noniridescent colors are generated by quasi-ordered structures of bird feathers. Adv Mater 22:2871–2880. doi:10.1002/adma.200903699
Ödeen A, Håstad O (2003) Complex distribution of avian color vision systems revealed by sequencing the SWS1 opsin from total DNA. Mol Biol Evol 20:855–861. doi:10.1093/molbev/msg108
Osorio D, Ham AD (2002) Spectral reflectance and directional properties of structural coloration in bird plumage. J Exp Biol 205:2017–2027
Pérez i de Lanuza G (2012) Visió en color i coloracions dels lacèrtids. PhD thesis, University of València, València
Pérez i de Lanuza G, Font E (2011) Lizard blues: blue body colouration and ultraviolet polychromatism in lacertids. Rev Esp Herp 24:67–84
Pérez i de Lanuza G, Font E (2014) Ultraviolet vision in lacertid lizards: evidence from retinal structure, eye transmittance, SWS1 visual pigment genes, and behaviour. J Exp Biol 217:1–11. doi:10.1242/jeb.104281
Pérez i de Lanuza G, Font E, Monterde JL (2013) Using visual modelling to study the evolution of lizard colouration: sexual selection drives the evolution of sexual dichromatism in lacertids. J Evol Biol 26:1826–1835. doi:10.1111/jeb.12185
Pérez-Mellado V (1998) Lacerta schreiberi Bedriaga, 1878. In: Salvador A (coord) Reptiles. Fauna Ibérica. Vol. 10. Museo Nacional de Ciencias Naturales, Madrid, pp 218–227
Prum RO (2006) Anatomy, physics, and evolution of avian structural colors. In: Hill GE, McGraw KJ (eds) Bird coloration, vol 1, Mechanisms and measurements. Harvard University Press, Cambridge, pp 295–355
Rohrlich ST, Porter KR (1972) Fine structural observations relating to the production of color by the iridophores of a lizard, Anolis carolinensis. J Cell Biol 53:38–52
Rowe C (2013) Receiver psychology: a receiver’s perspective. Anim Behav 85:517–523. doi:10.1016/j.anbehav.2013.01.004
Rutowski RL, Nahm AC, Macedonia JM (2010) Iridescent hindwing patches in the Pipevine Swallowtail: differences in dorsal and ventral surfaces relate to signal function and context. Funct Ecol 24:767–775. doi:10.1111/j.1365-2435.2010.01693.x
Saranathan V, Forster JD, Noh H, Liew S-F, Mochrie SGJ, Cao H, Dufresne ER, Prum RO (2012) Structure and optical function of amorphous photonic angle X-ray scattering (SAXS) analysis of 230 bird species nanostructures from avian feather barbs: a comparative small. J R Soc Interface 9:2563–2580. doi:10.1098/rsif.2012.0191
Shawkey MD, Estes AM, Sieferman L, Hill GE (2005) The anatomical basis of sexual dichromatism in non-iridescent ultraviolet-blue structural coloration of feathers. Biol J Linn Soc 84:259–271. doi:10.1111/j.1095-8312.2005.00428.x
Sicsú P, Manica LT, Maia R, Macedo RH (2013) Here comes the sun: multimodal displays are associated with sunlight incidence. Behav Ecol Sociobiol 67:1633–1642. doi:10.1007/s00265-013-1574-x
Stevens M (2013) Sensory Ecology, Behaviour, and Evolution. Oxford University Press, Oxford
Stoddard MC, Prum RO (2008) Evolution of avian plumage color in a tetrahedral color space: a phylogenetic analysis of new world buntings. Am Nat 171:755–776. doi:10.1086/587526
Stuart-Fox DM, Whiting MJ, Moussalli A (2006) Camouflage and colour change: antipredator responses to bird and snake predators across multiple populations in a dwarf chameleon. Biol J Linn Soc 88:437–446. doi:10.1111/j.1095-8312.2006.00631.x
Stuart-Fox D, Godinho R, de Bellocq JG, Irwin NR, Brito JC, Moussalli A, Široky P, Hugall AF, Baird SJE (2009) Variation in phenotype, parasite load and male competitive ability across a cryptic hybrid zone. PLoS ONE 4:e5677. doi:10.1371/journal.pone.0005677
Acknowledgments
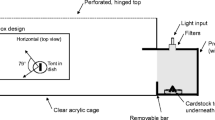
We are especially grateful to J. Heredero for the design and construction of the goniometer. We thank M. C. Stoddard for helping with TetraColorSpace, and L. J. Fleishman for generously sharing with us visual sensitivity data for Platysaurus broadleyi. This work was funded by a Portuguese FCT postdoctoral fellowship (SFRH/BPD/94582/2013) and a project from the Ministerio de Ciencia e Innovación (CGL2011-23751).
Ethical standards
The study was performed according to guidelines provided by the Association for the Study of Animal Behaviour (ASAB) and the Animal Behavior Society (ABS). The experiments complied with current EU and Spanish laws and permits were generously provided by the Sierra Norte de Guadalajara Natural Park (Junta de Castilla-la Mancha).
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by: Sven Thatje
Rights and permissions
About this article
Cite this article
Pérez i de Lanuza, G., Font, E. Now you see me, now you don’t: iridescence increases the efficacy of lizard chromatic signals. Naturwissenschaften 101, 831–837 (2014). https://doi.org/10.1007/s00114-014-1224-9
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00114-014-1224-9